Advanced Synthesis of L-Menthyl Glyoxylate: A Breakthrough for Antiviral API Manufacturing
The pharmaceutical industry's relentless pursuit of efficient antiviral therapies has placed significant demand on the supply chain for high-quality chiral intermediates. Patent CN102516078B introduces a transformative preparation method for L-menthyl glyoxylate and its monohydrate, a critical building block for nucleoside analogs like Lamivudine and Emtricitabine. This technology addresses long-standing inefficiencies in chiral synthesis by replacing hazardous and costly reagents with a robust DMSO-based oxidation system. By leveraging readily available L-menthol and simple haloacetyl derivatives, this route achieves total yields exceeding 72 percent for the ester and over 80 percent for the monohydrate, with purity levels surpassing 99.5 percent. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and economically viable manufacturing processes for complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-menthyl glyoxylate has been plagued by significant technical and economic barriers that hinder large-scale adoption. Early literature describes routes relying on silver nitrate for nitrate ester formation or potassium permanganate for oxidative cleavage, both of which involve prohibitively expensive reagents and generate substantial heavy metal waste. Other methods utilize ozonolysis of maleate or fumarate esters, which introduces severe safety hazards due to the explosive nature of ozone and peroxide intermediates, making them unsuitable for ton-scale production. Furthermore, approaches employing tributyltin hydride for reduction pose acute toxicity risks and complicate regulatory compliance for API manufacturing. These conventional pathways often suffer from low selectivity, requiring complex purification steps that erode overall yield and drive up the cost of goods sold, thereby limiting their utility for reliable agrochemical intermediate or pharma intermediate suppliers.
The Novel Approach
The methodology disclosed in CN102516078B fundamentally reengineers the synthetic pathway to prioritize safety, cost-efficiency, and scalability. Instead of relying on precious metals or dangerous oxidants, this novel approach utilizes a two-step sequence beginning with the esterification of L-menthol with haloacetyl halides or anhydrides. The subsequent oxidation step employs dimethylsulfoxide (DMSO) activated by phosphorus pentoxide or iodide salts, a strategy that effectively converts the haloacetate intermediate into the target glyoxylate under controlled thermal conditions. This eliminates the need for cryogenic ozonolysis or toxic tin reagents, drastically simplifying the operational workflow. The process demonstrates exceptional versatility, accommodating both monohalo and dihalo precursors to optimize yield and purity profiles. By streamlining the reaction sequence and utilizing commodity chemicals, this method offers a compelling solution for cost reduction in pharmaceutical intermediate manufacturing while maintaining the rigorous quality standards required for chiral drug synthesis.
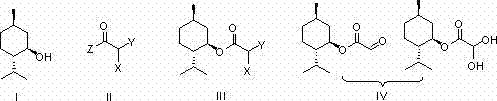
Mechanistic Insights into DMSO-Mediated Oxidative Conversion
The core innovation of this technology lies in the precise activation of DMSO to facilitate the oxidation of the alpha-halo ester moiety without compromising the chiral center of the menthol group. In the monohaloacetate pathway, the reaction initiates with the thermal activation of the substrate in DMSO, followed by the addition of phosphorus pentoxide (P2O5). This generates a highly reactive sulfonium species that promotes the displacement of the halide and subsequent hydrolysis to the aldehyde function. The reaction temperature is carefully maintained between 140°C and 160°C to ensure complete conversion while minimizing thermal degradation. For the dihaloacetate route, the mechanism shifts slightly to utilize iodide salts as catalysts, which likely facilitate the formation of an iodo-intermediate that is more susceptible to DMSO oxidation. This mechanistic flexibility allows manufacturers to select the most cost-effective starting material—whether chloroacetyl chloride or dichloroacetyl chloride—without sacrificing product quality, providing a strategic advantage in raw material sourcing for high-purity OLED material or pharma chemical production.
Impurity control is meticulously managed through a unique workup procedure involving sodium bisulfite and formaldehyde. Following the oxidation, the crude reaction mixture is treated with sodium bisulfite to form a water-soluble hydroxysulfonate adduct. This step effectively separates the desired aldehyde from non-reactive organic impurities and residual starting materials that remain in the organic phase. Subsequent hydrolysis with formaldehyde regenerates the free aldehyde as a stable monohydrate precipitate. This crystallization-driven purification is critical for achieving the reported purity of greater than 99.5 percent, as it physically excludes trace by-products that might otherwise co-elute during chromatographic separation. For quality assurance teams, this built-in purification mechanism ensures consistent batch-to-batch reproducibility and reduces the reliance on resource-intensive column chromatography, thereby enhancing the commercial scale-up of complex pharmaceutical additives.
How to Synthesize L-Menthyl Glyoxylate Efficiently
Implementing this synthesis route requires strict adherence to anhydrous conditions during the oxidation phase to prevent premature hydrolysis of the reactive intermediates. The process begins with the preparation of the haloacetate ester, where L-menthol is reacted with the acylating agent in dichloromethane using pyridine as an acid scavenger. Once the intermediate is isolated, it is subjected to the DMSO oxidation protocol, with careful monitoring of temperature and reagent stoichiometry to maximize conversion. The detailed standardized synthesis steps, including specific molar ratios and workup parameters, are outlined below to guide process engineers in replicating these high-yield results.
- Esterify L-menthol with haloacetyl halide or anhydride in dichloromethane using pyridine as a base to form the intermediate haloacetate.
- Oxidize the intermediate using DMSO activated by either P2O5 (for monohaloacetates) or iodide salts (for dihaloacetates) at elevated temperatures.
- Purify the crude product through bisulfite adduct formation and formaldehyde hydrolysis to obtain the high-purity monohydrate.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patent offers profound benefits by decoupling production from volatile markets for precious metals and hazardous gases. The substitution of silver nitrate and ozone with DMSO and phosphorus reagents significantly lowers the raw material cost baseline and mitigates supply risk associated with specialized reagents. The elimination of heavy metal catalysts also reduces the environmental burden of waste treatment, leading to substantial cost savings in effluent management and regulatory compliance. Furthermore, the high total yield of the process means that less raw material is required to produce the same amount of finished product, directly improving the margin structure for manufacturers of fine chemical intermediates. This efficiency translates into a more resilient supply chain capable of meeting surging demand for antiviral medications without the bottlenecks typical of older, lower-yielding technologies.
- Cost Reduction in Manufacturing: The replacement of expensive reagents like silver salts and tributyltin hydride with commodity chemicals such as DMSO and iodide salts drives down the direct material costs significantly. Additionally, the simplified workup procedure reduces solvent consumption and labor hours associated with complex purification steps. By avoiding the need for specialized equipment required for ozonolysis or cryogenic reactions, capital expenditure for new production lines is also minimized. These factors collectively contribute to a leaner manufacturing model that enhances competitiveness in the global market for specialty chemicals.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials like L-menthol and chloroacetyl chloride ensures a stable supply base that is less susceptible to geopolitical disruptions. The robustness of the reaction conditions allows for flexible scheduling and easier scale-up, reducing lead times for high-purity pharmaceutical intermediates. Manufacturers can maintain higher inventory turnover rates due to the predictable nature of the synthesis, ensuring continuous availability for downstream API producers. This reliability is crucial for maintaining uninterrupted production schedules in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing standard reactor configurations and avoiding exothermic runaways common in oxidation reactions. The absence of toxic heavy metals simplifies the disposal of chemical waste, aligning with increasingly stringent environmental regulations. The high atom economy of the esterification and oxidation steps minimizes waste generation, supporting sustainability goals. This eco-friendly profile not only reduces compliance costs but also enhances the brand reputation of suppliers committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process feasibility and product quality. Understanding these details is essential for technical teams evaluating the transition to this improved manufacturing route.
Q: What are the critical advantages of the DMSO/P2O5 oxidation system over traditional methods?
A: Unlike traditional methods requiring expensive silver salts or toxic tributyltin hydride, the DMSO/P2O5 system utilizes cost-effective reagents and avoids heavy metal contamination, significantly simplifying downstream purification and waste treatment.
Q: How does this process ensure high optical purity for chiral drug synthesis?
A: The process starts with naturally derived L-menthol and employs mild reaction conditions that prevent racemization. The final crystallization steps effectively remove minor impurities, ensuring the chiral integrity required for Lamivudine and Emtricitabine production.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the patent explicitly highlights the suitability for industrial production due to the use of common solvents like dichloromethane and DMSO, avoiding hazardous ozonolysis steps and achieving total yields exceeding 72%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Menthyl Glyoxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antiviral therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature are successfully translated into industrial reality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of L-menthyl glyoxylate meets the exacting standards required for GMP API synthesis. Our commitment to process excellence allows us to deliver consistent quality while optimizing the cost structure for our global partners.
We invite procurement leaders and R&D directors to collaborate with us on optimizing their supply chains for chiral intermediates. By leveraging our expertise in this DMSO-based oxidation technology, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical partner available in the market today.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
