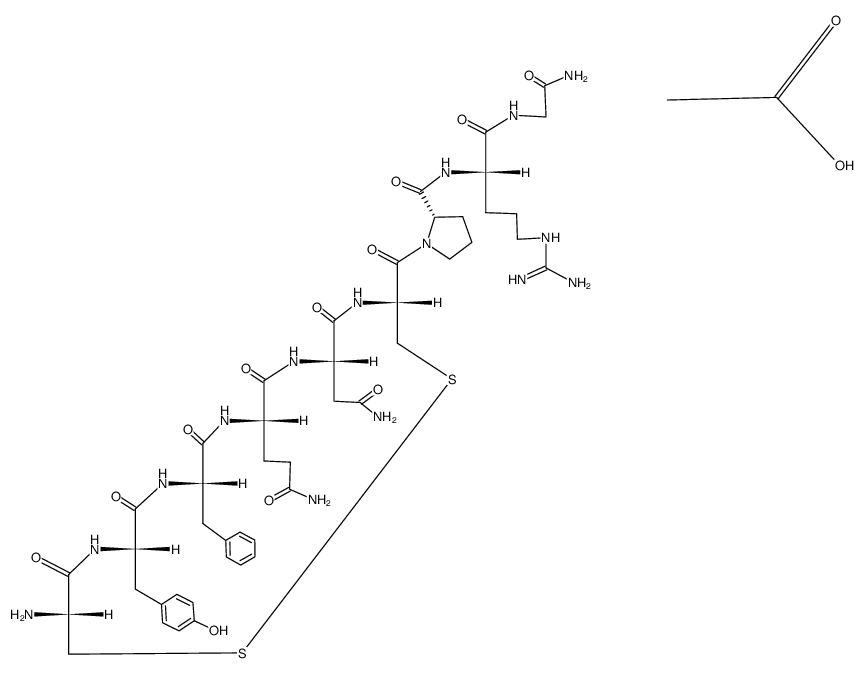
Argipressin Acetate
- CAS No.129979-57-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Argipressin Acetate (CAS 129979-57-3), a bioactive peptide derivative used in advanced cosmetic formulations for skin conditioning and neurocosmetic applications.
Request Bulk PricingProduct Technical Details
Product Overview
Argipressin Acetate is a synthetically derived, high-purity peptide compound widely recognized in the field of cosmetic science for its bioactive properties. With a molecular formula of C48H69N15O14S2 and a molecular weight of 1144.28 g/mol, this acetate salt form ensures enhanced stability and solubility for topical applications. It appears as a white to off-white powder and is manufactured under stringent quality control protocols to meet international cosmetic ingredient standards.
Specifications
| Appearance | White to off-white powder |
|---|---|
| Assay | ≥98.0% |
| Molecular Formula | C48H69N15O14S2 |
| Molecular Weight | 1144.28 g/mol |
| Packaging | 25 kg/drum; custom packaging available upon request |
| Storage | Store in a cool, dry, and well-ventilated area |
Industrial Applications
In the realm of cosmetic active ingredients, Argipressin Acetate serves as a functional biopeptide with potential neuromodulatory effects on skin physiology. While historically associated with endocrine research, its refined derivatives are now leveraged in premium skincare formulations targeting barrier support, hydration retention, and sensory comfort. As a non-hormonal cosmetic-grade material, it aligns with global regulatory frameworks for topical use in leave-on and rinse-off products.
- Suitable for neurocosmetic and anti-stress skincare formulations
- Compatible with aqueous and emulsion-based delivery systems
- Manufactured under ISO-compliant GMP conditions for cosmetic use
- Free from controlled substances and compliant with REACH, FDA, and EU Cosmetic Regulation
Our Argipressin Acetate is exclusively intended for external cosmetic applications and is not for human or veterinary therapeutic use. All batches are accompanied by a Certificate of Analysis (COA) and full regulatory documentation to support seamless integration into global product portfolios.