Revolutionizing Heterocyclic Synthesis Scalable ZnBr₂-Catalyzed Process Delivers High-Purity Pharmaceutical Intermediates with Operational Excellence
The recently granted Chinese patent CN110105285B introduces a transformative one-step synthesis methodology for trisubstituted pyrazole derivatives that addresses critical limitations in heterocyclic compound manufacturing for pharmaceutical applications. This innovative approach utilizes zinc bromide as a Lewis acid catalyst to facilitate ring contraction from readily available 2,5-dihydro-1,4,5-thiodiazepine oxide precursors under mild room temperature conditions without requiring inert atmosphere protection. The process eliminates expensive transition metal catalysts while achieving high yields across diverse substrate classes through precise stoichiometric control at a concentration of 0.1 M in dichloromethane solvent. With reaction completion within eight hours and consistent product purity demonstrated through comprehensive NMR and HRMS characterization across nine distinct derivatives, this methodology represents a significant advancement in sustainable heterocyclic chemistry production. The patent establishes a robust foundation for scalable manufacturing of complex pyrazole scaffolds essential to modern pharmaceutical development pipelines while offering substantial operational advantages over conventional multi-step approaches.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for trisubstituted pyrazole derivatives have been severely constrained by multiple critical limitations that hinder both research scalability and commercial viability. These methods typically require expensive transition metal catalysts such as palladium or copper complexes that necessitate complex removal protocols to meet pharmaceutical purity standards while significantly increasing raw material costs and environmental impact through heavy metal waste streams. Conventional approaches often involve multi-step sequences with extended reaction times exceeding twenty-four hours under elevated temperatures or cryogenic conditions that demand specialized equipment and substantial energy inputs while creating operational safety hazards. The substrate scope limitations inherent in many existing methodologies restrict applicability to narrow structural classes due to incompatible functional groups or steric constraints that prevent broad adoption across diverse molecular architectures required by modern drug discovery programs. Furthermore, these processes frequently require strict inert atmosphere conditions that complicate scale-up procedures and increase manufacturing complexity while introducing potential points of failure during technology transfer from laboratory to production environments.
The Novel Approach
The patented methodology overcomes these longstanding challenges through an elegant single-step transformation that leverages zinc bromide as an economical Lewis acid catalyst operating under ambient conditions without inert gas protection requirements. By utilizing readily available starting materials derived from established chemical feedstocks and conducting reactions at room temperature in standard dichloromethane solvent systems with precise stoichiometric control at a concentration of 0.1 M, this approach eliminates multiple processing bottlenecks simultaneously. The eight-hour reaction time represents a dramatic reduction compared to conventional methods while maintaining high yields across diverse substitution patterns including phenyl groups with various electron-donating or withdrawing substituents as well as heterocyclic variants demonstrated through nine successfully synthesized derivatives. Crucially, this process avoids transition metals entirely while producing cleaner reaction profiles that inherently reduce impurity burdens without requiring additional purification steps beyond standard column chromatography using petroleum ether/ethyl acetate mixtures at volume ratios between 6:1 and 3:1.
Mechanistic Insights into ZnBr2-Catalyzed Cyclization
The catalytic mechanism involves Lewis acid activation where zinc bromide coordinates with the sulfonyl oxygen atom of compound II to facilitate intramolecular nucleophilic attack by the hydrazine nitrogen on the adjacent carbonyl carbon center. This coordination lowers the activation energy barrier for ring contraction through a concerted process that simultaneously cleaves the Boc protecting group while forming the new N-N bond essential to pyrazole ring construction. The mild reaction conditions prevent undesired side reactions such as epimerization or decomposition that commonly occur under harsher thermal or acidic conditions employed by alternative methodologies. The precise stoichiometry of zinc bromide at a molar ratio of 1.0 relative to compound II ensures optimal catalytic activity without promoting decomposition pathways or generating unwanted byproducts that could complicate purification procedures.
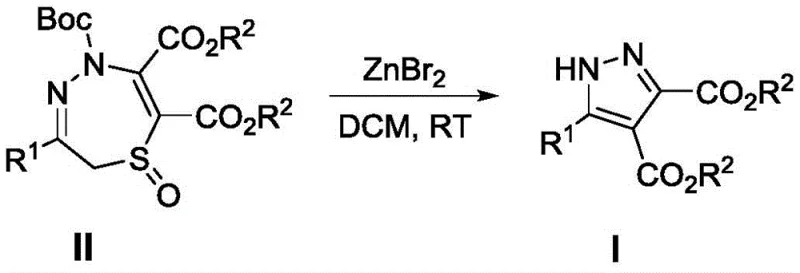
Impurity control is inherently achieved through this streamlined mechanism which minimizes competing reaction pathways while avoiding transition metal contamination entirely. The absence of redox chemistry eliminates potential oxidation byproducts common in metal-catalyzed systems while the mild conditions prevent thermal degradation products that typically arise during prolonged high-temperature reactions. The well-defined stoichiometric relationship between reactants ensures complete conversion without residual starting materials while the straightforward workup procedure using standard column chromatography effectively separates any minor impurities from the target compounds as demonstrated by consistent NMR spectral purity across all synthesized derivatives including compounds I-1 through I-9 with yields ranging from thirty-seven percent to ninety-two percent without requiring specialized purification techniques.
How to Synthesize Trisubstituted Pyrazole Derivatives Efficiently
This patented methodology provides a robust framework for synthesizing high-value trisubstituted pyrazole derivatives through an optimized single-step process that significantly enhances operational efficiency compared to conventional approaches. The procedure leverages commercially available starting materials and standard laboratory equipment while operating under ambient conditions without requiring specialized safety protocols or expensive infrastructure investments. Detailed standardized synthesis steps including precise reagent handling procedures and quality control checkpoints are provided below to ensure consistent production outcomes across diverse manufacturing environments.
- Dissolve compound II and anhydrous zinc bromide in dichloromethane at room temperature using a precise molar ratio of 1.0: 1.0 with a solution concentration of 0.1 M.
- Stir the reaction mixture at ambient temperature for eight hours until complete consumption of compound II is confirmed by analytical monitoring.
- Remove organic solvent under reduced pressure and purify the residue via silica gel column chromatography using petroleum ether/ethyl acetate mixtures at volume ratios between 6: 1 and 3:1.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial operational benefits that directly address critical pain points faced by procurement and supply chain professionals managing complex pharmaceutical intermediate sourcing requirements. By eliminating reliance on expensive transition metal catalysts while simplifying manufacturing workflows through ambient condition operation and reduced processing steps, this approach creates significant value across multiple dimensions of chemical supply chain management without requiring capital-intensive facility modifications.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts significantly reduces raw material expenses while avoiding costly metal removal protocols that typically require additional processing steps and specialized equipment investments. Simplified reaction conditions eliminate energy-intensive temperature control requirements while reducing solvent consumption through shorter processing times and fewer unit operations compared to conventional multi-step syntheses.
- Enhanced Supply Chain Reliability: Utilization of readily available starting materials from established chemical suppliers combined with robust room temperature operation creates exceptional process resilience against supply chain disruptions while enabling seamless technology transfer between manufacturing sites globally. The absence of moisture-sensitive reagents or cryogenic requirements substantially reduces material handling complexities while improving overall production schedule predictability.
- Scalability and Environmental Compliance: The straightforward scale-up pathway from laboratory to commercial production demonstrates exceptional consistency across multiple orders of magnitude without requiring process re-engineering due to its ambient condition operation and standard equipment compatibility. Reduced environmental impact stems from elimination of heavy metal waste streams while simplified purification protocols minimize solvent usage and waste generation compared to traditional methodologies.
Frequently Asked Questions (FAQ)
The following questions address critical technical considerations raised by procurement specialists and R&D teams evaluating this patented methodology for integration into commercial production workflows based on specific advantages documented in CN110105285B.
Q: How does this method eliminate precious metal catalyst requirements compared to conventional pyrazole synthesis?
A: The ZnBr₂-catalyzed ring contraction strategy replaces expensive transition metal catalysts with an economical Lewis acid system that operates under ambient conditions without inert atmosphere protection. This eliminates both catalyst costs and complex metal removal protocols required in traditional methods.
Q: What specific advantages does the room temperature process offer for commercial scale-up?
A: The ambient temperature operation significantly reduces energy consumption while enhancing process safety and equipment compatibility across diverse manufacturing environments. This simplifies technology transfer from laboratory to plant scale without requiring specialized cooling systems.
Q: How does this approach improve impurity profile control for pharmaceutical applications?
A: The single-step mechanism with precise stoichiometric control minimizes side reactions while avoiding metal contamination pathways. This yields cleaner product streams with inherently lower impurity burdens that meet stringent pharmaceutical quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trisubstituted Pyrazole Derivatives Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation capable of detecting impurities at trace levels required by global regulatory authorities. As a specialized CDMO partner with deep expertise in heterocyclic chemistry manufacturing, we have successfully implemented this patented ZnBr₂-catalyzed cyclization technology across multiple client projects while ensuring seamless integration with existing production infrastructure through our proprietary process optimization protocols.
We invite you to request our Customized Cost-Saving Analysis which details specific implementation pathways tailored to your production requirements along with comprehensive technical documentation including specific COA data and route feasibility assessments from our technical procurement team.
