Advanced Manufacturing of Fingolimod Hydrochloride: A Novel Wittig-Based Route for Commercial Scale-Up
The pharmaceutical landscape for immunosuppressants has been significantly reshaped by the introduction of Fingolimod Hydrochloride, a first-in-class sphingosine-1-phosphate receptor modulator approved for the treatment of relapsing-remitting multiple sclerosis. As global demand for this critical therapy escalates, the industry faces an urgent need for manufacturing processes that balance high purity with environmental sustainability and cost efficiency. Patent CN110938003B introduces a transformative preparation method that fundamentally re-engineers the synthetic pathway, moving away from legacy chemistries plagued by heavy metal waste and hazardous reagents. This technical insight report analyzes the strategic value of this novel route, specifically designed for reliable pharmaceutical intermediate supplier networks seeking to optimize their production portfolios. By leveraging a Wittig reaction strategy coupled with catalytic hydrogenation, this method addresses the critical bottlenecks of yield, safety, and scalability that have historically constrained the supply chain of complex immunosuppressants.
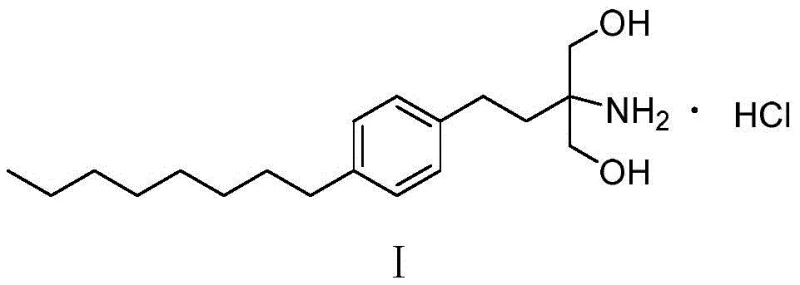
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Fingolimod Hydrochloride has relied heavily on Friedel-Crafts acylation strategies, which present profound challenges for modern green chemistry standards. Traditional routes, such as those utilizing beta-phenylethyl or benzene as starting materials, necessitate the use of stoichiometric amounts of aluminum trichloride, a Lewis acid that generates massive quantities of acidic wastewater and corrosive sludge upon quenching. Furthermore, electrophilic aromatic substitution reactions are inherently prone to forming regioisomers, particularly ortho-substituted byproducts, which are structurally similar to the desired para-isomer and notoriously difficult to separate, thereby compromising the overall purity profile. Beyond the environmental burden, these legacy processes often depend on aggressive chemical reducing agents like lithium aluminum hydride, triethylsilane-titanium tetrachloride, or sodium borohydride-calcium chloride complexes. These reagents not only escalate raw material costs due to their high price points but also introduce significant operational safety hazards, including pyrophoric risks and the generation of toxic silicon or boron-containing waste streams that complicate downstream processing and regulatory compliance.
The Novel Approach
In stark contrast to the cumbersome multi-step sequences of the past, the methodology disclosed in CN110938003B offers a streamlined, convergent synthesis that bypasses the pitfalls of Friedel-Crafts chemistry entirely. The core innovation lies in the construction of the carbon skeleton via a highly selective Wittig reaction between a specialized phosphonium salt and p-octylbenzaldehyde. This approach eliminates the formation of positional isomers at the aromatic ring, ensuring that the structural integrity of the intermediate is maintained with exceptional fidelity from the outset. Moreover, the route replaces dangerous stoichiometric reductions with a elegant catalytic hydrogenation step that simultaneously reduces the nitro group and the carbon-carbon double bond. This dual-reduction capability not only simplifies the process flow by combining multiple transformations into a single operational unit but also drastically improves the atom economy of the synthesis. The result is a robust manufacturing protocol that delivers high-purity Fingolimod Hydrochloride with significantly reduced environmental impact and enhanced operational safety, making it ideally suited for cost reduction in API manufacturing initiatives.
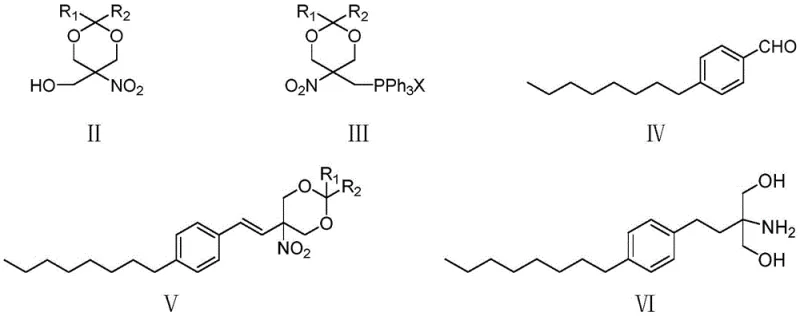
Mechanistic Insights into Wittig Coupling and Catalytic Hydrogenation
The mechanistic elegance of this synthesis begins with the preparation of the Wittig reagent, derived from 2,2-disubstituted-5-hydroxymethyl-5-nitro-1,3-dioxane. The initial halogenation of the hydroxymethyl group followed by nucleophilic substitution with triphenylphosphine creates a stable phosphonium salt that serves as a potent nucleophile. When reacted with p-octylbenzaldehyde under controlled alkaline conditions, typically using sodium methoxide or potassium hydroxide, the ylide attacks the carbonyl carbon to form the styryl double bond with high stereoselectivity. The precision of this coupling is critical; the patent specifies strict molar ratios of base to aldehyde to ensure complete consumption of the starting materials, preventing the accumulation of unreacted aldehyde which could lead to difficult-to-remove impurities in later stages. The use of a nitro-dioxane scaffold is particularly ingenious, as the nitro group acts as a latent amine precursor while the dioxane ring provides temporary protection for the diol functionality, preventing unwanted side reactions during the coupling phase.
Following the formation of the nitro-styryl intermediate, the process employs a sophisticated one-pot reduction and deprotection strategy. Under hydrogen pressure in the presence of palladium on carbon or Raney nickel catalysts, the nitro group is reduced to a primary amine, and the exocyclic double bond is saturated to form the ethyl linker. Crucially, the subsequent addition of aqueous hydrochloric acid triggers the hydrolysis of the acetal protecting group, releasing the free 1,3-diol moiety required for the final drug structure. This tandem operation is a masterclass in process efficiency, as it avoids the isolation of sensitive amine intermediates. The purification mechanism further enhances quality; by adjusting the pH of the reaction mixture, the basic amino product can be selectively partitioned into the aqueous phase while neutral organic impurities remain in the organic layer. This acid-base extraction technique effectively scrubs the product stream of non-amino contaminants, enabling the achievement of purity levels exceeding 99.9% without the need for complex chromatographic separations, a vital feature for high-purity pharmaceutical intermediates.
How to Synthesize Fingolimod Hydrochloride Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and minimize impurity formation. The process begins with the activation of the dioxane precursor, followed by the critical Wittig coupling where temperature and base concentration must be tightly regulated to prevent ylide decomposition. The subsequent hydrogenation step demands careful monitoring of hydrogen uptake to ensure complete reduction without over-reduction or catalyst poisoning. Finally, the hydrolysis and salt formation steps convert the free base into the stable hydrochloride salt suitable for pharmaceutical formulation. For a detailed breakdown of the specific reaction conditions, solvent systems, and workup procedures validated in the patent examples, please refer to the standardized guide below.
- Prepare the Wittig reagent by halogenating 2,2-disubstituted-5-hydroxymethyl-5-nitro-1,3-dioxane followed by substitution with triphenylphosphine.
- Perform a Wittig reaction between the prepared phosphonium salt and p-octylbenzaldehyde under alkaline conditions to form the nitro-styryl intermediate.
- Execute a one-pot catalytic hydrogenation to reduce both the nitro group and carbon-carbon double bond, followed by acid hydrolysis to remove the protecting group and final salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route represents a strategic opportunity to de-risk the supply of Fingolimod Hydrochloride while driving substantial cost efficiencies. The elimination of Friedel-Crafts reagents removes the logistical burden of handling and disposing of large volumes of corrosive aluminum salts, directly translating to lower waste treatment costs and reduced regulatory compliance overhead. Furthermore, the substitution of expensive and hazardous chemical reducing agents with catalytic hydrogenation significantly lowers the raw material cost per kilogram of finished product. The stability of the key intermediates, particularly the phosphonium salt and the aldehyde, ensures a consistent supply chain that is less susceptible to the degradation issues often encountered with reactive alkyl halides used in older methods. This robustness facilitates long-term inventory planning and reduces the risk of batch failures due to raw material instability.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the simplification of the process flow and the replacement of high-cost reagents. By avoiding the use of stoichiometric metal hydrides and silanes, manufacturers can eliminate the procurement costs associated with these premium chemicals. Additionally, the high selectivity of the Wittig reaction minimizes the formation of byproducts, which reduces the loss of valuable starting materials and lowers the cost of goods sold (COGS). The ability to perform reduction and deprotection in a single vessel further reduces utility consumption and labor hours, contributing to a leaner and more cost-effective manufacturing operation that aligns with aggressive cost reduction in API manufacturing targets.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of commercially abundant and stable starting materials. Unlike routes that rely on custom-synthesized halides with short shelf-lives, the precursors for this method, such as p-octylbenzaldehyde and triphenylphosphine, are widely available from multiple global vendors. This diversification of the supplier base mitigates the risk of single-source bottlenecks. Moreover, the improved stability of the intermediates allows for more flexible production scheduling and the potential for campaign manufacturing, ensuring that commercial scale-up of complex immunosuppressants can proceed without interruption even during periods of market volatility or raw material scarcity.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route offers a clear path to sustainable scale-up. The reduction in wastewater volume and the absence of heavy metal catalysts simplify the effluent treatment process, making it easier to meet stringent environmental regulations in major pharmaceutical manufacturing hubs. The use of catalytic hydrogenation, a well-established unit operation in the fine chemical industry, ensures that the process can be safely scaled from pilot plant to multi-ton production without encountering the thermal runaway risks associated with bulk chemical reductions. This alignment with green chemistry principles not only future-proofs the manufacturing asset but also enhances the corporate sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this new methodology for industrial production.
Q: How does this new route improve impurity control compared to Friedel-Crafts methods?
A: Traditional Friedel-Crafts acylation often generates regioisomers and requires harsh Lewis acids like aluminum trichloride, leading to difficult purification. This novel Wittig-based route avoids positional isomerism entirely and utilizes a specific acid-base extraction workup that leverages the solubility differences of the amino-functionalized product to achieve purity levels exceeding 99.9%.
Q: What are the safety advantages of using catalytic hydrogenation over chemical reducing agents?
A: Conventional routes often rely on hazardous reagents such as lithium aluminum hydride, triethylsilane, or sodium borohydride-calcium chloride complexes, which pose significant operational risks and generate stoichiometric metal waste. The patented method employs catalytic hydrogenation (Pd/C or Raney Nickel), which is inherently safer, generates water as the primary byproduct, and significantly reduces the environmental footprint of the manufacturing process.
Q: Is the starting material 4-n-octylbenzaldehyde stable for large-scale production?
A: Yes, unlike halide intermediates used in other routes which can be unstable, 4-n-octylbenzaldehyde is a robust and commercially available building block. Its stability ensures consistent reaction kinetics during the Wittig coupling step, facilitating reliable commercial scale-up without the degradation issues associated with reactive alkyl halides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fingolimod Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced therapeutics like Fingolimod depends on a manufacturing partner capable of navigating complex chemical landscapes with precision and reliability. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch meets the highest international pharmacopoeia standards. Our facility is equipped to handle the specific unit operations required by this novel route, including high-pressure hydrogenation and specialized crystallization processes, guaranteeing a consistent supply of high-quality material for your clinical and commercial needs.
We invite you to collaborate with us to optimize your supply chain for Fingolimod Hydrochloride. By leveraging our expertise in process development and scale-up, we can help you realize the full economic and operational potential of this advanced synthetic route. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your strategic sourcing decisions and ensure the uninterrupted availability of this critical immunosuppressant.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
