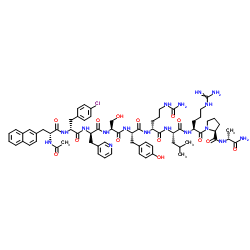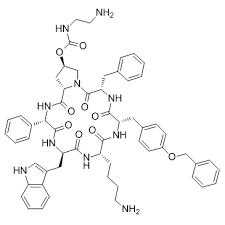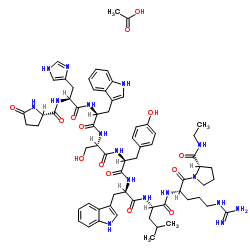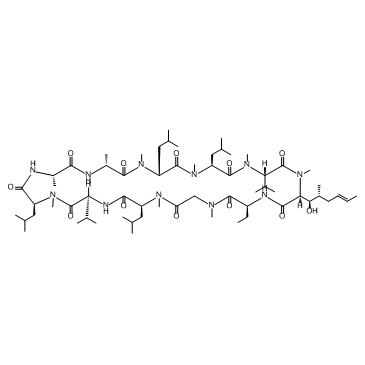
Cyclosporin A
- CAS No.59865-13-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Cyclosporin A available for pharmaceutical and research applications. Compliant manufacturing with full documentation support.
Request a Quote & Full COAProduct Technical Details
Product Overview
Cyclosporin A represents a cornerstone in the field of immunopharmacology and advanced peptide chemistry. As a cyclic nonribosomal peptide consisting of eleven amino acids, this compound is highly valued in the pharmaceutical industry for its potent immunosuppressive properties. Our facility specializes in the production and supply of high-quality Cyclosporin A, adhering to strict international quality standards to ensure consistency and reliability for downstream manufacturing processes.
We understand the critical nature of supply chain stability for active pharmaceutical ingredients. Our manufacturing processes are designed to maintain high purity levels while ensuring scalability for global demand. This product is suitable for licensed pharmaceutical manufacturers, research institutions, and development laboratories focusing on immunological therapeutics and dermatological formulations.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | Cyclosporin A |
| CAS Registry Number | 59865-13-3 |
| Molecular Formula | C62H111N11O12 |
| Molecular Weight | 1202.611 g/mol |
| Appearance | White to off-white powder |
| Assay (Purity) | ≥98.0% |
| Melting Point | 148-151°C |
| Density | 1.0±0.1 g/cm3 |
| Refractive Index | 1.468 |
Industrial Applications
The primary application of Cyclosporin A lies within the pharmaceutical sector, specifically in the formulation of medications designed to prevent organ transplant rejection. It is extensively utilized in protocols involving kidney, liver, and heart transplants. Beyond transplant medicine, this compound plays a significant role in the treatment of autoimmune conditions such as severe psoriasis and rheumatoid arthritis.
In the context of research and development, Cyclosporin A serves as a critical reference standard and active component for studying T-cell activation pathways. Its unique cyclic peptide structure makes it an valuable subject for biochemical research and the development of novel immunomodulatory agents. Our clients leverage this material for creating targeted therapies and advancing clinical research initiatives.
Quality Assurance and Compliance
Quality is the foundation of our operation. Every batch of Cyclosporin A undergoes rigorous testing using advanced analytical methods including HPLC and mass spectrometry. We provide comprehensive documentation, including Certificates of Analysis (COA), to verify identity, purity, and potency. Our production facilities operate under strict quality management systems to ensure compliance with global regulatory requirements.
- Consistent batch-to-batch reproducibility
- Full traceability of raw materials
- Comprehensive stability data available
- Support for regulatory filing documentation
Storage and Handling
To maintain the integrity of the chemical structure, proper storage conditions are essential. Cyclosporin A should be stored in a cool, ventilated environment away from direct sunlight and moisture. Containers must be kept tightly sealed when not in use to prevent degradation. We recommend following standard safety protocols for handling pharmaceutical powders, including the use of appropriate personal protective equipment during weighing and processing operations.
Packaging options are flexible to meet client needs, ranging from standard 25 kg drums to customized containers based on specific logistical requirements. Our logistics team ensures secure and timely global shipping, maintaining the cold chain or specific environmental controls where necessary to preserve product quality during transit.