Advanced Molybdenum-Copper Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
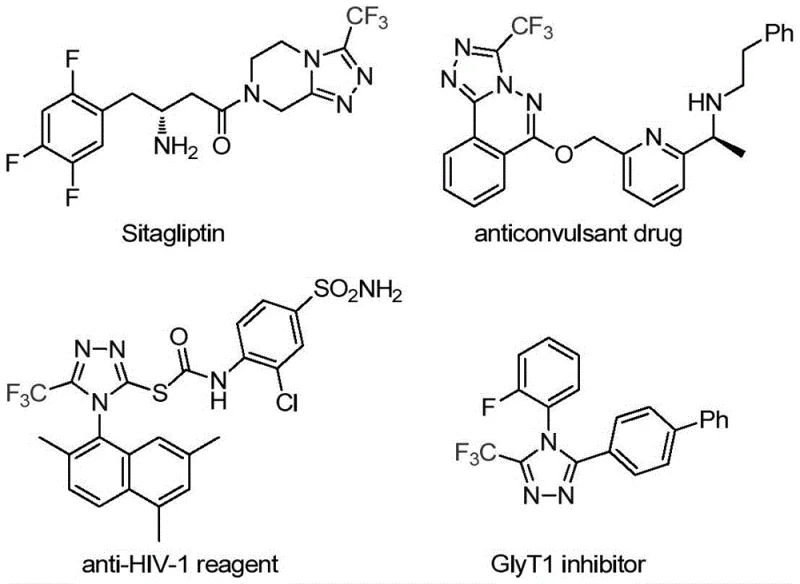
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are critical for enhancing metabolic stability and bioavailability in drug candidates. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, utilizing a synergistic molybdenum and copper co-catalytic system. This innovation addresses the longstanding challenges associated with synthesizing these privileged scaffolds, which are ubiquitous in high-value therapeutics such as Sitagliptin and various antifungal agents. By leveraging functionalized isonitriles and trifluoroethylimidoyl chloride as key building blocks, this technology offers a streamlined pathway that bypasses the need for hazardous reagents often found in legacy protocols. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for complex heterocyclic intermediates while maintaining stringent purity standards required for GMP manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on multistep sequences that often involve the cyclization of trifluoroacetyl hydrazine with amidine derivatives or the hydrazinolysis of oxazolinone precursors. These traditional routes frequently suffer from苛刻 reaction conditions, requiring extreme temperatures or highly reactive species like diazonium salts and trifluorodiazoethane, which pose significant safety hazards and handling difficulties on a large scale. Furthermore, many existing copper-catalyzed multi-component reactions necessitate precise control over stoichiometry and often result in poor atom economy due to the generation of substantial inorganic waste. The reliance on unstable intermediates not only increases the cost of goods sold but also complicates the purification process, leading to lower overall yields and inconsistent batch-to-batch quality that can disrupt downstream API synthesis timelines.
The Novel Approach
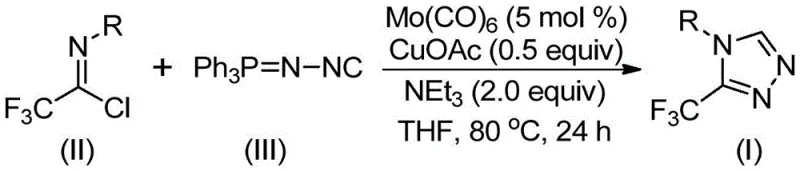
In stark contrast, the methodology disclosed in CN113307778A employs a sophisticated yet operationally simple one-pot cycloaddition strategy driven by molybdenum hexacarbonyl and cuprous acetate. This novel approach utilizes readily accessible trifluoroethylimidoyl chloride and functionalized isonitrile (specifically Ph3P=N-NC) to directly forge the triazole ring under mild thermal conditions ranging from 70 to 90°C. The use of triethylamine as a base and THF as the solvent creates a homogeneous reaction environment that facilitates efficient mass transfer and catalyst turnover. By eliminating the need for dangerous diazo compounds and reducing the number of isolation steps, this process drastically simplifies the workflow, making it highly attractive for cost reduction in pharmaceutical intermediate manufacturing. The ability to tolerate a wide range of functional groups on the aromatic ring further enhances the versatility of this platform for generating diverse chemical libraries.
Mechanistic Insights into Mo-Cu Co-Catalyzed Cycloaddition
The mechanistic elegance of this transformation lies in the cooperative interaction between the molybdenum and copper centers, which orchestrate the activation of distinct reaction partners to drive the cyclization forward. It is proposed that molybdenum hexacarbonyl initially coordinates with the functionalized isonitrile to form a reactive metal-isonitrile complex, thereby increasing the nucleophilicity of the carbon center. Subsequently, the cuprous acetate promoter facilitates a [3+2] cycloaddition between this activated species and the trifluoroethylimidoyl chloride, leading to the formation of a five-membered metallacycle intermediate. This step is critical as it establishes the core triazole architecture while incorporating the trifluoromethyl group with high regioselectivity. The final stage involves the elimination of triphenylphosphine oxide, likely assisted by trace water in the system or the basic environment, to release the stable 3-trifluoromethyl-1,2,4-triazole product and regenerate the catalytic species for subsequent cycles.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions commonly associated with radical pathways or uncontrolled nucleophilic attacks. The specific coordination geometry imposed by the metal ligands ensures that the cyclization proceeds through a defined trajectory, reducing the formation of regioisomers or polymeric byproducts that are difficult to separate. Moreover, the use of molecular sieves in the reaction mixture helps to sequester moisture that could otherwise hydrolyze the sensitive imidoyl chloride starting material, thereby preserving the integrity of the reactants and maximizing the conversion rate. This level of mechanistic control is paramount for R&D teams aiming to achieve high-purity outputs without resorting to extensive and costly chromatographic purification steps, ultimately supporting the production of high-purity pharmaceutical intermediates suitable for clinical applications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this synthesis effectively, operators must adhere to strict anhydrous conditions and precise stoichiometric ratios to ensure optimal catalyst performance and yield. The process begins with the careful charging of molybdenum hexacarbonyl, cuprous acetate, and triethylamine into a dry reaction vessel containing THF, followed by the sequential addition of the imidoyl chloride and isonitrile substrates. Maintaining the reaction temperature within the specified window of 70 to 90°C for a duration of 18 to 30 hours is essential to drive the reaction to completion while preventing thermal degradation of the sensitive triazole product. Detailed standardized synthetic steps see the guide below.
- Charge a reaction vessel with molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in anhydrous THF.
- Add trifluoroethylimidoyl chloride and the functionalized isonitrile (Ph3P=N-NC) to the mixture under inert atmosphere.
- Heat the reaction mixture to 80°C for 24 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits regarding cost efficiency and supply reliability. The shift towards using commercially abundant starting materials like trifluoroethylimidoyl chloride and functionalized isonitrile eliminates the dependency on custom-synthesized, high-cost precursors that often bottleneck production schedules. Additionally, the robustness of the molybdenum-copper catalyst system allows for extended campaign runs with consistent performance, reducing the frequency of catalyst replenishment and minimizing downtime associated with reactor cleaning and changeovers. This operational stability is crucial for maintaining continuous supply lines to downstream API manufacturers who demand just-in-time delivery of critical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the use of relatively inexpensive metal catalysts such as cuprous acetate and molybdenum hexacarbonyl compared to precious metals like palladium or rhodium. Furthermore, the simplified workup procedure, which involves basic filtration and standard column chromatography, reduces the consumption of solvents and silica gel, leading to substantial cost savings in waste disposal and raw material usage. By avoiding the need for cryogenic conditions or specialized high-pressure equipment, capital expenditure for plant retrofitting is also minimized, allowing for immediate integration into existing manufacturing infrastructure.
- Enhanced Supply Chain Reliability: The reliance on off-the-shelf reagents ensures that the supply chain is resilient against market fluctuations or shortages of exotic chemicals. Since the key substrates are derived from common aromatic amines and trifluoroacetic acid derivatives, sourcing can be diversified across multiple global vendors to mitigate risk. The high tolerance of the reaction to various substituents means that a single production line can be easily adapted to manufacture a wide portfolio of triazole derivatives, providing flexibility to respond rapidly to changing customer demands without significant retooling costs.
- Scalability and Environmental Compliance: The patent explicitly notes the successful expansion of this chemistry to gram-level scales, demonstrating its readiness for kilogram and tonne-scale production. The use of THF as a solvent, which is widely accepted in pharmaceutical processing, combined with the absence of toxic heavy metal waste streams, aligns well with modern green chemistry principles and environmental regulations. This compliance reduces the regulatory burden on the supply chain team, facilitating smoother audits and faster approval times for new process validations in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their current process development pipelines. The answers provided reflect the specific conditions and outcomes reported in the invention disclosure to ensure accuracy and reliability.
Q: What are the key advantages of the Mo-Cu co-catalytic system over traditional methods?
A: Unlike traditional methods requiring harsh cyclization conditions or unstable diazonium salts, this patent utilizes a mild Mo-Cu dual catalytic system that operates at moderate temperatures (70-90°C) with commercially available starting materials, significantly simplifying the operational complexity.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly demonstrates that the reaction can be expanded to gram-level scales with high efficiency, indicating strong potential for commercial scale-up due to the use of robust catalysts and simple post-treatment procedures.
Q: What is the substrate scope for the R-group in this triazole synthesis?
A: The method exhibits broad substrate tolerance, successfully accommodating various aryl groups including those with electron-donating (methyl, methoxy) and electron-withdrawing (fluoro, chloro, nitro) substituents, as well as phenethyl groups, allowing for diverse derivative synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics, and we are fully equipped to leverage this innovative Mo-Cu catalytic technology for your projects. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in early-stage discovery or late-stage clinical trials. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the highest industry standards for impurity profiles and physicochemical properties.
We invite you to engage with our technical procurement team to discuss how this efficient synthesis route can be tailored to your specific molecule requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing your intermediate supply can impact your overall project budget and timeline. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being a transparent and capable partner in your supply chain.
