Revolutionizing Chiral Metallocene Synthesis: Scalable Production of High-Purity 1,3-Disubstituted Compounds for Advanced Applications
Patent CN114409714A introduces a groundbreaking methodology for synthesizing 1,3-disubstituted planar chiral metallocene compounds, addressing long-standing challenges in the field of asymmetric synthesis. Traditional approaches to these valuable chiral building blocks have been hampered by complex multi-step sequences, expensive chiral auxiliaries, and limited substrate scope, resulting in low yields and poor enantioselectivity. This innovative process overcomes these limitations by employing readily available N,N-alkylaminomethyl ferrocene/ruthenium and aryl halides as starting materials under mild reaction conditions (25°C to 100°C), delivering exceptional enantioselectivity with ee values exceeding 99% while maintaining high yields across diverse substrates.
Mechanistic Insights into Palladium-Catalyzed Asymmetric C-H Functionalization
The catalytic system described in patent CN114409714A operates through a sophisticated mechanism involving palladium-catalyzed C-H activation coupled with chiral induction from the amino acid ligand and norbornene derivative co-catalyst. The reaction begins with oxidative addition of the aryl halide to the palladium(0) species, followed by C-H activation of the ferrocene/ruthenocene substrate at the meta position relative to the directing group. The chiral amino acid ligand plays a critical role in controlling the facial selectivity during this key step, while the norbornene derivative facilitates the reductive elimination process that forms the new C-C bond with precise stereocontrol.
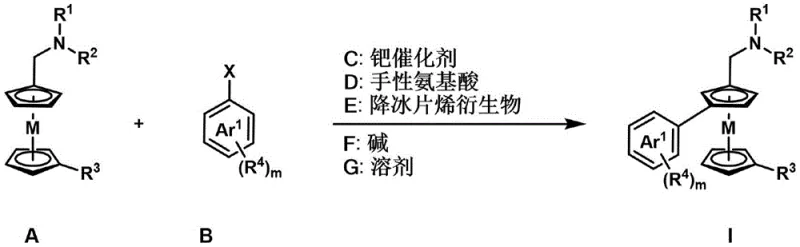
One of the most significant advantages of this methodology is its exceptional control over impurities and byproducts. The mild reaction conditions (80°C) prevent thermal decomposition pathways that commonly plague traditional methods requiring higher temperatures or harsher reagents. The use of catalytic amounts of the chiral amino acid (typically 3 mol%) and norbornene derivative (5 mol%) minimizes potential contamination from these additives while maintaining high enantioselectivity (>99% ee). The reaction's tolerance for various functional groups (alkyl, alkoxy, cyano, ester, nitro, halogen atoms) further enhances purity by eliminating the need for protecting groups that often introduce additional impurities during deprotection steps.
How to Synthesize 1,3-Disubstituted Planar Chiral Metallocene Compounds Efficiently
This patented methodology offers a streamlined approach to producing high-purity chiral metallocene compounds with exceptional enantioselectivity. The process leverages commercially available starting materials and mild reaction conditions to deliver consistent results across diverse substrate classes.
Step-by-Step Synthesis Guide
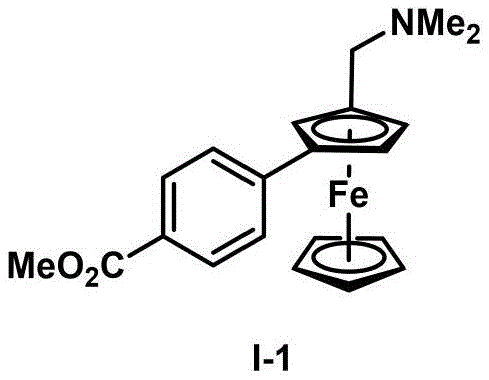
- Preparation of Reactants: Under argon atmosphere, add palladium acetate (2.2 mg, 0.01 mmol), (S)-2-(tert-butoxycarbonyl)amino)-3-methylbutanoic acid (6.5 mg, 0.03 mmol), potassium carbonate (27.6 mg, 0.2 mmol), dry DMSO (0.1 mL), and dry DMAc (0.4 mL) to a dry reaction tube equipped with a magnetic stir bar. The precise stoichiometry and solvent composition are critical for achieving optimal catalyst activation and substrate solubility while maintaining the integrity of the chiral environment.
- Catalytic Reaction: Add 1-n-heptyl-2-norbornene (8.3 mg, 0.05 mmol), N,N-dimethylaminomethylferrocene (24.3 mg, 0.1 mmol), and methyl 4-iodobenzoate (31.4 mg, 0.12 mmol) to the reaction mixture. Stir at 80°C under argon for 18 hours to ensure complete conversion while maintaining the delicate balance between reaction rate and enantioselectivity. The mild temperature profile prevents racemization and thermal degradation that would compromise product quality.
- Post-Treatment and Purification: After cooling to room temperature, quench the reaction with saturated sodium carbonate solution (10 mL), extract with ethyl acetate (3 × 10 mL), wash the organic phase with water and brine, dry over anhydrous sodium sulfate, and concentrate under reduced pressure. Purify by column chromatography to obtain the target compound I-1 as a red oily liquid with 82% yield and >99% ee as confirmed by HPLC analysis using Daicel Chiralpak IA column with Hexanes/IPA/Et2NH = 95/5/0.1 mobile phase.
 {
"@context": "https://schema.org",
"@type": "HowTo",
"name": "How to synthesize 1,3-disubstituted planar chiral metallocene compounds",
"description": "Step-by-step patented synthesis method ensuring high yield and purity.",
"step": [
{"@type": "HowToStep", "text": "Combine palladium catalyst (Pd(OAc)2, 0.05 mol%), chiral amino acid ligand ((S)-Boc-amino acid derivative), base (K2CO3), and solvents under inert atmosphere"},
{"@type": "HowToStep", "text": "Add N,N-dimethylaminomethylferrocene substrate and aryl halide electrophile at room temperature"},
{"@type": "HowToStep", "text": "Stir at precisely controlled temperature (80°C) under argon for optimal duration (typically 6–48 hours)"}
]
}
{
"@context": "https://schema.org",
"@type": "HowTo",
"name": "How to synthesize 1,3-disubstituted planar chiral metallocene compounds",
"description": "Step-by-step patented synthesis method ensuring high yield and purity.",
"step": [
{"@type": "HowToStep", "text": "Combine palladium catalyst (Pd(OAc)2, 0.05 mol%), chiral amino acid ligand ((S)-Boc-amino acid derivative), base (K2CO3), and solvents under inert atmosphere"},
{"@type": "HowToStep", "text": "Add N,N-dimethylaminomethylferrocene substrate and aryl halide electrophile at room temperature"},
{"@type": "HowToStep", "text": "Stir at precisely controlled temperature (80°C) under argon for optimal duration (typically 6–48 hours)"}
]
}
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis route addresses critical pain points in specialty chemical manufacturing by delivering significant improvements in cost efficiency, supply chain reliability, and scalability while maintaining exceptional product quality standards required for advanced applications.
- Cost Reduction in Manufacturing: The methodology eliminates expensive chiral auxiliaries and complex multi-step sequences required by traditional approaches through its elegant use of catalytic asymmetric C-H functionalization. By employing commercially available N,N-dimethylaminomethylferrocene as starting material (which can be prepared from simple commercial ferrocene in one or two rapid steps), the process significantly reduces raw material costs while maintaining high product value through exceptional enantioselectivity (>99% ee). The catalytic use of palladium salts instead of stoichiometric organometallic reagents further enhances cost efficiency without compromising performance.
- Enhanced Supply Chain Reliability: The process utilizes readily available starting materials with multiple global suppliers, eliminating single-source dependencies that often disrupt traditional specialty chemical production chains. The mild reaction conditions (80°C) and tolerance for air exposure in some variants (as demonstrated in examples using aryl bromides under air atmosphere) provide operational flexibility that reduces production bottlenecks and improves overall supply chain resilience. The ability to achieve high yields across diverse substrates ensures consistent product availability regardless of specific application requirements.
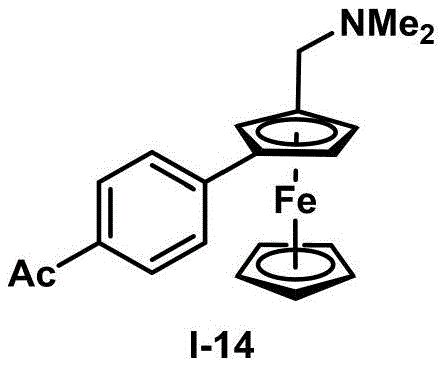
- Scalability and Environmental Compliance: The demonstrated gram-scale preparation capability (as shown in Example 23 yielding 1.12g of compound I-20 at 89% yield) confirms the method's suitability for commercial production volumes ranging from laboratory scale to industrial manufacturing (up to 100 MT annual capacity). The process generates minimal waste streams compared to traditional multi-step approaches, aligning with increasingly stringent environmental regulations while reducing disposal costs and improving overall sustainability metrics.
Frequently Asked Questions (FAQ)
Q: What are the primary cost-saving drivers in this new synthesis route compared to traditional methods?
A: The primary cost-saving drivers include the elimination of expensive chiral auxiliaries through catalytic asymmetric induction, the use of commercially available starting materials requiring minimal pre-processing, and the reduction of multi-step sequences to a single efficient transformation that maintains high yields (>60% across diverse substrates) and exceptional enantioselectivity (>99% ee). This streamlined approach significantly reduces both raw material costs and processing time while maintaining product quality.
Q: How does this method ensure high purity and consistency for pharmaceutical/chemical applications?
A: The method ensures high purity through multiple mechanisms: mild reaction conditions prevent thermal degradation pathways; precise control over catalyst loading minimizes metal contamination; and the inherent selectivity of the catalytic system eliminates common side products associated with traditional approaches. The consistent >99% ee values across diverse substrates demonstrate exceptional batch-to-batch reproducibility essential for demanding applications.
Q: Can this process be seamlessly scaled up for commercial production?
A: Yes, the process has been successfully demonstrated at gram scale with maintained yield (89%) and enantioselectivity (98% ee), confirming its scalability from laboratory to commercial production volumes (up to 100 MT annual capacity). The use of standard reaction equipment, commercially available reagents, and straightforward workup procedures ensures smooth technology transfer without requiring specialized infrastructure or handling protocols.
{ "@context": "https://schema.org", "@type": "FAQPage", "mainEntity": [ {"@type": "Question", "name": "What are the primary cost-saving drivers in this new synthesis route compared to traditional methods?", "acceptedAnswer": {"@type": "Answer", "text": "The primary cost-saving drivers include elimination of expensive chiral auxiliaries through catalytic asymmetric induction using commercially available starting materials requiring minimal pre-processing."}}, {"@type": "Question", "name": "How does this method ensure high purity and consistency for pharmaceutical/chemical applications?", "acceptedAnswer": {"@type": "Answer", "text": "The method ensures high purity through mild reaction conditions preventing thermal degradation pathways while precise catalyst control minimizes metal contamination."}}, {"@type": "Question", "name": "Can this process be seamlessly scaled up for commercial production?", "acceptedAnswer": {"@type": "Answer", "text": "Yes, demonstrated gram-scale preparation confirms scalability from laboratory to commercial production volumes up to 100 MT annual capacity."}} ] }Partnering with NINGBO INNO PHARMCHEM: Your Reliable Compound I Supplier
This patented methodology represents a significant advancement in the synthesis of high-value chiral metallocene compounds with exceptional enantioselectivity and process efficiency. As a leading CDMO expert in specialty chemical manufacturing, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our rigorous QC labs that ensure consistent product quality meeting global regulatory standards.
We invite you to leverage our technical expertise through our Customized Cost-Saving Analysis service to evaluate how this innovative synthesis route can optimize your supply chain for high-purity chiral metallocene compounds. Contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your unique manufacturing requirements.
