Scalable Electrochemical Recycling of Chiral Naphthyridine Intermediates for Commercial API Production
Introduction to Advanced Electrochemical Recycling in Pharma Synthesis
The pharmaceutical industry continuously seeks robust, scalable, and economically viable synthetic routes for complex active pharmaceutical ingredients (APIs). Patent CN108137587B introduces a groundbreaking methodology for the preparation and recovery of (4S)-4-(4-cyano-2-methoxyphenyl)-5-ethoxy-2,8-dimethyl-1,4-dihydro-1,6-naphthyridine-3-carboxamide, a critical intermediate for mineralocorticoid receptor antagonists. This technology addresses the longstanding challenge of low atom economy in chiral synthesis by implementing a sophisticated electrochemical recycling loop. Instead of discarding the unwanted enantiomer, a common practice that halves potential yield, this process converts it back into a reusable racemic form. For R&D directors and process chemists, this represents a paradigm shift from linear synthesis to circular chemical manufacturing, ensuring higher purity and significantly reduced waste generation while maintaining strict regulatory compliance for GMP production environments.
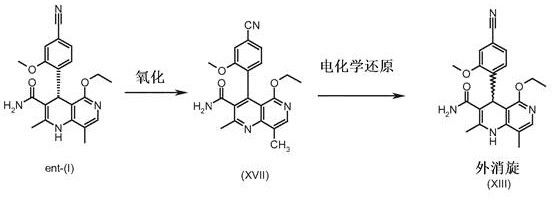
The core innovation lies in the seamless integration of indirect electrochemical oxidation and reduction steps. By leveraging sub-stoichiometric mediators, the process avoids the pitfalls of traditional stoichiometric oxidation, such as the use of toxic heavy metals or explosive peroxides. This approach not only enhances the safety profile of the manufacturing process but also simplifies downstream purification, as fewer by-products are generated. The ability to recover and reuse the ent-(I) enantiomer transforms what was once a waste stream into a valuable feedstock, fundamentally altering the cost structure of producing high-purity chiral intermediates. This patent provides a detailed roadmap for achieving these efficiencies, making it a vital reference for any organization aiming to optimize their supply chain for complex heterocyclic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for naphthyridine derivatives, as described in prior art like WO2008/104306, suffer from severe inefficiencies that hinder commercial viability. A primary bottleneck is the reliance on research-grade synthesis conditions that are impractical for multi-ton scale production. These methods often necessitate extremely high dilutions and vast excesses of reagents to drive reactions to completion, leading to disproportionately low overall yields, historically reported around 3.76% from simple starting materials. Furthermore, the purification burden is immense, requiring multiple chromatographic steps including preparative chiral HPLC, which is notoriously solvent-intensive and difficult to scale. Safety is another critical concern; conventional pathways frequently employ hazardous reagents such as osmium tetroxide for oxidative cleavage or trifluoromethanesulfonic anhydride for activation, posing significant occupational health risks and waste disposal challenges. The formation of plastic-like polymeric residues during scale-up of Heck reactions further complicates reactor maintenance and operational continuity.
The Novel Approach
The novel process outlined in CN108137587B overcomes these barriers through a strategic redesign of the synthetic pathway, focusing on telescoped reactions and electrochemical regeneration. By avoiding the isolation of unstable intermediates like the methyl ester (XV) and aldehyde (XVI), the new method reduces the number of discrete unit operations, thereby minimizing material handling losses. The most transformative aspect is the recycling strategy: the unwanted ent-(I) enantiomer, typically a waste product of chiral resolution, is oxidized to the achiral pyridine derivative (XVII) and subsequently reduced back to the racemic amide (XIII). This allows the material to re-enter the chiral separation stage, theoretically enabling near-quantitative conversion of all starting material into the desired (4S)-enantiomer over multiple cycles. This approach elevates the overall yield to approximately 27.7%, a substantial improvement that directly translates to reduced raw material costs and a smaller environmental footprint for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into Indirect Electrochemical Oxidation and Reduction
The heart of this technological advancement is the precise control of redox potentials to achieve selective transformations without degrading sensitive functional groups. The oxidation of the 1,4-dihydropyridine derivative ent-(I) to the pyridine analog (XVII) is achieved via indirect electrochemical oxidation using a catalytic amount of 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) as a mediator. Unlike direct electrolysis which requires high potentials (> +1.0 V vs. Ag/Ag+) that can lead to non-selective side reactions and electrode fouling, this mediated process operates within a narrow, optimized potential window of +0.3 V to +0.5 V. In this regime, the DDQ mediator is efficiently regenerated at the anode, while the substrate is chemically oxidized in the bulk solution. This decoupling of electron transfer from the chemical reaction ensures high current efficiency (>95%) and exceptional selectivity, preventing the oxidation of other susceptible moieties like the amide or nitrile groups.
Following oxidation, the resulting pyridine derivative (XVII) undergoes electrochemical reduction to regenerate the racemic 1,4-dihydropyridine (XIII). This step is equally critical and is performed under controlled conditions, often in a flow cell configuration to enhance mass transfer. The reduction selectively targets the pyridine ring without affecting the nitrile or ester functionalities, a challenge that chemical reducing agents like sodium borohydride often fail to meet due to lack of chemoselectivity. The use of a divided cell with a cation exchange membrane prevents the re-oxidation of the product at the anode, ensuring high faradaic efficiency. The mechanistic elegance of this two-step redox cycle lies in its reversibility and controllability; by tuning parameters such as current density (10-30 mA/cm²) and solvent composition (methanol/DMF mixtures), the process can be finely adjusted to maximize the recovery of the racemate. This level of control is essential for maintaining the stringent purity specifications required for API intermediates, ensuring that the recycled material is indistinguishable in quality from virgin synthesis batches.
How to Synthesize Finerenone Intermediate Efficiently
Implementing this electrochemical recycling protocol requires careful attention to reaction engineering and parameter optimization to ensure reproducibility and safety. The process begins with the chiral separation of the racemic amide (XIII) using Simulated Moving Bed (SMB) chromatography, which isolates the target (I) and the byproduct ent-(I). The ent-(I) fraction is then subjected to the electrochemical oxidation step in an undivided or divided cell using a platinum or carbon electrode system. Following isolation of the pyridine intermediate (XVII), it is dissolved in a methanol/DMF electrolyte solution containing a supporting salt like tetraethylammonium tetrafluoroborate. The detailed standardized synthesis steps see the guide below.
- Oxidize the unwanted enantiomer ent-(I) to the pyridine derivative (XVII) using indirect electrochemical oxidation with a DDQ mediator.
- Subject the resulting pyridine derivative (XVII) to electrochemical reduction in a flow cell or batch reactor to regenerate the racemic amide (XIII).
- Separate the regenerated racemate (XIII) using Simulated Moving Bed (SMB) chromatography to isolate the target (4S)-enantiomer (I) and recycle the remaining ent-(I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical recycling technology offers compelling strategic advantages beyond mere technical feasibility. The primary benefit is the drastic reduction in raw material costs associated with chiral synthesis. By recovering and reprocessing the unwanted enantiomer, the effective cost of the chiral building block is significantly lowered, as the yield loss inherent in classical resolution is mitigated. This leads to substantial cost savings in API manufacturing, making the final drug product more competitive in the marketplace. Furthermore, the elimination of hazardous stoichiometric oxidants like osmium tetroxide removes the need for expensive waste treatment protocols and specialized handling equipment, further driving down operational expenditures. The process's compatibility with continuous flow electrochemistry also means that production capacity can be scaled incrementally by adding more cell modules rather than building massive batch reactors, offering greater flexibility in meeting fluctuating market demand.
Supply chain reliability is enhanced through the simplification of the synthetic route. The new method reduces the number of isolation and purification steps, particularly avoiding intermediate chromatography which is a common bottleneck in production schedules. This streamlining shortens the overall lead time for high-purity pharmaceutical intermediates, ensuring a more consistent and predictable supply of critical materials. Additionally, the use of commercially available starting materials and standard electrochemical equipment reduces dependency on exotic reagents that might be subject to supply disruptions. From an environmental compliance perspective, the reduction in solvent consumption and toxic waste generation aligns with increasingly stringent global regulations on pharmaceutical manufacturing, reducing the risk of regulatory delays and enhancing the sustainability profile of the supply chain. This makes the process not only economically attractive but also a future-proof investment for long-term production stability.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this electrochemical recycling process. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these nuances is crucial for evaluating the feasibility of integrating this technology into existing manufacturing infrastructure. The insights cover aspects ranging from equipment selection to impurity control, providing a comprehensive overview for decision-makers.
Q: How does the electrochemical recycling process improve overall yield compared to conventional methods?
A: Conventional research syntheses often discard the unwanted enantiomer, resulting in a maximum theoretical yield of 50% for the chiral step and overall yields as low as 3.76%. The patented electrochemical recycling process converts the unwanted ent-(I) enantiomer back into the racemic mixture, allowing for multiple passes through the chiral separation unit. This iterative recovery significantly boosts the overall yield to approximately 27.7% and maximizes raw material utilization.
Q: What are the safety advantages of using indirect electrochemical oxidation over chemical oxidants?
A: Traditional chemical oxidation often requires stoichiometric amounts of hazardous reagents like osmium tetroxide or large excesses of DDQ, generating significant toxic waste. The patented method utilizes sub-stoichiometric amounts of a DDQ mediator regenerated electrochemically at mild potentials (+0.3 to +0.5 V). This eliminates the need for dangerous heavy metal oxidants and reduces the thermal risks associated with exothermic chemical oxidation reactions.
Q: Is this electrochemical process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for scalability. The patent demonstrates successful operation in flow cells (e.g., MicroFlow Cell or Multipurpose Cell) which allow for continuous processing and better heat/mass transfer compared to batch reactors. The method has been validated for the preparation of hundreds of kilograms of material for clinical trials, proving its viability for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Finerenone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modern pharmaceutical manufacturing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN108137587B can be seamlessly transferred from the lab to the plant. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and SMB chromatography units, allowing us to implement complex recycling loops with precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Finerenone intermediate meets the highest international standards, providing our clients with peace of mind regarding quality and consistency.
We invite you to collaborate with us to leverage these advanced synthetic technologies for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how electrochemical recycling can optimize your budget. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that is not only robust and compliant but also driven by cutting-edge chemistry designed to maximize efficiency and minimize environmental impact.
