Advanced Chiral Zinc-Nitrogen Coordination Compounds for Scalable Asymmetric Catalysis in Fine Chemicals
Advanced Chiral Zinc-Nitrogen Coordination Compounds for Scalable Asymmetric Catalysis in Fine Chemicals
The landscape of asymmetric synthesis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A significant advancement in this field is documented in patent CN102850383A, which discloses a novel chiral zinc-nitrogen coordination compound. This organometallic complex, specifically a bis(4-isopropyl-4,5-dihydro-2-oxazolinyl)acetonitrile zinc complex, represents a strategic breakthrough for manufacturers seeking cost-effective alternatives to precious metal catalysts. The invention details a robust synthetic pathway utilizing tetracyanoethylene and D-valinol, resulting in a stable crystalline structure capable of inducing chirality in various organic transformations. For R&D directors and procurement specialists, this technology offers a compelling value proposition by combining the economic benefits of zinc chemistry with the stereocontrol typically associated with more expensive systems.
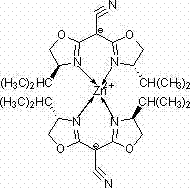
The structural integrity of this catalyst is confirmed through rigorous single-crystal X-ray diffraction analysis, revealing a precise orthorhombic crystal system. This level of structural definition is critical for process chemists who need to understand the steric environment around the metal center to predict substrate compatibility. By leveraging this specific coordination geometry, the complex facilitates reactions such as allyl alkylation, Baylis-Hillman coupling, Henry reactions, and silylcyanation with notable conversion efficiencies. The ability to achieve up to 68% conversion in silylcyanation reactions underscores the potential of this material to serve as a reliable workhorse in the synthesis of complex pharmaceutical building blocks, addressing the industry's need for versatile and scalable catalytic solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral intermediates has relied heavily on catalysts based on precious transition metals such as palladium, rhodium, or ruthenium. These conventional methods often suffer from significant drawbacks, including the exorbitant cost of the metal precursors and the stringent requirement for removing trace metal residues from the final active pharmaceutical ingredients (APIs). Furthermore, many established chiral ligands require multi-step synthetic routes involving hazardous reagents and protecting group strategies, which inflate the overall cost of goods sold (COGS). The environmental footprint of these processes is also a growing concern, as the disposal of heavy metal waste streams necessitates complex treatment protocols. For supply chain managers, the reliance on geographically concentrated sources of precious metals introduces volatility and risk into the procurement strategy, potentially leading to disruptions in the continuity of supply for critical drug substances.
The Novel Approach
In contrast, the methodology presented in patent CN102850383A introduces a streamlined approach centered on zinc, an abundant and non-toxic metal. The novel synthesis involves a direct reflux reaction between tetracyanoethylene and D-valinol in the presence of anhydrous zinc chloride within a chlorobenzene solvent system. This one-pot style coordination effectively bypasses the need for pre-synthesized, expensive chiral ligands, thereby simplifying the supply chain for the catalyst itself. The process operates under anhydrous and anaerobic conditions to ensure the formation of the desired coordination sphere, yielding a stable complex that can be isolated as white crystals. This shift towards base metal catalysis not only aligns with green chemistry principles by reducing toxicity but also offers a pathway to substantial cost reduction in fine chemical manufacturing by eliminating the dependency on volatile precious metal markets.
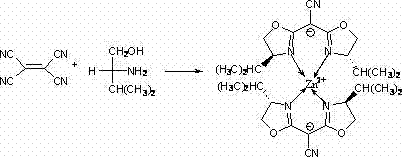
Mechanistic Insights into Zinc-Catalyzed Asymmetric Transformations
The catalytic efficacy of this chiral zinc-nitrogen complex stems from its unique ability to activate electrophilic substrates through Lewis acid coordination while simultaneously providing a chiral pocket for nucleophilic attack. The zinc center, coordinated by the nitrogen atoms of the bis(oxazoline) moiety, creates a rigid chiral environment that differentiates between the enantiotopic faces of the substrate. In reactions such as the silylcyanation of aldehydes, the zinc atom likely coordinates to the carbonyl oxygen, increasing its electrophilicity and directing the approach of the cyanide source. The steric bulk provided by the isopropyl groups on the oxazoline rings plays a pivotal role in shielding one face of the molecule, thereby inducing enantioselectivity. Understanding this mechanistic nuance is vital for R&D teams aiming to optimize reaction parameters such as temperature, solvent polarity, and catalyst loading to maximize both yield and optical purity for specific target molecules.
Furthermore, the stability of the catalyst under reaction conditions is a key factor in its practical utility. The patent data indicates that the complex maintains its integrity during prolonged reflux periods, suggesting a high activation energy for decomposition. This thermal stability is crucial for scaling up reactions where heat transfer limitations might otherwise lead to catalyst degradation. Additionally, the mechanism likely involves a dynamic equilibrium between the catalyst and the substrate, allowing for turnover without permanent deactivation. For process development, this implies that the catalyst loading could potentially be optimized to lower levels once the kinetics are fully understood, further driving down the cost per kilogram of the produced intermediate. The ability to function effectively in diverse reaction types, from carbon-carbon bond formation to nucleophilic additions, highlights the versatility of this zinc coordination sphere.
How to Synthesize Bis(4-isopropyl-4,5-dihydro-2-oxazolinyl)acetonitrile Zinc Complex Efficiently
The preparation of this high-value catalyst is designed to be robust and reproducible, utilizing standard laboratory equipment that translates well to pilot plant operations. The process begins with the careful exclusion of moisture and oxygen, which is critical for preventing the hydrolysis of the zinc chloride and the oxidation of sensitive intermediates. The reaction mixture is subjected to extended reflux in chlorobenzene, a high-boiling solvent that ensures sufficient thermal energy for the cyclization and coordination steps to proceed to completion. Following the reaction, the workup involves a straightforward extraction and purification sequence that yields the product in a crystalline form suitable for characterization and immediate use in downstream catalytic applications. Detailed standardized synthesis steps are provided in the guide below to ensure consistency across batches.
- Combine tetracyanoethylene and D-valinol in chlorobenzene solvent under strict anhydrous and anaerobic conditions with anhydrous ZnCl2 catalyst.
- Maintain the reaction mixture at reflux temperature for approximately 36 to 72 hours to ensure complete coordination and cyclization.
- Remove solvent under reduced pressure, dissolve residue in water, extract with chloroform, and purify the precipitate via column chromatography using petroleum ether and dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-based catalytic technology presents a multitude of strategic benefits that extend beyond simple reagent substitution. The primary advantage lies in the decoupling of production costs from the fluctuating prices of precious metals, offering a more predictable and stable cost structure for long-term manufacturing contracts. The use of commodity chemicals like tetracyanoethylene and amino alcohols as precursors ensures a reliable supply chain, as these materials are produced on a massive scale for various industrial applications. This abundance mitigates the risk of supply shortages that often plague niche chiral ligands, thereby enhancing the overall resilience of the manufacturing operation against market volatility and geopolitical disruptions.
- Cost Reduction in Manufacturing: The transition to a zinc-based catalyst system inherently lowers the raw material costs associated with the catalytic cycle. Unlike palladium or rhodium catalysts which require expensive recovery processes to meet regulatory limits on heavy metals in APIs, zinc residues are generally less toxic and easier to manage, potentially simplifying the purification workflow. This elimination of complex metal scavenging steps reduces both the consumption of auxiliary materials and the time required for downstream processing. Consequently, the overall operational expenditure is significantly reduced, allowing for more competitive pricing of the final pharmaceutical intermediates without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: The synthetic route for the catalyst itself relies on widely available starting materials, ensuring that the production of the catalyst can be scaled up rapidly to meet surging demand. The robustness of the synthesis, which tolerates standard reflux conditions, means that it can be manufactured in existing multipurpose reactors without the need for specialized high-pressure or cryogenic equipment. This ease of manufacture translates to shorter lead times for catalyst replenishment, ensuring that production schedules for the final API intermediates remain uninterrupted. Suppliers can thus guarantee a continuous flow of materials, a critical metric for just-in-time manufacturing environments prevalent in the modern pharmaceutical industry.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the use of zinc aligns better with increasingly stringent global regulations regarding heavy metal discharge. The waste streams generated from this process are less hazardous compared to those containing platinum group metals, reducing the cost and complexity of waste treatment and disposal. Furthermore, the high thermal stability of the complex supports scale-up to multi-ton production volumes, as the reaction kinetics remain favorable even in larger reactor vessels where heat transfer is less efficient. This scalability ensures that the technology remains viable from early-stage clinical trial material production through to commercial launch, providing a seamless path for process validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral zinc complex in industrial settings. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering a clear picture of the technology's capabilities and limitations. Understanding these factors is essential for making informed decisions about integrating this catalyst into existing synthetic routes for high-value fine chemicals.
Q: What are the primary advantages of this zinc complex over traditional transition metal catalysts?
A: This chiral zinc-nitrogen complex offers a distinct advantage by utilizing earth-abundant zinc rather than expensive precious metals like palladium or rhodium. The synthesis utilizes readily available precursors such as tetracyanoethylene and D-valinol, significantly reducing raw material costs. Furthermore, the complex demonstrates robust catalytic activity across multiple reaction types including allyl alkylation and silylcyanation, providing a versatile tool for process chemists.
Q: How does the purification process impact the overall yield and scalability?
A: The purification protocol described in the patent involves standard workup procedures including solvent removal, aqueous dissolution, and organic extraction followed by column chromatography. While column chromatography can be a bottleneck at very large scales, the formation of white crystals with a defined melting point and solubility profile suggests that recrystallization could be developed as a more scalable alternative for industrial production, thereby enhancing supply chain reliability.
Q: Is this catalyst suitable for continuous flow manufacturing processes?
A: Given the thermal stability indicated by the reflux conditions in chlorobenzene and the solid-state stability confirmed by single-crystal X-ray diffraction, this complex holds potential for immobilization on solid supports. Such immobilization would facilitate its adaptation into continuous flow reactors, a key requirement for modern pharmaceutical manufacturing aiming to reduce lead times and improve safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Zinc Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the chiral zinc-nitrogen complex described in CN102850383A. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of catalyst or intermediate meets the exacting standards required by global regulatory bodies. We are committed to delivering high-purity pharmaceutical intermediates that drive your drug development programs forward with speed and certainty.
We invite you to collaborate with our technical team to explore how this cost-effective zinc catalysis can be integrated into your specific synthetic challenges. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your production volume. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for efficiency, sustainability, and long-term success in the competitive global marketplace.
