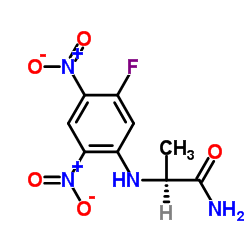
Nα-(2,4-Dinitro-5-fluorophenyl)-L-alaninamide
- CAS No.95713-52-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Marfey's reagent (FDAA) designed for precise chiral derivatization and HPLC analysis of amino acids. Ideal for pharmaceutical quality control and research laboratories requiring reliable enantiomer separation.
Request Bulk PricingProduct Technical Details
Product Overview
Nα-(2,4-Dinitro-5-fluorophenyl)-L-alaninamide, widely recognized in the analytical chemistry community as Marfey's reagent or FDAA, represents a critical tool for the stereoisomeric analysis of chiral compounds. Our manufacturing process ensures exceptional purity levels, making this reagent the preferred choice for laboratories engaged in complex pharmaceutical development and quality assurance protocols. This advanced derivatization agent facilitates the precise separation and identification of amino acid enantiomers using high-performance liquid chromatography (HPLC).
As a leading global manufacturer, we understand the stringent requirements of modern analytical workflows. Our production facilities adhere to rigorous quality management systems, guaranteeing batch-to-batch consistency. Whether you are conducting routine quality control or pioneering research into novel peptide structures, this reagent provides the reliability and sensitivity necessary for accurate results. The compound's unique chemical structure allows for stable derivative formation, ensuring long-term sample integrity during analysis.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | Nα-(2,4-Dinitro-5-fluorophenyl)-L-alaninamide |
| CAS Number | 95713-52-3 |
| Molecular Formula | C9H9FN4O5 |
| Molecular Weight | 272.19 g/mol |
| Purity | >99% |
| Appearance | Yellow Powder |
| Melting Point | 229°C |
| Storage Temperature | 2-8°C |
Industrial Applications
The primary application of this specialty chemical lies in the field of chiral analysis within the pharmaceutical and biotechnology sectors. It is extensively used for the derivatization of amino acids, amines, and peptides prior to chromatographic separation. By converting non-chromophoric analytes into highly detectable derivatives, Marfey's reagent enhances the sensitivity of UV detection systems. This capability is indispensable for determining the enantiomeric purity of drug substances, where even minor impurities can have significant biological implications.
- Pharmaceutical quality control for chiral drug intermediates.
- Research and development of peptide-based therapeutics.
- Food science analysis for amino acid profiling.
- Forensic chemistry applications requiring precise stereoisomer identification.
- Academic research focused on protein structure and function.
Quality Assurance and Storage
Maintaining the integrity of analytical reagents is paramount for obtaining reproducible data. Our product is packaged under controlled conditions to prevent moisture absorption and degradation. We recommend storing the material at refrigerated temperatures between 2°C and 8°C. Each batch is accompanied by a comprehensive Certificate of Analysis (COA), detailing purity assessments and physical constants verified through advanced spectroscopic methods. Our commitment to excellence ensures that researchers worldwide can rely on our chemicals for their most critical analytical challenges.