Advanced One-Step Synthesis of Trisubstituted Pyrazole Derivatives for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously demand more efficient pathways to access privileged heterocyclic scaffolds, among which the pyrazole core remains paramount due to its ubiquitous presence in bioactive molecules. A significant technological breakthrough in this domain is documented in Chinese Patent CN110105285B, which discloses a novel, highly efficient preparation method for trisubstituted pyrazole derivatives. This intellectual property introduces a transformative Lewis acid-catalyzed ring contraction strategy that converts 2,5-dihydro-1,4,5-thiadiazepine oxides directly into highly functionalized pyrazoles. Unlike conventional methodologies that often suffer from harsh reaction conditions and poor atom economy, this innovation utilizes zinc bromide as a cost-effective catalyst in dichloromethane at room temperature. For R&D directors and process chemists seeking to optimize their synthetic routes, this patent offers a compelling solution that merges high yield with operational simplicity, representing a critical advancement for the reliable supply of complex pharmaceutical intermediates.
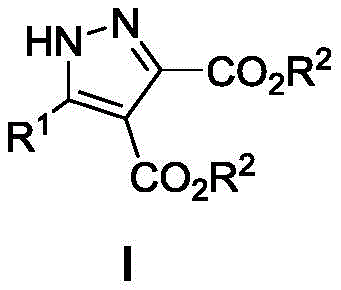
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrazole ring, particularly when heavily substituted at the 3, 4, and 5 positions, has presented substantial synthetic challenges that impact both cost and timeline in drug development. Traditional approaches frequently rely on the condensation of hydrazines with 1,3-dicarbonyl compounds, a process that often lacks regioselectivity, leading to difficult-to-separate isomeric mixtures that compromise the purity profile essential for API manufacturing. Furthermore, many existing protocols necessitate the use of expensive transition metal catalysts, such as palladium or rhodium complexes, which not only inflate raw material costs but also introduce stringent requirements for residual metal control in the final product. These legacy methods often involve multi-step sequences with intermediate isolation, requiring extreme temperatures or hazardous reagents that pose safety risks and complicate the commercial scale-up of complex polymer additives or active ingredients. The cumulative effect of these inefficiencies is a prolonged lead time and elevated production costs that strain supply chain reliability.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology described in the patent data leverages a unique ring contraction mechanism that streamlines the synthesis into a single, high-yielding step. By utilizing 2,5-dihydro-1,4,5-thiadiazepine oxides as the starting material, the process bypasses the regioselectivity issues inherent in classical pyrazole formation. The use of zinc bromide as a Lewis acid catalyst activates the substrate effectively under ambient conditions, eliminating the need for energy-intensive heating or cooling systems. This approach not only simplifies the reaction setup by removing the requirement for inert gas protection but also ensures a cleaner reaction profile with fewer byproducts. The result is a robust protocol that delivers trisubstituted pyrazole derivatives with exceptional efficiency, directly addressing the industry's need for cost reduction in electronic chemical manufacturing and pharmaceutical intermediate production through process intensification.
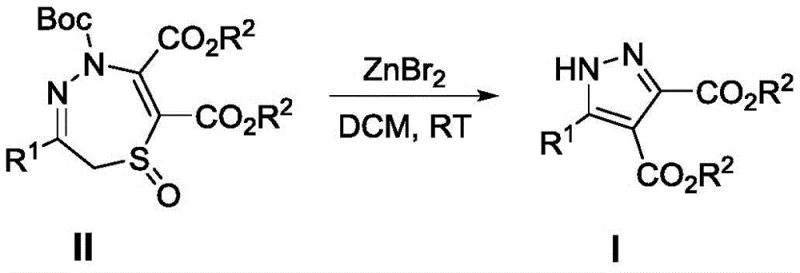
Mechanistic Insights into ZnBr2-Catalyzed Ring Contraction
The core chemical innovation driving this synthesis lies in the Lewis acid-mediated activation of the thiadiazepine oxide scaffold, which triggers a cascade of bond cleavages and rearrangements to form the stable pyrazole aromatic system. Zinc bromide acts as a potent electrophile, coordinating with the oxygen atom of the sulfoxide group and potentially the nitrogen atoms within the seven-membered ring. This coordination weakens the sulfur-nitrogen and carbon-sulfur bonds, facilitating the extrusion of the sulfur moiety and the subsequent contraction of the seven-membered heterocycle into the five-membered pyrazole ring. This mechanistic pathway is particularly elegant because it avoids the formation of reactive radical intermediates that often lead to polymerization or decomposition in other metal-catalyzed systems. The mild nature of the zinc salt ensures that sensitive functional groups on the R1 substituent, such as esters or halogens, remain intact throughout the transformation, preserving the chemical integrity required for downstream coupling reactions.
From an impurity control perspective, this mechanism offers distinct advantages for maintaining high-purity OLED material or API standards. Because the reaction proceeds through a concerted ionic pathway rather than a radical chain reaction, the generation of tarry byproducts or oligomeric impurities is significantly suppressed. The stoichiometry is straightforward, typically employing a 1:1 molar ratio of substrate to catalyst, which minimizes the introduction of excess reagents that would require extensive washing or purification steps. The stability of the intermediate species under the reaction conditions (room temperature in DCM) allows for a controlled conversion rate, preventing runaway exotherms that could degrade product quality. Consequently, the crude reaction mixture is often clean enough that simple column chromatography yields the final product with the stringent purity specifications demanded by regulatory bodies, thereby reducing the burden on QC labs and analytical teams.
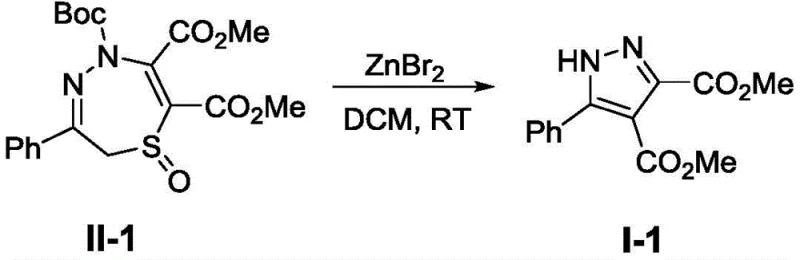
How to Synthesize Trisubstituted Pyrazole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and reproducibility as outlined in the patent examples. The process begins with the dissolution of the thiadiazepine oxide precursor in anhydrous dichloromethane to maintain a concentration of approximately 0.1M, ensuring optimal interaction between the substrate and the catalyst. The addition of anhydrous zinc bromide is critical, and the mixture is stirred at ambient temperature (25°C) for a duration of roughly 8 hours to ensure complete consumption of the starting material. Following the reaction, the workup is remarkably simple, involving the removal of the solvent under reduced pressure followed by purification via silica gel chromatography. For a comprehensive understanding of the precise operational parameters and safety considerations, the detailed standardized synthesis steps are provided in the guide below.
- Dissolve the 2,5-dihydro-1,4,5-thiadiazepine oxide precursor (Compound II) and anhydrous zinc bromide (1.0 equivalent) in dichloromethane to achieve a concentration of 0.1M.
- Stir the reaction mixture at room temperature (approximately 25°C) for 8 hours until the starting material is completely consumed as monitored by TLC.
- Remove the solvent under reduced pressure and purify the resulting crude residue via silica gel column chromatography using a petroleum ether and ethyl acetate gradient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc bromide-catalyzed route represents a strategic opportunity to optimize the cost structure and reliability of the supply chain for heterocyclic intermediates. By shifting away from precious metal catalysts to abundant zinc salts, the direct material costs are drastically simplified, removing the volatility associated with the pricing of rare earth or noble metals. Furthermore, the elimination of inert atmosphere requirements reduces the capital expenditure needed for specialized reactor setups, allowing for utilization of standard glass-lined or stainless steel equipment that is readily available in most multipurpose chemical plants. This accessibility translates into a more resilient supply network where production is not bottlenecked by equipment availability or specialized operator training.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive catalytic systems with inexpensive zinc bromide and the reduction of unit operations. Since the reaction occurs in a single step at room temperature, there is a substantial saving in energy costs associated with heating or cryogenic cooling, which are typical in traditional pyrazole syntheses. Additionally, the high conversion rates and clean reaction profiles minimize the loss of valuable starting materials, improving the overall mass balance and reducing the cost of goods sold (COGS) per kilogram of finished intermediate. The simplified workup procedure also lowers labor costs and solvent consumption during the purification phase.
- Enhanced Supply Chain Reliability: The robustness of this chemical transformation enhances supply continuity by reducing the risk of batch failures due to sensitive reaction conditions. Because the process does not rely on air-sensitive reagents or strict moisture exclusion beyond standard anhydrous solvent practices, the risk of operational errors is minimized. The starting materials, specifically the thiadiazepine oxides, are accessible via established synthetic routes, ensuring that the upstream supply chain remains stable. This reliability is crucial for maintaining consistent inventory levels and meeting the just-in-time delivery schedules required by downstream pharmaceutical manufacturers.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is facilitated by the absence of exothermic hazards and the use of common organic solvents like dichloromethane, which are well-understood in industrial waste management systems. The avoidance of heavy metal contaminants simplifies the environmental compliance landscape, as there is no need for complex heavy metal scavenging steps or specialized disposal protocols for toxic catalyst residues. This aligns with modern green chemistry principles, making the process more attractive for companies aiming to reduce their environmental footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrazole synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide clarity on feasibility and application. Understanding these details is essential for technical teams evaluating the integration of this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this ZnBr2-catalyzed method over traditional pyrazole synthesis?
A: This method eliminates the need for expensive precious metal catalysts and complex multi-step sequences. It operates under mild room temperature conditions in a common solvent (DCM), significantly simplifying the operational workflow and reducing energy consumption compared to high-temperature or cryogenic traditional methods.
Q: What is the substrate scope for the R1 and R2 groups in this synthesis?
A: The process demonstrates excellent versatility. R1 can accommodate phenyl, substituted phenyl (including electron-donating methoxy/methyl and electron-withdrawing trifluoromethyl/fluoro groups), heterocyclic, alkyl, and alkenyl substituents. R2 is compatible with both methyl and ethyl ester groups, allowing for diverse downstream functionalization.
Q: Is inert gas protection required for this reaction?
A: No, one of the key operational benefits highlighted in the patent data is that the reaction proceeds efficiently without the need for inert gas protection (such as nitrogen or argon atmosphere), which further reduces equipment complexity and operational costs for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trisubstituted Pyrazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative academic and patent research into commercially viable reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in laboratory settings are faithfully reproduced at an industrial level. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of trisubstituted pyrazole derivatives meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to quality assurance ensures that the structural integrity and purity of these complex intermediates are maintained throughout the manufacturing process.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your supply chain.
