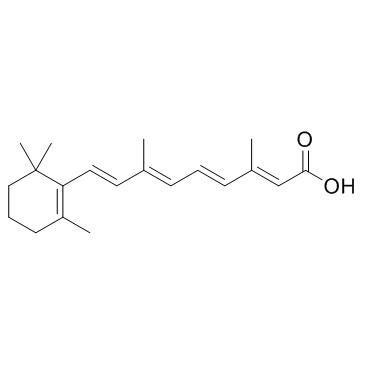
Tretinoin
- CAS No.302-79-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Tretinoin (Vitamin A Acid) for pharmaceutical and cosmetic formulations, offering proven efficacy in skin renewal and acne treatment.
Request Bulk PricingProduct Technical Details
Product Overview
Tretinoin, scientifically identified as all-trans-retinoic acid, stands as a pivotal active ingredient within the global pharmaceutical and cosmetic sectors. As a metabolite of Vitamin A, this compound is highly valued for its ability to influence cellular differentiation and proliferation. Our organization specializes in the supply of high-purity Tretinoin, catering to licensed manufacturers who require reliable raw materials for product development. We maintain rigorous quality control protocols to ensure that every batch meets international standards for purity and stability.
This chemical substance is essential for the creation of advanced topical formulations aimed at skin renewal. By providing consistent quality, we enable our partners to develop effective solutions for various dermatological concerns. Our manufacturing processes are designed to minimize impurities, ensuring that the final material is suitable for sensitive applications where safety and efficacy are paramount. We understand the critical nature of supply chain reliability in the chemical industry and strive to deliver on time without compromising specifications.
Technical Specifications
Quality assurance is the foundation of our chemical supply service. Each production lot of Tretinoin undergoes extensive analytical testing to verify its chemical identity and purity profile. We utilize high-performance liquid chromatography (HPLC) alongside spectroscopic methods to confirm compliance with established monographs. The data below summarizes the critical physical and chemical properties that define our standard commercial grade.
| Parameter | Specification |
|---|---|
| CAS Number | 302-79-4 |
| Assay (HPLC) | 98.0% - 102.0% |
| Appearance | Fine Yellow Crystal Powder |
| Melting Point | 179-184°C |
| Molecular Formula | C20H28O2 |
| Loss on Drying | ≤0.50% |
| Heavy Metals | ≤20ppm |
| Total Impurities | ≤1.00% |
Industrial Applications
The utility of Tretinoin extends across multiple segments of the health and beauty industry. It is primarily incorporated into topical preparations designed to manage acne vulgaris and mitigate signs of photoaging. The compound functions by normalizing keratinization and promoting epidermal turnover, resulting in improved skin texture and tone. Manufacturers utilize this ingredient to formulate products that address both therapeutic and cosmetic needs.
- Pharmaceutical creams and gels for acne management and skin disorder treatment.
- Anti-aging serums and lotions targeting fine wrinkles and pigmentation.
- Adjuvant therapy formulations for psoriasis and disorders of keratinization.
- Research and development of novel retinoid delivery systems and derivatives.
Compliance with regulatory frameworks is essential when handling potent active ingredients. Our Tretinoin is produced in facilities that adhere to good manufacturing practices, ensuring traceability and safety. We support our customers with technical data packages that assist in regulatory filings for finished products. This commitment to compliance reduces time-to-market for our partners and ensures consumer safety.
Our clients benefit from a stable supply chain that supports continuous production schedules. The material is engineered to be compatible with various lipid bases and emulsion systems commonly employed in topical product manufacturing. We work closely with formulators to ensure that the physical properties of our powder meet their specific processing requirements.
Storage and Handling
Proper handling is crucial to maintain the integrity of retinoic acid derivatives due to their sensitivity to light and oxidation. The material must be stored in opaque, tightly sealed containers to prevent degradation. We recommend keeping the product in a cool, dry environment with temperatures maintained between 2-8°C for optimal stability. For long-term storage, freezing conditions at -20°C are advised to extend shelf life.
Packaging options include standard 25kg drums or customized solutions based on client logistics needs. All shipments are accompanied by comprehensive documentation, including Certificate of Analysis (COA) and Safety Data Sheets (SDS), to facilitate regulatory compliance and quality assurance checks upon receipt. Our logistics team ensures secure transport to prevent exposure to adverse conditions during transit.