Advanced Synthesis of 2-Alkynyl Indoles: A Green, Scalable Route for Pharmaceutical Intermediates
Advanced Synthesis of 2-Alkynyl Indoles: A Green, Scalable Route for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective methods for constructing complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN108864164B, which discloses a novel synthesis method for primary amine-guided 2-alkynyl indole compounds. This technology represents a paradigm shift from traditional halogenation-coupling sequences to a direct, transition metal-catalyzed C-H activation strategy. By leveraging a simple primary amine as a directing group, this method achieves high regioselectivity for the challenging C2-position of the indole ring, a feat that is notoriously difficult due to the higher electron density at the C3-position. The process operates under relatively mild conditions, utilizing water as a green solvent, which drastically reduces the environmental footprint and operational hazards associated with volatile organic compounds. For R&D directors and process chemists, this patent offers a robust pathway to access highly functionalized indole derivatives that are critical building blocks for various bioactive molecules and material science applications.
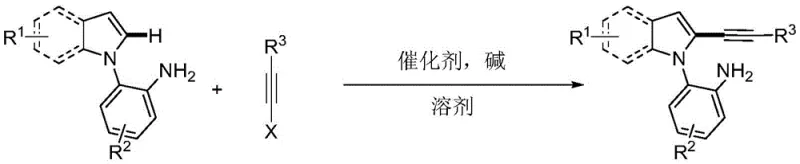
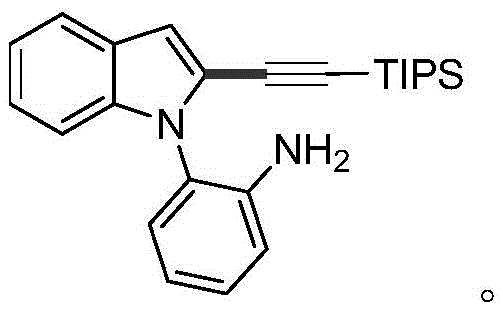
The core innovation lies in the cross-coupling reaction between 2-(1H-indol-1-yl)aniline compounds and alkyne halides. Unlike conventional methods that often require pre-functionalization of the indole core or the use of expensive and toxic reagents, this approach utilizes readily available starting materials. The reaction system employs a palladium salt catalyst, such as palladium chloride or palladium acetate, in conjunction with a cesium-based alkali. The versatility of the substrate scope is impressive, accommodating various substituents on both the indole and aniline rings, including methyl, fluoro, chloro, methoxy, and cyano groups. This broad tolerance ensures that the method can be adapted for the synthesis of a diverse library of analogs, facilitating rapid structure-activity relationship (SAR) studies in drug discovery programs. Furthermore, the use of triisopropylsilyl (TIPS) protected alkynes allows for further downstream functionalization, adding significant value to the synthetic utility of the resulting intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 2-substituted indoles has been plagued by inherent chemoselectivity challenges. The indole nucleus possesses a higher electron cloud density at the C3-position compared to the C2-position, making the C3-site kinetically favored for electrophilic substitution and metallation. Traditional strategies to access 2-alkynyl indoles often involve multi-step sequences, such as initial C-H halogenation followed by Sonogashira coupling. These classical routes are not only atom-uneconomical but also generate substantial amounts of halogenated waste. Moreover, achieving exclusive C2-selectivity often requires bulky protecting groups or harsh reaction conditions that can degrade sensitive functional groups present in complex drug candidates. Other reported methods utilizing transition metal catalysis have struggled with low yields, poor regioselectivity, or the necessity for expensive hypervalent iodine reagents and specialized ligands. These limitations create bottlenecks in the supply chain, increasing the cost of goods sold (COGS) and extending the lead time for producing high-purity pharmaceutical intermediates.
The Novel Approach
The methodology described in CN108864164B elegantly circumvents these obstacles by employing a primary amine-directed C-H activation mechanism. By coordinating the palladium catalyst with the nitrogen atom of the ortho-amino group on the N-phenyl ring, the system forms a thermodynamically stable six-membered palladacycle intermediate. This coordination effectively overrides the natural electronic bias of the indole ring, directing the metal center specifically to the C2-position for activation. The subsequent oxidative addition of the alkyne halide and reductive elimination proceeds smoothly to yield the desired 2-alkynyl product with excellent regioselectivity. Crucially, this reaction can be performed in water or water-toluene mixtures, a feature that is rare for palladium-catalyzed cross-couplings involving organometallic intermediates. This aqueous compatibility simplifies the workup procedure, eliminates the need for extensive solvent exchange, and aligns perfectly with modern green chemistry mandates. The result is a streamlined process that delivers high-purity products with reduced operational complexity.
Mechanistic Insights into Pd-Catalyzed C-H Activation
To fully appreciate the technical sophistication of this synthesis, one must delve into the catalytic cycle that drives the transformation. The reaction initiates with the coordination of the palladium(II) species to the primary amine nitrogen of the 2-(1H-indol-1-yl)aniline substrate. This coordination is the key determinant of regioselectivity, as it positions the metal center in close proximity to the C2-H bond of the indole moiety. Through a concerted metalation-deprotonation (CMD) pathway facilitated by the base (such as cesium pivalate), the C2-H bond is cleaved to form a cyclometallated palladium(II) intermediate. This five- or six-membered ring structure is robust enough to withstand the reaction conditions yet reactive enough to undergo the subsequent transmetallation or oxidative addition steps. The use of cesium salts is particularly beneficial here, as the large cation radius helps to stabilize the anionic intermediates and enhance the solubility of the base in the reaction medium, thereby accelerating the rate-determining step of the catalytic cycle.
Following the formation of the palladacycle, the alkyne halide (e.g., bromoalkyne) undergoes oxidative addition to the palladium center, generating a high-valent palladium(IV) or palladium(II) species depending on the specific mechanistic pathway. This step introduces the alkynyl fragment onto the metal center. Finally, a reductive elimination event occurs, forging the new C-C bond between the indole C2-position and the alkyne carbon, releasing the final 2-alkynyl indole product and regenerating the active palladium catalyst. This mechanism explains the broad substrate tolerance observed in the patent examples; as long as the directing group can coordinate effectively and the steric bulk does not prevent the formation of the metallacycle, the reaction proceeds efficiently. Impurity control is inherently managed by the high selectivity of the directing group, minimizing the formation of C3-alkynylated byproducts or homocoupling side reactions, which simplifies the purification burden and ensures a cleaner impurity profile for regulatory submissions.
How to Synthesize 2-Alkynyl Indole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and purity. The general procedure involves charging a reactor with the 2-(1H-indol-1-yl)aniline substrate, the alkyne halide coupling partner, a palladium catalyst source, a suitable base, and the solvent system. The patent specifies a molar ratio of catalyst to substrate ranging from 0.03:1 to 0.1:1, which is economically viable for large-scale operations. The reaction mixture is then heated to a temperature between 80°C and 110°C, typically maintained for 12 to 24 hours to ensure full conversion. Detailed standardized synthesis steps are provided below to guide process development teams in replicating these results.
- Charge a reactor with 2-(1H-indol-1-yl)aniline substrate, alkyne halide (e.g., TIPS-protected), palladium catalyst, cesium base, and water solvent.
- Stir the reaction mixture at elevated temperatures between 80°C and 110°C for 12 to 24 hours to ensure complete conversion.
- Cool to room temperature, extract with ethyl acetate, dry over magnesium sulfate, and purify via column chromatography to isolate the target 2-alkynyl indole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers compelling advantages for procurement managers and supply chain leaders looking to optimize their sourcing strategies for indole-based intermediates. The primary driver for cost reduction is the simplification of the synthetic sequence. By eliminating the need for pre-halogenation steps and avoiding the use of expensive, specialized ligands or hypervalent iodine oxidants, the overall material cost is significantly lowered. Furthermore, the ability to use water as a solvent translates to drastic savings in solvent purchase, recovery, and disposal costs. Organic solvents like DMF or NMP, often used in traditional couplings, are expensive and subject to strict environmental regulations regarding emissions and waste treatment. Replacing them with water not only reduces the direct financial burden but also mitigates regulatory risks associated with volatile organic compound (VOC) emissions.
- Cost Reduction in Manufacturing: The economic benefits of this process extend beyond simple reagent costs. The high atom economy of the direct C-H activation means that a larger proportion of the starting material mass is incorporated into the final product, reducing waste generation. Additionally, the use of earth-abundant palladium catalysts at relatively low loadings minimizes the cost associated with precious metal recovery. The simplified workup procedure, which involves basic extraction and chromatography, reduces labor hours and energy consumption compared to multi-step purification protocols required for less selective methods. These cumulative efficiencies result in a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and stability of raw materials. This synthesis relies on 2-(1H-indol-1-yl)aniline derivatives and simple alkyne halides, both of which are commercially available from multiple global suppliers. Unlike proprietary reagents that may be sourced from a single vendor, these commodity chemicals ensure a diversified supply base, reducing the risk of shortages or price volatility. The robustness of the reaction conditions (80-110°C) also means that the process is less sensitive to minor fluctuations in temperature or mixing, leading to consistent batch-to-batch quality. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to kilogram or ton scale often reveals hidden pitfalls, but this aqueous-based method is inherently scalable. Water has a high heat capacity, which aids in temperature control during exothermic events, enhancing process safety on a large scale. Moreover, the environmental profile of the process aligns with increasingly stringent global environmental, social, and governance (ESG) standards. By minimizing hazardous waste and utilizing green solvents, manufacturers can easily comply with environmental regulations in jurisdictions with strict pollution controls. This compliance facilitates smoother regulatory approvals and enhances the corporate reputation of the supply chain partners, making it a preferred choice for multinational corporations with strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this primary amine-guided alkynylation technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating this synthetic route for their specific applications.
Q: What is the key advantage of using water as a solvent in this indole alkynylation?
A: Using water as the primary solvent significantly reduces environmental impact and waste disposal costs compared to traditional organic solvents, aligning with green chemistry principles while maintaining high reaction efficiency.
Q: How does the primary amine directing group improve regioselectivity?
A: The primary amine group coordinates with the palladium catalyst to form a stable six-membered ring intermediate, effectively guiding the activation to the C2 position of the indole ring rather than the inherently more reactive C3 position.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process utilizes inexpensive, commercially available starting materials and mild conditions (80-110°C), making it highly adaptable for commercial scale-up with robust safety profiles and simplified downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkynyl Indole Supplier
The technological potential of primary amine-guided C-H activation is immense, offering a pathway to complex heterocycles that were previously difficult to access economically. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of 2-alkynyl indole intermediates we produce. We understand that consistency and quality are non-negotiable in the pharmaceutical supply chain, and our dedicated process chemistry team is ready to optimize this route further to meet your specific throughput and purity requirements.
We invite you to collaborate with us to leverage this green synthesis technology for your next project. Whether you require custom synthesis of specific analogs or large-scale manufacturing of the core scaffold, our technical procurement team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us today to request specific COA data, route feasibility assessments, and quotations. Let us help you secure a reliable, cost-effective, and sustainable supply of high-quality 2-alkynyl indole compounds for your global operations.
