Scalable Production of (S)-3-Hydroxytetrahydrofuran via Dual-Catalyst Asymmetric Hydrogenation for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for critical chiral intermediates, and patent CN110396072B presents a significant breakthrough in the synthesis of (S)-3-hydroxytetrahydrofuran. This optically pure compound serves as a pivotal building block for a diverse array of high-value therapeutic agents, including the diabetes medication Empagliflozin, the breast cancer treatment Afatinib, and anti-AIDS drugs such as Amprenavir and Fosamprenavir. 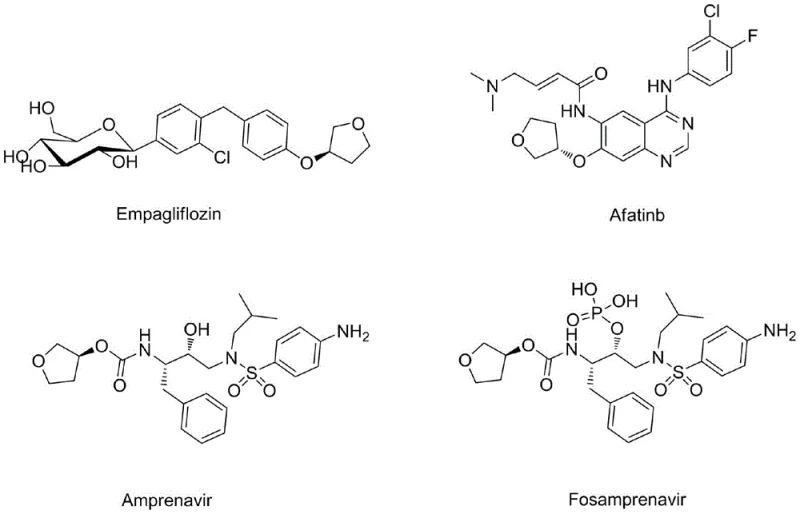 The structural complexity of these final active pharmaceutical ingredients demands a precursor with exceptional stereochemical integrity, making the efficient production of (S)-3-hydroxytetrahydrofuran a strategic priority for supply chain stability. The disclosed invention addresses historical bottlenecks by introducing a streamlined two-step protocol that leverages a sophisticated dual-catalyst system to achieve high yields and optical purity from readily available starting materials.
The structural complexity of these final active pharmaceutical ingredients demands a precursor with exceptional stereochemical integrity, making the efficient production of (S)-3-hydroxytetrahydrofuran a strategic priority for supply chain stability. The disclosed invention addresses historical bottlenecks by introducing a streamlined two-step protocol that leverages a sophisticated dual-catalyst system to achieve high yields and optical purity from readily available starting materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 3-hydroxytetrahydrofuran has been plagued by reliance on the chiral pool strategy, which often dictates prohibitive costs and supply constraints. Early methodologies, such as those described by Tadon, utilized chiral malic acid as a starting material, necessitating the use of hazardous and expensive reducing agents like lithium aluminum hydride in large stoichiometric quantities. Subsequent improvements attempted to mitigate these issues but still suffered from multi-step sequences involving esterification and dehydration, leading to cumulative yield losses and significant wastewater generation. Other approaches employing optically pure (S)-4-chloro-3-hydroxybutyric acid ethyl ester faced similar economic hurdles due to the high cost of the raw material and the environmental burden of sodium borohydride reduction. Furthermore, biological enzyme catalysis, while green in principle, has historically struggled with long reaction periods, low volumetric productivity, and the high expense of specialized enzymes, rendering them unsuitable for the tonnage-scale production required by the global API market.
The Novel Approach
In stark contrast to these legacy processes, the method disclosed in patent CN110396072B revolutionizes the manufacturing landscape by utilizing ethyl 4-chloroacetoacetate, a cheap and commercially abundant commodity chemical, as the primary feedstock. This innovative route bypasses the need for chiral starting materials entirely by installing chirality through a highly selective asymmetric hydrogenation reaction. The process employs a synergistic dual-catalyst system comprising an iridium complex generated in situ from [Ir(COD)Cl]2 and a specific phosphine-pyridine ligand, working in concert with a Ru-MACHO complex. 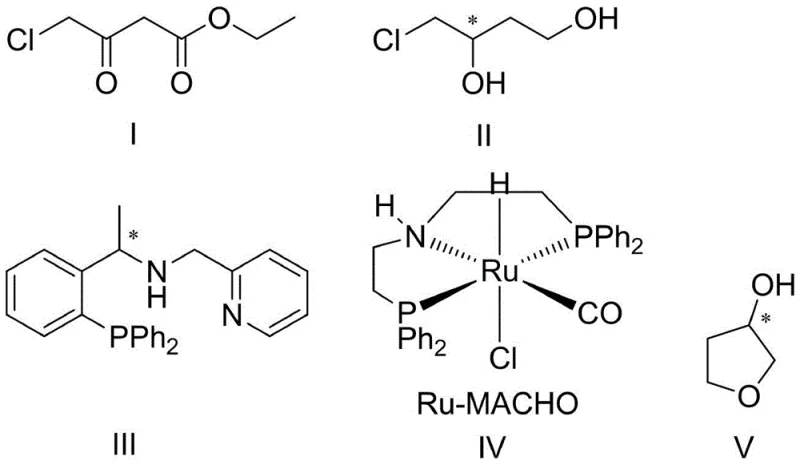 This catalytic synergy allows for the direct transformation of the achiral beta-keto ester into the chiral intermediate (S)-4-chloro-3-hydroxy-1-butanol with exceptional efficiency. The subsequent cyclization step is equally elegant, utilizing simple acid catalysis to close the ring, thereby eliminating the need for complex protecting group strategies or harsh dehydrating conditions that typically degrade product quality.
This catalytic synergy allows for the direct transformation of the achiral beta-keto ester into the chiral intermediate (S)-4-chloro-3-hydroxy-1-butanol with exceptional efficiency. The subsequent cyclization step is equally elegant, utilizing simple acid catalysis to close the ring, thereby eliminating the need for complex protecting group strategies or harsh dehydrating conditions that typically degrade product quality.
Mechanistic Insights into Dual-Catalyst Asymmetric Hydrogenation
The core of this technological advancement lies in the intricate interplay between the iridium-based chiral catalyst and the ruthenium-based hydrogenation catalyst during the first synthetic step. The phosphine-pyridine ligand coordinates with the iridium precursor [Ir(COD)Cl]2 to form a chiral environment that dictates the facial selectivity of the hydrogen addition to the ketone moiety of the substrate. Simultaneously, the Ru-MACHO complex acts as a powerful hydrogenation engine, facilitating the activation of molecular hydrogen under moderate pressures ranging from 1 MPa to 8 MPa. This cooperative catalysis ensures that the reduction of the carbonyl group proceeds with rigorous stereocontrol, effectively suppressing the formation of the unwanted (R)-enantiomer and other reduced byproducts. The reaction conditions are optimized to balance kinetics and selectivity, typically operating between 80°C and 120°C, which provides sufficient thermal energy to drive the reaction to completion within 5 to 6 hours without compromising the delicate chiral integrity of the intermediate.
Following the asymmetric hydrogenation, the resulting (S)-4-chloro-3-hydroxy-1-butanol undergoes an intramolecular nucleophilic substitution to form the tetrahydrofuran ring. This cyclization is catalyzed by strong acids such as hydrochloric acid or sulfuric acid, which protonate the hydroxyl group or activate the leaving group to facilitate ring closure. The mechanism is straightforward yet highly effective, proceeding cleanly in solvents like water, methanol, or ethanol at temperatures between 50°C and 80°C. Crucially, the mild acidic conditions prevent the racemization of the chiral center established in the previous step, preserving the 99% optical purity achieved during hydrogenation. The purification process involves simple distillation techniques, including short-path and reduced-pressure distillation, which efficiently separate the volatile product from the reaction matrix, yielding a final substance with chemical purity exceeding 98% and minimal residual solvent content.
How to Synthesize (S)-3-Hydroxytetrahydrofuran Efficiently
The synthesis protocol outlined in the patent offers a clear pathway for laboratory and pilot-scale execution, emphasizing precise control over catalyst loading and reaction parameters to maximize yield. The process begins with the in situ preparation of the iridium catalyst under inert atmosphere, followed by the introduction of the substrate and the ruthenium co-catalyst into a high-pressure reactor. Detailed operational guidelines regarding solvent ratios, base selection (such as potassium tert-butoxide or sodium methoxide), and hydrogen pressurization cycles are critical for reproducing the high enantioselectivity reported in the examples. For a comprehensive understanding of the specific molar ratios, solvent volumes, and workup procedures required to achieve the reported 90%+ yields, please refer to the standardized synthesis guide below.
- Prepare the first catalyst by reacting [Ir(COD)Cl]2 with a phosphine-pyridine ligand in a solvent like ethanol under nitrogen protection.
- Perform asymmetric hydrogenation of ethyl 4-chloroacetoacetate using the iridium catalyst and Ru-MACHO complex under 2-4 MPa hydrogen pressure at 80-120°C to yield (S)-4-chloro-3-hydroxy-1-butanol.
- Dissolve the intermediate in a second solvent, add an acid catalyst such as hydrochloric acid, and react at 50-80°C to cyclize and obtain (S)-3-hydroxytetrahydrofuran.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from chiral pool synthesis to this catalytic asymmetric hydrogenation route represents a substantial opportunity for cost optimization and risk mitigation. By replacing expensive, supply-constrained chiral starting materials with a bulk commodity chemical like ethyl 4-chloroacetoacetate, manufacturers can decouple their production costs from the volatility of the fine chemical chiral market. The elimination of stoichiometric reducing agents such as lithium aluminum hydride or sodium borohydride not only reduces raw material expenses but also drastically simplifies waste management protocols, as the process generates significantly less hazardous inorganic waste. Furthermore, the use of common, recyclable solvents like ethanol and methanol aligns with modern green chemistry principles, reducing the environmental compliance burden and associated disposal costs for large-scale facilities.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy of the hydrogenation step and the low catalyst loading required to achieve turnover. Although the iridium and ruthenium complexes are precious metal-based, their catalytic nature means they are used in minute quantities relative to the substrate, and the potential for catalyst recovery or recycling further amortizes this cost over large production batches. The streamlined two-step sequence reduces the number of unit operations, labor hours, and equipment occupancy time compared to multi-step chiral pool routes, leading to a lower overall cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: Sourcing reliability is significantly improved because the primary raw material, ethyl 4-chloroacetoacetate, is produced on a massive industrial scale for various applications, ensuring a stable and continuous supply unlike niche chiral acids. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures without sensitive enzymatic components, minimizes the risk of batch failures due to minor process deviations. This reliability translates into more predictable lead times for API manufacturers, allowing for tighter inventory control and reduced safety stock requirements for this critical diabetes and oncology drug intermediate.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, utilizing standard high-pressure hydrogenation reactors that are commonplace in modern pharmaceutical manufacturing plants. The absence of cryogenic conditions or extremely high vacuum requirements simplifies the engineering controls needed for scale-up, facilitating a smoother technology transfer from R&D to production. Additionally, the reduction in hazardous waste streams and the use of less toxic reagents contribute to a smaller environmental footprint, helping companies meet increasingly stringent regulatory standards for sustainable manufacturing practices without sacrificing production volume.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for (S)-3-hydroxytetrahydrofuran. These answers are derived directly from the experimental data and comparative analysis provided in patent CN110396072B, offering clarity on the practical benefits of adopting this dual-catalyst methodology over traditional alternatives. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the advantages of this method over traditional chiral pool synthesis?
A: Unlike traditional methods relying on expensive chiral malic acid or carnitine, this patent utilizes cheap ethyl 4-chloroacetoacetate. The dual-catalyst system achieves high optical purity (99% ee) without the need for stoichiometric reducing agents like lithium aluminum hydride, significantly reducing waste and cost.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scale-up. It operates at moderate hydrogen pressures (1-8 MPa) and temperatures (20-150°C) using standard high-pressure reactors. The use of common solvents like ethanol and methanol further facilitates easy solvent recovery and process safety.
Q: What is the optical purity of the final product?
A: The method consistently yields (S)-3-hydroxytetrahydrofuran with an optical purity of 99%. This high enantiomeric excess is critical for its application in synthesizing potent APIs like Empagliflozin and Afatinib, ensuring minimal impurity burden in downstream drug synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-Hydroxytetrahydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development and commercialization of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering (S)-3-hydroxytetrahydrofuran with stringent purity specifications, leveraging our rigorous QC labs to verify optical purity and chemical integrity before every shipment. Our facility is equipped to handle the specific hydrogenation and cyclization requirements of this patent, guaranteeing a consistent supply of this vital building block for your API synthesis needs.
We invite you to collaborate with us to optimize your supply chain for diabetes and oncology therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this advanced catalytic technology can enhance your project's economic and operational performance.
