Advanced Chiral Zinc Complexes for Asymmetric Catalysis and Commercial Scale-up
Introduction to Next-Generation Chiral Zinc Catalysts
The landscape of asymmetric catalysis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A pivotal advancement in this domain is documented in patent CN102627616A, which discloses a novel class of chiral zinc-nitrogen coordination compounds. These complexes, specifically bis(oxazolinyl)acetonitrile zinc derivatives, represent a strategic leap forward in catalyst design, moving away from expensive precious metals toward more sustainable and cost-effective earth-abundant alternatives. The technology leverages the unique reactivity of tetracyanoethylene combined with chiral amino alcohols to construct robust catalytic architectures. For R&D directors and procurement strategists, this innovation signals a potential paradigm shift in how key chiral building blocks are manufactured, offering a route that balances high enantioselectivity potential with economic viability. The ability to synthesize these complexes through a relatively straightforward reflux process underscores their potential for immediate integration into existing fine chemical supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral oxazoline ligands and their corresponding metal complexes has been a bottleneck in process chemistry. Conventional routes often involve multi-step syntheses requiring harsh conditions, sensitive reagents, and extensive purification protocols that drive up both time and cost. Many established catalysts rely on scarce transition metals such as rhodium, iridium, or ruthenium, which introduce significant volatility in pricing and supply chain risks. Furthermore, the preparation of the ligands themselves frequently necessitates the use of protecting groups and difficult cyclization steps that result in lower overall yields and higher waste generation. These factors collectively impede the scalability of processes relying on older generation catalysts, making them less attractive for the production of commodity-level chiral intermediates where margin compression is a constant pressure. The environmental footprint associated with heavy metal residues also poses substantial regulatory hurdles for pharmaceutical manufacturers aiming for greener processes.
The Novel Approach
In stark contrast, the methodology outlined in the patent presents a streamlined, one-pot strategy that dramatically simplifies the construction of the catalytic center. By utilizing tetracyanoethylene as a versatile precursor, the process facilitates the in situ formation of the bis-oxazoline framework directly upon reaction with chiral amino alcohols like L-phenylglycinol or L-phenylalaninol. This approach eliminates the need for pre-synthesized, complex ligands, thereby reducing the number of unit operations and the associated handling costs. The use of anhydrous zinc chloride as the metal source not only lowers the raw material cost but also ensures that the resulting complex is free from the toxicological concerns associated with heavy metals. The reaction proceeds efficiently in chlorobenzene under reflux, a condition that is easily replicable in standard industrial reactors. This simplicity translates directly into a more robust and reliable manufacturing protocol that can be scaled with confidence.
Mechanistic Insights into Zinc-Catalyzed Cyanosilylation
The efficacy of these chiral zinc complexes stems from the precise coordination geometry established between the zinc ion and the nitrogen atoms of the newly formed oxazoline rings. Mechanistically, the zinc center acts as a potent Lewis acid, activating the carbonyl group of the substrate, such as benzaldehyde, towards nucleophilic attack by trimethylsilyl cyanide (TMSCN). The chiral environment provided by the phenyl or benzyl substituents on the oxazoline rings dictates the facial selectivity of this attack, leading to the formation of enantiomerically enriched cyanohydrin ethers. The presence of the nitrile groups on the acetonitrile backbone may further influence the electronic properties of the metal center, fine-tuning its Lewis acidity and stability during the catalytic cycle. Understanding this interplay is crucial for R&D teams looking to optimize reaction parameters for specific substrates beyond benzaldehyde. The stability of the complex under the reaction conditions ensures that the catalyst maintains its integrity throughout the transformation, minimizing decomposition and the formation of achiral background products.
From an impurity control perspective, the mechanism offers distinct advantages. The well-defined coordination sphere reduces the likelihood of non-selective background reactions that often plague less organized catalytic systems. The byproduct profile is generally cleaner, primarily consisting of silylated species that are easily removed during workup. This inherent selectivity reduces the burden on downstream purification processes, such as crystallization or chromatography, which are often the most costly steps in API intermediate manufacturing. For quality assurance teams, this means a more consistent impurity profile and a higher probability of meeting stringent pharmacopeial standards without extensive reprocessing. The ability to predict and control the stereochemical outcome based on the choice of amino alcohol (phenylglycinol vs. phenylalaninol) provides a powerful tool for accessing either enantiomer of the desired product simply by switching the starting material.
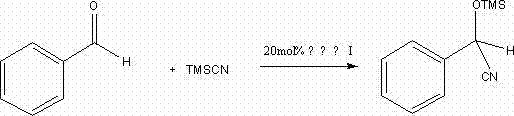
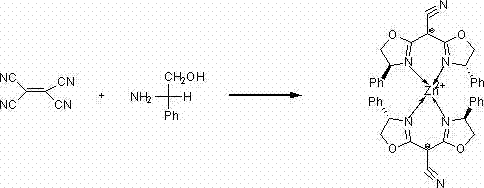
How to Synthesize Bis(oxazolinyl)acetonitrile Zinc Complexes Efficiently
The synthesis protocol described in the patent is designed for reproducibility and scalability, making it an ideal candidate for technology transfer. The process begins with the careful preparation of an anhydrous and oxygen-free environment, typically achieved through nitrogen or argon purging, which is critical for preventing the hydrolysis of the zinc chloride catalyst and the degradation of the sensitive nitrile intermediates. The reaction mixture, comprising tetracyanoethylene, the chosen chiral amino alcohol, and the zinc catalyst in chlorobenzene, is subjected to prolonged reflux to ensure complete conversion and cyclization. Following the reaction, the workup involves a straightforward aqueous extraction and solvent removal, followed by purification via column chromatography to isolate the pure white crystalline complex. Detailed standardized operating procedures for scaling this synthesis from gram to kilogram quantities are essential for maintaining batch-to-batch consistency.
- Reflux tetracyanoethylene with L-phenylglycinol or L-phenylalaninol in chlorobenzene solvent using anhydrous ZnCl2 as a catalyst for 48 to 72 hours under inert atmosphere.
- Remove the chlorobenzene solvent under reduced pressure, dissolve the residue in water, and perform extraction using chloroform to isolate the organic phase.
- Dry the organic phase, remove the solvent, and purify the crude product via column chromatography using petroleum ether and dichloromethane to obtain white crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-based catalytic technology offers compelling strategic benefits that extend beyond mere technical performance. The primary driver is the substantial reduction in raw material costs achieved by replacing precious metal catalysts with zinc, a commodity metal with a stable and abundant global supply. This shift mitigates the risk of price spikes associated with geopolitical instability in platinum-group metal mining regions. Additionally, the simplified synthetic route reduces the number of processing steps, which directly correlates to lower energy consumption, reduced solvent usage, and decreased labor hours per kilogram of product. These efficiencies compound to create a significantly lower cost of goods sold (COGS), providing a competitive edge in price-sensitive markets. The robustness of the chlorobenzene reflux process also implies a high degree of operational reliability, minimizing the risk of batch failures that can disrupt production schedules.
- Cost Reduction in Manufacturing: The elimination of expensive noble metals and complex ligand pre-synthesis steps leads to a drastic simplification of the bill of materials. By utilizing tetracyanoethylene and common chiral amino alcohols, manufacturers can leverage existing supply chains for bulk chemicals, avoiding the premiums associated with specialty catalysts. The reduction in process complexity also lowers the capital expenditure required for specialized equipment, as standard glass-lined or stainless steel reactors are sufficient for the reflux conditions. Furthermore, the high atom economy of the cyclization reaction minimizes waste disposal costs, contributing to a leaner and more profitable manufacturing model. These cumulative savings allow for more aggressive pricing strategies or improved margins on high-value chiral intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures a resilient supply chain that is less susceptible to shortages. Unlike proprietary ligands that may be sourced from a single vendor, the precursors for these zinc complexes are commoditized chemicals with multiple global suppliers. This diversification of supply sources reduces the risk of production stoppages due to vendor-specific issues. The stability of the final zinc complex also facilitates easier storage and transportation, extending shelf life and reducing the need for cold-chain logistics. For supply chain planners, this translates to greater flexibility in inventory management and the ability to respond more rapidly to fluctuations in market demand without compromising on quality or lead times.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations that are standard in the fine chemical industry. The use of chlorobenzene, while requiring appropriate handling, is a well-understood solvent in industrial settings with established recovery and recycling protocols. The absence of heavy metal contaminants in the final product simplifies the regulatory approval process for pharmaceutical applications, as residual metal limits are easier to meet with zinc than with palladium or nickel. This alignment with green chemistry principles enhances the sustainability profile of the manufacturing site, aiding in compliance with increasingly stringent environmental regulations. The potential for solvent recycling further reduces the environmental footprint, making the process attractive for companies with aggressive carbon reduction targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral zinc complex technology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent documentation. They serve to clarify the operational parameters and potential benefits for stakeholders evaluating this technology for adoption. Understanding these nuances is critical for making informed decisions about process development and investment.
Q: What are the primary advantages of using these chiral zinc complexes over traditional transition metal catalysts?
A: These zinc-based complexes offer a significant cost advantage due to the abundance and low price of zinc compared to precious metals like palladium or ruthenium. Furthermore, the synthesis utilizes readily available starting materials like tetracyanoethylene and chiral amino alcohols, simplifying the supply chain and reducing overall production costs.
Q: What is the catalytic efficiency of these complexes in cyanosilylation reactions?
A: According to patent data, these complexes demonstrate effective catalytic performance in the cyanosilylation of benzaldehyde. Specific embodiments report conversion rates reaching up to 60% and 48% respectively for the different complex variants, indicating viable activity for fine chemical synthesis applications.
Q: Are the synthesis conditions suitable for large-scale manufacturing?
A: Yes, the process relies on standard industrial unit operations such as reflux in chlorobenzene and column chromatography purification. The use of common solvents and stable reagents suggests that the pathway is amenable to scale-up from laboratory to commercial production volumes with appropriate engineering controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Zinc Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the chiral zinc complexes described in CN102627616A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these laboratory-scale innovations into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from pilot plant to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify enantiomeric excess and chemical purity. Our commitment to quality ensures that every batch of chiral intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to unlock the full value of this catalytic system for your specific application. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing bottlenecks. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only cost-effective but also technically superior, positioning your organization for long-term success in the competitive landscape of chiral chemical manufacturing.
