Optimizing L-Menthyl Glyoxylate Production for Commercial Scale-Up and Purity
Optimizing L-Menthyl Glyoxylate Production for Commercial Scale-Up and Purity
The pharmaceutical industry continuously seeks robust synthetic routes for critical chiral intermediates that balance high purity with economic viability. A pivotal development in this domain is documented in patent CN102516078A, which outlines an innovative preparation method for L-menthyl glyoxylate and its monohydrate. This compound serves as a fundamental chiral building block for the industrial production of vital antiviral agents such as Lamivudine and Emtricitabine. The technological breakthrough described herein addresses long-standing challenges regarding yield optimization and impurity control, offering a streamlined pathway that eliminates the need for hazardous reagents often associated with traditional synthesis. By leveraging a specific esterification-oxidation sequence, this methodology ensures consistent quality essential for regulatory compliance in global drug manufacturing.
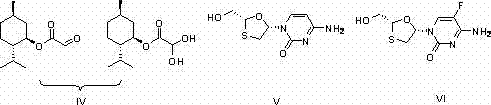
For R&D directors evaluating process feasibility, the structural integrity and stereochemical purity of the intermediate are paramount. The patent details a route that preserves the chiral center of L-menthol throughout the transformation, minimizing racemization risks that could compromise the efficacy of the final active pharmaceutical ingredient. The ability to produce both the anhydrous ester and the stable monohydrate form provides flexibility for downstream processing, allowing manufacturers to select the most stable form for storage or immediate reaction. This dual capability enhances the reliability of the supply chain, ensuring that high-purity pharmaceutical intermediates are available without the bottlenecks typically caused by unstable reactive species.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-menthyl glyoxylate has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Early literature describes routes relying on expensive and toxic reagents such as silver nitrate or tributyltin hydride, which not only escalate raw material costs but also introduce severe environmental and safety liabilities. For instance, methods involving ozonolysis require specialized infrastructure to manage explosion risks associated with ozone and peroxide by-products, making them unsuitable for large-scale facilities. Furthermore, traditional esterification processes often suffer from poor selectivity, leading to difficult purification steps and substantial product loss, thereby reducing the overall economic feasibility of the manufacturing process.
The Novel Approach
In contrast, the novel approach presented in the patent data utilizes a highly efficient two-step sequence that drastically simplifies the operational workflow while enhancing safety profiles. By reacting L-menthol with readily available haloacetyl halides or anhydrides, the process achieves esterification yields exceeding 95% under mild conditions without the need for azeotropic water removal. The subsequent oxidation step employs dimethyl sulfoxide (DMSO) activated by phosphorus pentoxide or iodide salts, avoiding the use of heavy metal oxidants like potassium permanganate. This strategic shift not only reduces the burden on waste treatment systems but also ensures cost reduction in API manufacturing by minimizing the consumption of precious catalysts and simplifying the downstream isolation of the target molecule.
Mechanistic Insights into DMSO-Mediated Oxidation
The core of this technological advancement lies in the precise mechanistic control of the oxidation step, where DMSO acts as both solvent and oxygen donor. In the presence of activators like P2O5, the reaction proceeds through a sulfonium intermediate that facilitates the conversion of the alpha-halo ester to the corresponding glyoxylate with high chemoselectivity. This mechanism is particularly advantageous because it operates effectively at temperatures between 140-160°C, providing sufficient energy to drive the reaction to completion while maintaining the stability of the sensitive chiral ester linkage. The careful control of stoichiometry, specifically the molar ratios of DMSO to substrate, ensures that over-oxidation to carboxylic acids is suppressed, thereby maximizing the yield of the desired aldehyde functionality.
Impurity control is another critical aspect addressed by this mechanistic design, as the formation of side products is minimized through the specific choice of halogenated precursors. The process allows for the formation of a bisulfite adduct, which serves as a powerful purification tool to separate the target glyoxylate from unreacted starting materials and non-carbonyl impurities. Upon hydrolysis of this adduct with formaldehyde, the pure monohydrate is recovered with purity levels reaching 99.5% or higher. This level of purity is essential for meeting the stringent specifications required by regulatory bodies for intermediates used in the synthesis of nucleoside analogues, ensuring that the final drug substance is free from genotoxic impurities associated with halogenated by-products.
How to Synthesize L-Menthyl Glyoxylate Efficiently
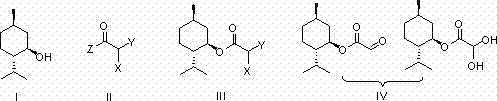
Implementing this synthesis route requires strict adherence to anhydrous conditions during the initial esterification and oxidation phases to prevent hydrolysis of the reactive acyl halides and the intermediate esters. The protocol dictates the use of dichloromethane as a solvent for the first step, facilitating easy removal of pyridine hydrochloride salts via filtration, which streamlines the workup procedure significantly. Operators must monitor the reaction progress via TLC to ensure complete consumption of L-menthol before proceeding to the oxidation stage, as residual alcohol can interfere with the activation of DMSO. Detailed standardized synthesis steps see the guide below for precise temperature ramps and quenching procedures.
- React L-menthol with haloacetyl halide or anhydride in dichloromethane with pyridine to form the intermediate haloacetate ester.
- Oxidize the intermediate using DMSO activated by P2O5 or iodide salts at controlled temperatures between 140-160°C.
- Purify the crude product via recrystallization or convert to monohydrate using sodium bisulfite and formaldehyde treatment.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this manufacturing route offers substantial strategic benefits by decoupling production from volatile markets for precious metals and hazardous gases. The reliance on commodity chemicals such as DMSO, chloroacetyl chloride, and L-menthol ensures a stable supply chain that is less susceptible to geopolitical disruptions or sudden price spikes associated with specialty reagents. Moreover, the elimination of expensive catalysts like silver salts directly translates to significant cost savings in raw material procurement, allowing for more competitive pricing structures in long-term supply agreements. The simplified workup also reduces solvent consumption and waste disposal costs, further enhancing the overall economic efficiency of the production lifecycle.
- Cost Reduction in Manufacturing: The process eliminates the need for costly reagents such as silver nitrate and toxic tributyltin hydride, which traditionally inflate the bill of materials for chiral intermediates. By substituting these with economical alternatives like DMSO and iodide salts, the direct material costs are significantly lowered without compromising reaction efficiency. Additionally, the high yield of the esterification step minimizes the loss of the chiral pool starting material, L-menthol, which is a value-added component, thereby optimizing the utilization of expensive raw inputs.
- Enhanced Supply Chain Reliability: Utilizing widely available industrial chemicals ensures that production schedules are not dictated by the lead times of niche reagents. The robustness of the reaction conditions allows for flexible manufacturing planning, reducing the risk of batch failures that could disrupt the supply of critical antiviral intermediates. This reliability is crucial for maintaining continuous operations in the pharmaceutical sector, where downtime can have cascading effects on the availability of life-saving medications.
- Scalability and Environmental Compliance: The absence of ozone generation and heavy metal waste simplifies the environmental permitting process for new production lines. The aqueous workup and recrystallization steps are easily adaptable to large-scale reactors, facilitating the transition from pilot plant to full commercial production. This scalability ensures that suppliers can rapidly respond to increased market demand for nucleoside analogues while adhering to increasingly strict global environmental regulations regarding chemical discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific synthetic methodology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, focusing on practical aspects of industrial application. Understanding these nuances helps stakeholders make informed decisions about process adoption and supplier qualification.
Q: What are the primary advantages of this synthesis method over conventional ozonolysis?
A: Unlike ozonolysis which poses explosion hazards and requires complex equipment, this method utilizes mild DMSO-based oxidation, significantly improving operational safety and reducing infrastructure costs for industrial production.
Q: How does this process impact the purity profile of the final intermediate?
A: The specific workup procedures involving sodium bisulfite adduct formation effectively remove trace impurities, consistently achieving purity levels exceeding 99.5% suitable for stringent pharmaceutical applications.
Q: Is this route scalable for large volume API manufacturing?
A: Yes, the process avoids expensive reagents like silver nitrate and toxic tin hydrides, utilizing readily available raw materials and standard solvent systems that facilitate seamless commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Menthyl Glyoxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of antiviral therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of L-menthyl glyoxylate meets the exacting standards required for GMP manufacturing environments. Our commitment to quality assurance guarantees that your downstream synthesis remains uninterrupted by impurity-related issues.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production needs, ensuring a partnership built on transparency, technical excellence, and mutual success.
