Tranexamic Acid
- CAS No.701-54-2
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Tranexamic Acid (CAS 701-54-2) designed for cosmetic whitening and pharmaceutical applications, ensuring superior stability and efficacy.
Request Bulk PricingProduct Technical Details
Product Overview
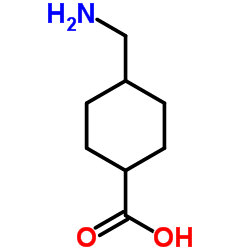
Tranexamic Acid, chemically known as Trans-4-(Aminomethyl)cyclohexanecarboxylic acid, is a high-value synthetic derivative widely recognized for its exceptional stability and functional versatility. As a leading manufacturer, we provide premium-grade material that meets stringent international standards for both pharmaceutical and cosmetic industries. Our production process ensures consistent quality, making it an ideal choice for formulators seeking reliable performance in complex matrices.
This compound functions primarily as a plasminogen activator inhibitor in medical contexts, but its ability to interfere with melanin production has made it a cornerstone ingredient in advanced skincare formulations. We specialize in supplying bulk quantities that adhere to rigorous purity specifications, supporting global supply chains with efficiency and compliance.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 701-54-2 |
| Molecular Formula | C8H15NO2 |
| Molecular Weight | 157.21 g/mol |
| Appearance | White crystalline powder |
| Assay (HPLC) | ≥99.0% |
| Melting Point | 233°C |
| Loss on Drying | ≤0.5% |
| Storage Condition | Refrigerator, cool and dry place |
Industrial Applications
In the cosmetic sector, Tranexamic Acid is highly valued for its skin-brightening properties. It effectively inhibits the interaction between melanocytes and keratinocytes, reducing hyperpigmentation and preventing the formation of age spots. It is commonly incorporated into whitening essences, moisturizing lotions, and firming dew formulations. Its stability across various pH levels allows for flexible formulation strategies without compromising efficacy.
Pharmaceutically, it serves as a critical antifibrinolytic agent used to manage excessive bleeding during surgeries or specific medical conditions. Our manufacturing capabilities support both sectors with dedicated production lines that prevent cross-contamination, ensuring safety and regulatory compliance for diverse end-uses.
Quality Assurance and Safety
We maintain a robust quality management system aligned with GMP standards. Every batch undergoes comprehensive testing, including identity confirmation, purity analysis, and residual solvent checks. We provide a complete Certificate of Analysis (COA) with each shipment, guaranteeing transparency and traceability. Our commitment to safety extends to packaging, which is designed to protect the integrity of the product during international transit.
Packaging and Storage
Standard packaging includes 25kg fiber drums with inner polyethylene bags, ensuring moisture protection and ease of handling. Custom packaging solutions are available upon request to meet specific logistical requirements. For optimal stability, the product should be stored in a cool, well-ventilated area away from direct sunlight and heat sources. Containers must remain tightly closed when not in use to prevent absorption of atmospheric moisture.