Scalable Production of (S)-3-Hydroxytetrahydrofuran via Novel Solid Acid Catalysis
The pharmaceutical industry continuously seeks robust, scalable pathways for synthesizing chiral building blocks essential for antiretroviral therapies. A pivotal development in this domain is detailed in Chinese Patent CN108620103B, which discloses a highly efficient catalytic system for the production of (S)-3-hydroxytetrahydrofuran. This compound serves as a critical intermediate in the synthesis of Amprenavir and Fosamprenavir, key protease inhibitors used in the treatment of HIV/AIDS. The patent introduces a novel catalyst comprising tetravalent metal oxides, specifically titanium dioxide (TiO2) or tin dioxide (SnO2), modified with phosphoric acid (H3PO4). This innovation represents a significant departure from traditional stoichiometric reduction methods, offering a greener, one-step dehydration route from (S)-1,2,4-butanetriol. By leveraging solid acid catalysis, the process achieves yields up to 95% while rigorously maintaining optical purity, addressing long-standing challenges in impurity control and process intensification for fine chemical manufacturing.
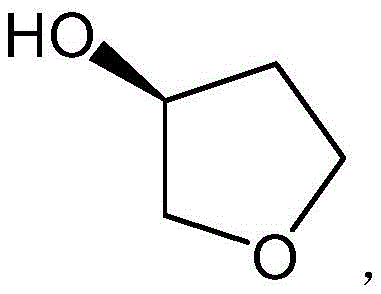
The Limitations of Conventional Methods vs. The Novel Approach
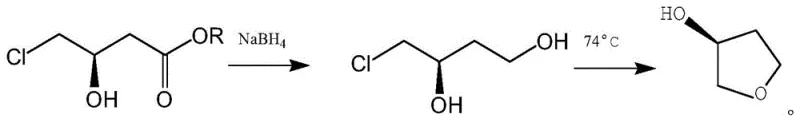
The Limitations of Conventional Methods
Historically, the industrial synthesis of (S)-3-hydroxytetrahydrofuran has relied heavily on multi-step sequences originating from (S)-4-chloro-3-hydroxybutyric acid ethyl ester. As illustrated in the traditional pathway, this route necessitates a reduction step typically employing sodium borohydride (NaBH4) in a tetrahydrofuran solvent system, followed by acid-catalyzed dehydrochlorination and cyclization. While chemically feasible, this legacy approach suffers from severe operational drawbacks that hinder large-scale efficiency. The reduction product contains multiple hydroxyl groups, resulting in extremely high water solubility. This physicochemical property creates a nightmare for downstream processing, as extracting the product from the aqueous phase becomes notoriously difficult and inefficient, leading to substantial yield losses during isolation. Furthermore, the use of stoichiometric reducing agents generates significant amounts of boron-containing waste, complicating environmental compliance and increasing disposal costs for manufacturers aiming for sustainable operations.
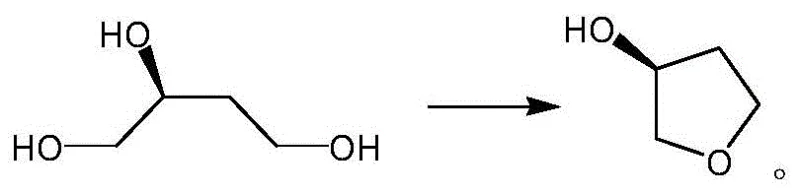
The Novel Approach
In stark contrast to the cumbersome reduction-dehydration sequence, the methodology outlined in CN108620103B streamlines the synthesis into a single, elegant catalytic dehydration step. By utilizing (S)-1,2,4-butanetriol as the direct precursor, the process bypasses the need for halogenated intermediates and stoichiometric reductants entirely. The core of this innovation lies in the use of a fixed-bed reactor loaded with H3PO4-modified metal oxides. This setup allows for the continuous vaporization and reaction of the triol substrate over the solid acid catalyst surface. The reaction operates under mild thermal conditions, typically between 100°C and 150°C, facilitated by an inert gas flow such as nitrogen. This shift from batch liquid-phase chemistry to a potentially continuous gas-solid phase process drastically simplifies the workflow. The product, being more volatile than the polyol starting material, can be separated directly via distillation at the reactor outlet, effectively eliminating the aqueous extraction bottleneck that plagues the conventional chloro-ester route.
Mechanistic Insights into H3PO4-Modified Metal Oxide Catalysis
The exceptional performance of this catalytic system stems from the synergistic interaction between the tetravalent metal oxide support and the phosphate modifier. During the catalyst preparation, the metal oxide (TiO2 or SnO2) is soaked in phosphoric acid and subsequently calcined at high temperatures ranging from 600°C to 650°C. This thermal treatment induces the formation of strong Brønsted and Lewis acid sites on the catalyst surface. The phosphate groups anchor onto the metal oxide lattice, creating a superacidic environment that is sufficiently potent to protonate the hydroxyl groups of the (S)-1,2,4-butanetriol substrate. This protonation facilitates the elimination of a water molecule, triggering an intramolecular nucleophilic attack by the remaining hydroxyl group onto the adjacent carbon, thereby closing the tetrahydrofuran ring. Crucially, the solid nature of the catalyst ensures that the reaction environment remains heterogeneous, preventing the leaching of acidic species into the product stream which could otherwise catalyze unwanted side reactions or racemization.
From an impurity control perspective, the mechanism offers distinct advantages for maintaining stereochemical integrity. Conventional liquid-phase acid catalysis often risks epimerization of the chiral center at the 3-position due to prolonged exposure to harsh acidic media. However, the fixed-bed configuration employed in this novel method minimizes the residence time of the product in the hot zone. As soon as the (S)-3-hydroxytetrahydrofuran is formed, it is swept away by the inert gas flow and condensed. Experimental data from the patent indicates that this rapid removal, combined with the specific acidity profile of the P/TiO2 and P/SnO2 catalysts, preserves the optical purity of the starting material almost perfectly. Yields reaching 95% with optical purities (o.p.) exceeding 99% demonstrate that the catalyst selectively promotes the desired 5-exo-tet cyclization without compromising the chiral information inherent in the (S)-1,2,4-butanetriol feedstock.
How to Synthesize (S)-3-Hydroxytetrahydrofuran Efficiently
Implementing this catalytic dehydration process requires precise control over catalyst preparation and reactor parameters to maximize throughput and selectivity. The protocol involves generating the active catalyst phase by impregnating the metal oxide precursor with phosphoric acid at molar ratios ranging from 1:9 to 81:9 (P:Metal), followed by rigorous calcination. Once the catalyst is granulated and loaded into the fixed-bed reactor, the system is purged with nitrogen to establish an oxygen-free environment. The reactant, an aqueous solution of (S)-1,2,4-butanetriol, is then pumped into the heated zone where it vaporizes and contacts the catalyst bed. The efficiency of this transformation is governed by the mass space velocity and the temperature gradient across the bed. For a comprehensive understanding of the specific operational parameters, including exact calcination times and flow rates required for optimal conversion, please refer to the standardized synthesis guide below.
- Prepare the catalyst by soaking tetravalent metal oxides (TiO2 or SnO2) in phosphoric acid solution, drying, and calcining at 600-650°C to create active acidic sites.
- Load the granular catalyst into a batch-type fixed-bed reactor and purge with inert gas (nitrogen) while heating the catalytic bed to 100-150°C.
- Pump an aqueous solution of (S)-1,2,4-butanetriol through the vaporized catalytic bed at a controlled mass space velocity, collecting the product via distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this solid-acid catalyzed dehydration route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The most significant advantage lies in the drastic simplification of the unit operations. By eliminating the sodium borohydride reduction step, manufacturers remove the need for handling hazardous reducing agents and the subsequent quenching procedures that generate large volumes of saline wastewater. This reduction in chemical consumption directly translates to lower raw material costs and reduced expenditure on waste treatment infrastructure. Furthermore, the shift from a batch extraction process to a continuous distillation-based separation significantly shortens the production cycle time, allowing for faster turnover of batches and improved asset utilization within the manufacturing facility.
- Cost Reduction in Manufacturing: The economic impact of replacing stoichiometric reagents with a reusable solid catalyst cannot be overstated. In the conventional route, the cost of sodium borohydride and the solvents required for extraction constitutes a major portion of the variable costs. The new method utilizes a durable catalyst that can operate for extended periods in a fixed bed, amortizing the catalyst cost over tons of product. Additionally, the elimination of complex aqueous workups reduces the energy load associated with drying organic layers and recovering solvents. This streamlined process flow results in substantial cost savings per kilogram of API intermediate produced, enhancing the overall margin profile for the final drug substance.
- Enhanced Supply Chain Reliability: Supply chain resilience is often compromised by dependencies on specialized reagents with long lead times or volatile pricing. Sodium borohydride and specific chloro-ester precursors can be subject to market fluctuations and regulatory scrutiny regarding transport. In contrast, the feedstock for this new method, (S)-1,2,4-butanetriol, is a stable polyol that is easier to store and handle. The robustness of the fixed-bed reactor system also means that production is less susceptible to the bottlenecks typical of batch processing, such as filtration delays or emulsion formation during extraction. This reliability ensures a more consistent output of high-purity pharmaceutical intermediates, securing the supply line for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling chemical processes often magnifies safety and environmental risks, particularly when dealing with exothermic reductions and corrosive acids. The fixed-bed dehydration process described in the patent is inherently safer and more scalable. The heat of reaction is managed efficiently within the catalyst bed, and the absence of bulk liquid acid reduces corrosion risks in large-scale reactors. From an environmental standpoint, the process aligns with green chemistry principles by minimizing waste generation (E-factor) and avoiding the use of heavy metals or toxic halogenated solvents. This compliance facilitates smoother regulatory approvals and reduces the carbon footprint of the manufacturing site, a critical metric for modern pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of the H3PO4-modified catalyst system for producing (S)-3-hydroxytetrahydrofuran. These insights are derived directly from the experimental data and claims presented in the patent literature, providing clarity on reaction conditions, catalyst longevity, and product quality assurance for technical decision-makers evaluating this technology for adoption.
Q: What are the primary advantages of the H3PO4-modified catalyst over conventional p-toluenesulfonic acid methods?
A: The solid acid catalyst described in patent CN108620103B eliminates the need for homogeneous acid neutralization and separation steps, allowing for continuous fixed-bed operation and significantly simplifying downstream purification while maintaining optical purity above 98%.
Q: How does this process address the extraction difficulties associated with traditional reduction routes?
A: Unlike conventional routes starting from chloro-esters which generate highly water-soluble byproducts requiring complex extraction, this direct dehydration method utilizes a gas-phase or vapor-phase reaction in a fixed bed, allowing the product to be separated directly via distillation without aqueous workup.
Q: Is the optical purity of the starting material preserved during the cyclization?
A: Yes, the mild reaction conditions (80-180°C) and the specific nature of the modified metal oxide catalyst ensure that the chiral center is not racemized, yielding (S)-3-hydroxytetrahydrofuran with optical purity equivalent to the starting (S)-1,2,4-butanetriol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-Hydroxytetrahydrofuran Supplier
The technological advancements detailed in patent CN108620103B underscore the evolving landscape of chiral intermediate synthesis, where efficiency and purity are paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex catalytic dehydrations and fixed-bed operations, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of (S)-3-hydroxytetrahydrofuran meets the exacting standards required for antiretroviral drug synthesis.
We invite procurement leaders and R&D teams to collaborate with us to optimize their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable sourcing strategy available in the market.
