Advanced Synthetic Route for Fingolimod Hydrochloride: Commercial Scalability and Purity Control
The pharmaceutical landscape for immunosuppressants has been significantly transformed by the introduction of Fingolimod hydrochloride, widely known as FTY-720, which stands as the first oral disease-modifying therapy approved for relapsing-remitting multiple sclerosis. The commercial viability of this critical active pharmaceutical ingredient relies heavily on the efficiency and robustness of its synthetic pathway. Patent CN103804123A discloses a groundbreaking method and intermediate for synthesizing Fingolimod hydrochloride that addresses the longstanding inefficiencies of prior art. This technical insight report analyzes the novel route which utilizes a unique octyl halopropyl benzene intermediate to streamline production. By shifting away from traditional multi-step esterifications and complex protecting group strategies, this methodology offers a direct path to high-purity material. For R&D directors and procurement managers, understanding the mechanistic advantages of this patent is crucial for securing a reliable Fingolimod hydrochloride supplier capable of meeting stringent global regulatory standards while optimizing manufacturing costs.
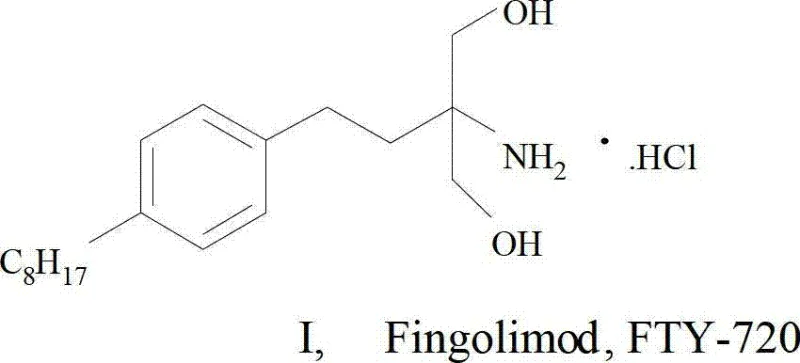
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Fingolimod hydrochloride has been plagued by excessive reaction steps and poor overall yields, creating significant bottlenecks for commercial scale-up of complex pharmaceutical intermediates. Early methods, such as those described in US5604229, relied on phenylethyl ethanoate as a starting material and required up to ten distinct reaction steps to construct the necessary amino-propanediol structure. This elongated sequence resulted in a dismal total recovery rate of approximately 4%, rendering the process economically unfeasible for large-volume production. Furthermore, alternative routes attempting to shorten the sequence often introduced hazardous reagents or unstable intermediates, such as alpha-bromoacetophenone, which possesses lachrymatory properties and poses safety risks in an industrial setting. Other approaches utilizing vinylbenzene suffered from raw material instability due to spontaneous polymerization at room temperature, increasing the difficulty of industrialized production and compromising supply chain continuity. These conventional methods frequently necessitated rigorous column chromatography for purification, a technique that is notoriously difficult to scale and generates substantial chemical waste.
The Novel Approach
In stark contrast to the cumbersome legacy processes, the novel approach detailed in the patent data utilizes a strategically designed intermediate, octyl halopropyl benzene, to drastically simplify the synthetic architecture. This new route reduces the total reaction steps from ten down to merely five, representing a fundamental shift in process efficiency. The core innovation lies in the construction of the carbon skeleton via Friedel-Crafts acylation followed by a highly efficient reduction using triethylsilane, which avoids the use of expensive and difficult-to-remove transition metal catalysts often found in hydrogenation steps for ketone reduction. By employing this specific intermediate, the synthesis bypasses the need for unstable enolate chemistry and complex ester manipulations. The result is a process that is not only shorter but also operates under milder reaction conditions, significantly reducing energy consumption and operational complexity. This streamlined methodology ensures that the commercial scale-up of complex pharmaceutical intermediates is achievable with standard reactor equipment, thereby enhancing supply chain reliability and reducing lead time for high-purity pharmaceutical intermediates.
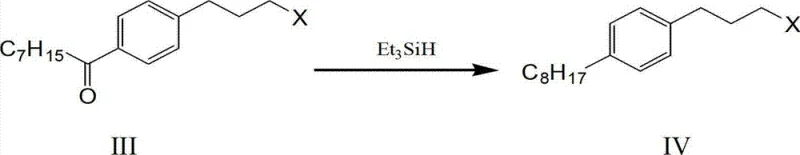
Mechanistic Insights into Triethylsilane Reduction and One-Pot Condensation
The mechanistic elegance of this synthesis is best observed in the reduction step where capryloyl halopropyl benzene is converted to the key octyl halopropyl benzene intermediate. Unlike traditional catalytic hydrogenation which requires high pressure and specialized safety protocols, this method employs triethylsilane in the presence of trifluoroacetic acid. This reagent combination facilitates a rapid and selective reduction of the ketone carbonyl group to a methylene group without affecting the halogen substituent on the propyl chain. The reaction proceeds with exceptional conversion rates, often exceeding 95% yield for this specific intermediate, which is critical for maintaining mass balance throughout the synthesis. The selectivity of this reduction prevents the formation of alcohol byproducts that would otherwise complicate downstream purification. Furthermore, the use of trifluoroacetic acid as a promoter ensures that the reaction kinetics are favorable at relatively low temperatures, typically between 6°C and 12°C, minimizing thermal degradation of sensitive functional groups. This level of control over the reaction mechanism is essential for R&D directors focused on purity and impurity profile management.
Another critical mechanistic advancement is the implementation of a 'one-pot' strategy for the nitro substitution and formaldehyde condensation steps. In conventional routes, the intermediate octyl phenyl-3-nitropropane often requires isolation and purification, which can lead to yield losses due to the instability of the nitro group. The patented method overcomes this by directly utilizing the reaction filtrate from the nitro substitution step for the subsequent condensation with formaldehyde under alkaline conditions. This telescoping of steps prevents the decomposition of the nitro intermediate and eliminates a unit operation, thereby saving time and solvent. The condensation reaction constructs the 1,3-propanediol backbone with high regioselectivity, ensuring that the amino group is positioned correctly for the final immunosuppressive activity. By avoiding the isolation of unstable intermediates, the process significantly reduces the generation of hazardous waste and simplifies the overall workflow, making it highly attractive for cost reduction in pharmaceutical intermediates manufacturing.
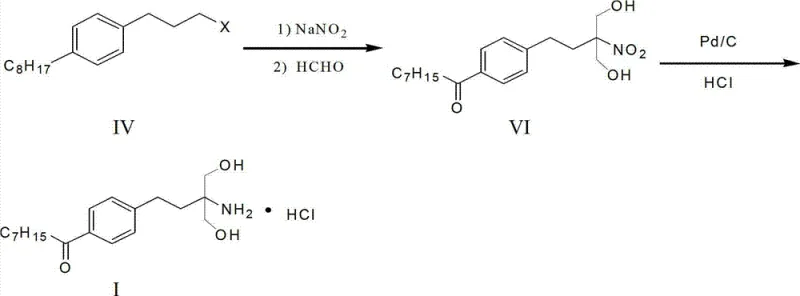
How to Synthesize Fingolimod Hydrochloride Efficiently
The efficient synthesis of Fingolimod hydrochloride relies on the precise execution of the novel intermediate pathway described in the patent data. The process begins with the preparation of the capryloyl halopropyl benzene precursor, followed by the critical silane reduction to generate the octyl halopropyl benzene core. Once this key intermediate is secured, the subsequent nitro substitution and condensation steps can be performed in a telescoped manner to maximize throughput. The final stage involves catalytic hydrogenation to reduce the nitro group to an amine, followed by salification with hydrochloric acid to yield the stable hydrochloride salt. Detailed standardized synthesis steps see the guide below.
- Perform Friedel-Crafts acylation on 3-halogenophenyl propane with capryloyl chloride using Lewis acid to form capryloyl halopropyl benzene.
- Reduce the ketone intermediate using triethylsilane and trifluoroacetic acid to generate the key octyl halopropyl benzene intermediate.
- Execute nitro substitution and formaldehyde condensation followed by catalytic hydrogenation and salification to obtain the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the significant simplification of the supply chain for raw materials. The starting material, 3-bromophenyl propane, is cheaply and easily available on the global market, reducing the risk of supply disruptions that often plague specialized fine chemical manufacturing. By reducing the number of reaction steps from ten to five, the process inherently lowers the consumption of solvents, reagents, and labor hours, leading to substantial cost savings without the need for complex economic modeling. The elimination of column chromatography purification steps further reduces operational expenditures and waste disposal costs, aligning with modern environmental compliance standards. Additionally, the mild reaction conditions mean that existing general-purpose chemical reactors can be utilized, avoiding the need for capital investment in specialized high-pressure or cryogenic equipment. This flexibility enhances supply chain reliability and allows for rapid scaling to meet market demand fluctuations.
- Cost Reduction in Manufacturing: The streamlined five-step sequence drastically reduces the cumulative loss of material associated with multi-step synthesis, directly improving the cost of goods sold. By eliminating the need for expensive transition metal catalysts and complex purification techniques like column chromatography, the operational overhead is significantly lowered. The high yield of the key intermediate ensures that raw material utilization is optimized, minimizing waste generation. Furthermore, the ability to telescope the nitro substitution and condensation steps reduces solvent consumption and energy usage, contributing to a leaner manufacturing process. These factors combine to create a robust economic model that supports competitive pricing strategies in the global API market.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 3-bromophenyl propane and capryloyl chloride ensures a stable supply chain foundation. Unlike routes dependent on unstable monomers like vinylbenzene, this process uses stable intermediates that can be stored and transported without special precautions. The robustness of the reaction conditions means that production is less susceptible to variations in environmental factors or equipment performance. This stability translates to consistent delivery schedules and reduced lead times for high-purity pharmaceutical intermediates. Procurement teams can confidently forecast inventory needs knowing that the production process is resilient to common industrial perturbations.
- Scalability and Environmental Compliance: The process is designed with industrial amplification in mind, utilizing simple separation techniques like extraction and crystallization rather than complex chromatography. This simplicity facilitates easy scale-up from pilot plant to commercial production volumes without significant re-engineering. The reduction in solvent usage and the avoidance of hazardous reagents lower the environmental footprint of the manufacturing process. Waste streams are easier to treat, ensuring compliance with increasingly stringent environmental regulations. The high purity of the final product, exceeding 99.8%, reduces the need for re-processing, further enhancing the sustainability and efficiency of the production lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the patented synthesis of Fingolimod hydrochloride. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. They provide clarity on impurity control, scalability, and the specific advantages of the novel intermediate. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What is the primary advantage of the new intermediate IV in Fingolimod synthesis?
A: The novel octyl halopropyl benzene intermediate allows for a significantly shortened reaction sequence, reducing the total steps from 10 to 5 while improving overall yield from 4% to approximately 31%.
Q: How does this method address impurity control compared to conventional routes?
A: By utilizing a one-pot nitro substitution and condensation strategy, the formation of unstable nitro-ester byproducts is minimized, eliminating the need for complex column purification and ensuring product purity exceeds 99.8%.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process utilizes readily available raw materials like 3-bromophenyl propane and operates under mild reaction conditions without requiring specialized high-pressure equipment, facilitating easy commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fingolimod Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes for high-value immunosuppressants like Fingolimod hydrochloride. Our technical team has extensively analyzed the pathway disclosed in CN103804123A and possesses the expertise to implement this novel intermediate strategy effectively. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the >99.8% purity required for pharmaceutical applications. We are committed to delivering a reliable Fingolimod hydrochloride supplier experience that prioritizes quality, consistency, and regulatory compliance.
We invite global partners to collaborate with us to optimize their supply chains for this critical medication. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. By leveraging our manufacturing capabilities and technical insights, we can help you secure a stable supply of high-quality Fingolimod hydrochloride while achieving significant operational efficiencies.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
