Revolutionizing Triazole Synthesis: A Cost-Effective DMF-Based Route for Pharmaceutical Intermediates
Revolutionizing Triazole Synthesis: A Cost-Effective DMF-Based Route for Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks more efficient pathways to construct privileged heterocyclic scaffolds, particularly those containing fluorine motifs which are ubiquitous in modern drug design. Patent CN114920707B introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, leveraging the common organic solvent N,N-dimethylformamide (DMF) not merely as a medium, but as a critical carbon source. This innovation addresses long-standing challenges in heterocyclic chemistry by merging solvent and reactant roles, thereby streamlining the synthetic workflow. The significance of this chemical transformation cannot be overstated, as the 1,2,4-triazole core is a fundamental building block found in numerous bioactive molecules, ranging from anticonvulsants to Factor IXa inhibitors. By utilizing a readily available commodity chemical like DMF to construct these complex rings, the technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
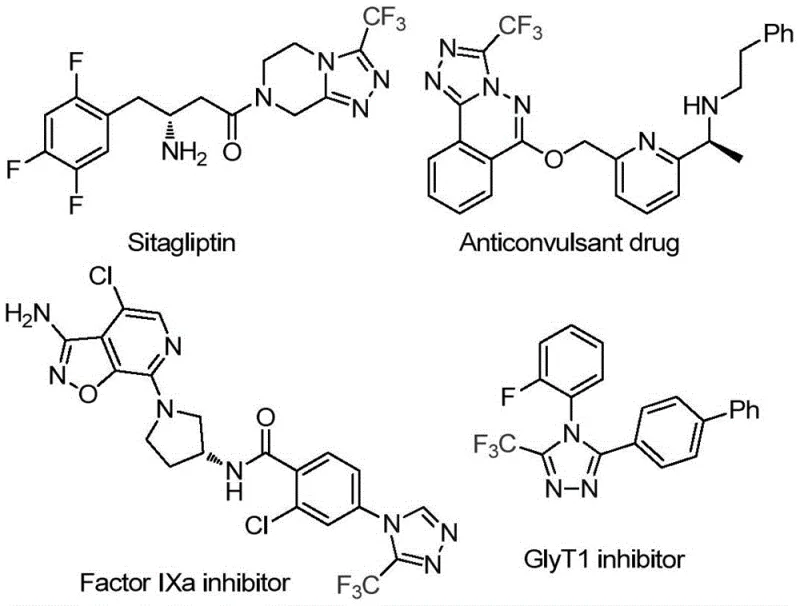
The strategic importance of this synthesis route is highlighted by the prevalence of the trifluoromethyl-triazole motif in high-value therapeutics. As illustrated in the structural diversity of known drugs, incorporating a trifluoromethyl group often enhances metabolic stability and lipophilicity, crucial parameters for oral bioavailability. Traditional approaches to accessing these scaffolds often involve multi-step sequences with expensive reagents or harsh conditions that limit scalability. In contrast, the methodology disclosed in CN114920707B utilizes a molecular iodine-promoted tandem cyclization that proceeds under relatively mild thermal conditions. This shift represents a paradigm change in how we approach cost reduction in API manufacturing, moving away from specialized reagents toward bulk solvents that double as synthetic building blocks. For procurement managers and supply chain heads, this translates to reduced dependency on niche chemical suppliers and a more robust, resilient production pipeline capable of withstanding market fluctuations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,2,4-triazole rings bearing trifluoromethyl groups has been fraught with synthetic inefficiencies that hinder large-scale commercialization. Conventional methodologies frequently rely on the condensation of hydrazides with specific nitriles or amidines, which often necessitates the pre-synthesis of these precursors through separate, resource-intensive steps. Furthermore, many established protocols demand stringent reaction environments, such as strictly anhydrous conditions or inert gas atmospheres, to prevent side reactions or hydrolysis of sensitive intermediates. These requirements impose significant operational burdens on manufacturing facilities, necessitating specialized equipment and increasing energy consumption. Additionally, the use of stoichiometric amounts of expensive coupling agents or toxic heavy metal catalysts in traditional routes generates substantial chemical waste, complicating downstream purification and environmental compliance. The cumulative effect of these factors is a high cost of goods sold (COGS) and extended lead times, creating bottlenecks for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
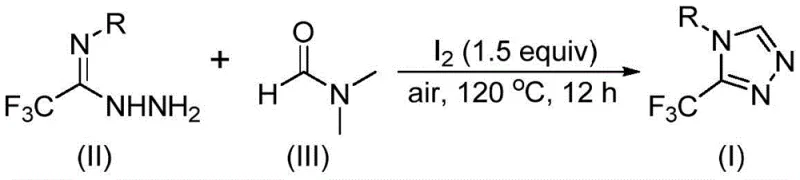
The inventive method described in the patent circumvents these obstacles by employing DMF as a versatile C1 synthon in an iodine-mediated cyclization. This approach fundamentally simplifies the reaction matrix by eliminating the need for external formylating agents; instead, the solvent itself provides the necessary carbon atom to close the triazole ring. The reaction operates effectively in an air atmosphere at temperatures between 110°C and 130°C, removing the logistical complexity associated with glovebox operations or extensive nitrogen purging. This operational simplicity is a key driver for reducing lead time for high-purity pharmaceutical intermediates, as it allows for faster batch turnover and reduced setup times. Moreover, the substrate scope is remarkably broad, tolerating a wide array of functional groups on the aromatic ring, including electron-donating and electron-withdrawing substituents. This versatility ensures that the process can be adapted for the synthesis of diverse analog libraries without requiring extensive re-optimization, making it an ideal platform technology for process research and development teams.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for R&D directors evaluating its feasibility for process integration. The reaction initiates with the activation of DMF by molecular iodine, which facilitates the cleavage of the C-N bond or activates the carbonyl species depending on the specific pathway taken. In one plausible mechanistic manifold, the formyl group of DMF acts as the carbon source, undergoing condensation with the trifluoroethyliminohydrazide to generate a hydrazone intermediate. Subsequent intramolecular cyclization followed by the elimination of dimethylamine yields the desired 3-trifluoromethyl-1,2,4-triazole product. Alternatively, the N-methyl group of DMF may participate, reacting with iodine to form an amine salt which then undergoes nucleophilic addition. Regardless of the specific pathway, the presence of iodine is critical for promoting the oxidative aromatization step that drives the formation of the stable triazole ring system.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. The absence of transition metals eliminates the risk of heavy metal contamination, a critical quality attribute for pharmaceutical ingredients that often requires costly scavenging steps. The byproducts generated, primarily dimethylamine salts or iodine derivatives, are generally easier to remove via standard aqueous workups or crystallization techniques compared to organometallic residues. Furthermore, the reaction demonstrates high chemoselectivity, minimizing the formation of regioisomers or polymeric byproducts that can plague heterocyclic syntheses. This clean reaction profile contributes to higher crude purity, reducing the burden on final purification stages such as column chromatography or recrystallization. For quality assurance teams, this implies a more predictable impurity profile and a smoother path to regulatory approval, ensuring that the final high-purity pharmaceutical intermediates meet stringent global pharmacopeial standards.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for scalability and ease of handling, making it accessible for both laboratory discovery and pilot plant operations. The protocol involves a straightforward mixing of reagents followed by a thermal cycle, avoiding the need for slow addition pumps or cryogenic cooling. Detailed standardized operating procedures regarding stoichiometry, agitation rates, and quenching protocols are essential to maximize yield and consistency across different batch sizes. The following guide outlines the critical operational parameters derived from the patent data to ensure successful implementation.
- Combine molecular iodine and trifluoroethyliminohydrazide in DMF solvent within a reaction vessel.
- Heat the mixture to 110-130°C under air atmosphere for 10-15 hours to facilitate the tandem cyclization.
- Perform post-treatment including filtration, washing, and column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this DMF-based synthesis route offers tangible economic and logistical benefits that directly impact the bottom line. The primary driver of value is the drastic simplification of the bill of materials (BOM). By utilizing DMF, a solvent that is produced on a massive industrial scale and available at commodity prices, the process replaces expensive, specialized carbon sources. This substitution leads to substantial cost savings in raw material procurement, insulating the manufacturing process from the volatility often seen in the pricing of fine chemical reagents. Additionally, the elimination of anhydrous and anaerobic requirements reduces capital expenditure on specialized reactor infrastructure and lowers utility costs associated with maintaining inert atmospheres. These factors combine to create a highly competitive cost structure for the production of trifluoromethyl-triazole derivatives.
- Cost Reduction in Manufacturing: The economic efficiency of this process is anchored in the dual functionality of DMF. Since the solvent acts as a reactant, there is no need to purchase separate formylating agents or C1 synthons, which are often priced at a premium. This consolidation of materials significantly lowers the direct material cost per kilogram of product. Furthermore, the use of molecular iodine as a promoter is advantageous because it is relatively inexpensive compared to palladium or copper catalysts often used in similar cyclizations. The avoidance of noble metals also removes the necessity for expensive metal scavenging resins during purification, further driving down processing costs. Overall, the streamlined reagent profile ensures a leaner, more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available starting materials. DMF and molecular iodine are commodity chemicals with robust global supply networks, minimizing the risk of shortages that can disrupt production schedules. Unlike specialized reagents that may have single-source suppliers or long lead times, the inputs for this reaction can be sourced from multiple vendors, providing procurement teams with greater negotiating power and flexibility. The simplified reaction conditions also mean that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without significant requalification efforts, ensuring continuity of supply even in the face of regional disruptions.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this method aligns well with green chemistry principles. The reaction does not generate heavy metal waste, simplifying effluent treatment and reducing the environmental footprint of the manufacturing site. The ability to run the reaction under air atmosphere reduces the consumption of nitrogen or argon gases, contributing to lower operational emissions. Moreover, the high reaction efficiency and selectivity minimize the generation of organic waste streams associated with extensive purification steps. These attributes facilitate easier regulatory compliance and support sustainability goals, making the process attractive for companies aiming to reduce their environmental impact while scaling up production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating the technology for potential licensing or production partnerships.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (C1 synthon), eliminating the need for separate, expensive formylating agents and significantly reducing raw material costs.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, the patented method operates efficiently under standard air atmosphere without the need for rigorous anhydrous or oxygen-free environments, simplifying operational requirements.
Q: What is the substrate scope for the R group in this reaction?
A: The method demonstrates broad substrate tolerance, accommodating various substituted aryl groups including those with alkyl, alkoxy, halogen, and trifluoromethyl substituents at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of efficient synthetic methodologies like the one described in CN114920707B for the pharmaceutical industry. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory processes into robust commercial manufacturing lines. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale discovery to multi-ton supply is seamless and risk-mitigated. We are committed to delivering stringent purity specifications through our state-of-the-art rigorous QC labs, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole intermediate meets the exacting standards required for downstream API synthesis.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective technology for your drug development programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain and accelerate your path to market with our reliable supply of high-quality pharmaceutical intermediates.
