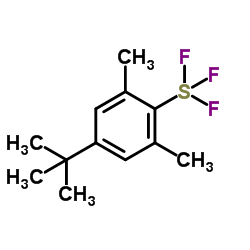
4-tert-Butyl-2,6-dimethylphenylsulfur Trifluoride
- CAS No.947725-04-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-performance fluorinating reagent for organic synthesis, offering mild reaction conditions and excellent thermal stability for pharmaceutical and material science applications.
Request Bulk PricingProduct Technical Details
Product Overview
4-tert-Butyl-2,6-dimethylphenylsulfur Trifluoride, commonly known in the industry as Fluolead, represents a significant advancement in modern organofluorine chemistry. This specialized fluorinating agent is designed to facilitate the introduction of fluorine atoms into complex organic molecules under remarkably mild conditions. Unlike traditional fluorinating reagents that may require harsh environments, this compound operates effectively between 0 degrees Celsius and room temperature, ensuring high yields while preserving sensitive functional groups within the substrate.
Our commitment to quality ensures that every batch meets stringent industrial purity standards, making it an ideal choice for research and development in pharmaceuticals, agrochemicals, and new material synthesis. The unique steric hindrance provided by the tert-butyl and dimethyl groups enhances the reagent stability, reducing the risk of premature decomposition during storage or handling.
Key Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 947725-04-4 |
| Molecular Formula | C12H17F3S |
| Molecular Weight | 250.32 g/mol |
| Density | 1.1 +/- 0.1 g/cm3 |
| Boiling Point | 92-93 degrees C (0.5 Torr) |
| Melting Point | 60-61 degrees C (pentane) |
| Refractive Index | 1.450 |
| Storage Temperature | 0-10 degrees C |
Industrial Applications
This fluorinating reagent is versatile and serves critical roles in various synthetic pathways. Its primary application involves the deoxygenative fluorination of hydroxyl and carbonyl groups, enabling chemists to convert alcohols and ketones into their corresponding fluorinated derivatives with exceptional efficiency. This capability is particularly valuable in the late-stage functionalization of drug candidates where metabolic stability is enhanced by fluorine substitution.
- Conversion of carboxyl groups (-COOH) to trifluoromethyl groups (-CF3), expanding the chemical space for medicinal chemistry.
- Fluorination of propylthioketones and thioesters, providing access to sulfur-containing fluorinated building blocks.
- Substitute for hazardous fluorinating reagents, offering a safer profile with reduced smoke generation during reactions.
- Compatible with a wide range of substrates, allowing for broad application in custom synthesis projects.
Storage and Stability
To maintain optimal performance, 4-tert-Butyl-2,6-dimethylphenylsulfur Trifluoride should be stored in a cool, dry environment between 0 and 10 degrees Celsius. The compound exhibits high thermal stability, which minimizes degradation over time and ensures consistent results across multiple production runs. Proper handling protocols should be followed to prevent exposure to moisture, which can hydrolyze the sulfur-fluorine bonds. Our packaging is designed to protect the integrity of the reagent during global shipping, ensuring it arrives ready for immediate use in your laboratory or production facility.