Advanced Synthesis of 2-Alkynyl Indoles via Primary Amine Guidance for Pharma
Advanced Synthesis of 2-Alkynyl Indoles via Primary Amine Guidance for Pharma
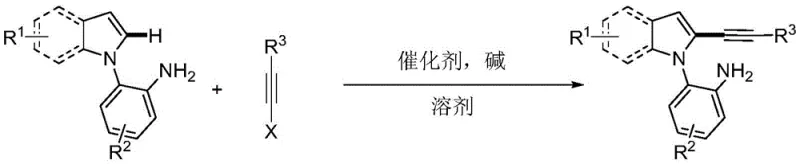
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for more efficient and selective synthetic routes for pharmaceutical intermediates. A significant breakthrough in this domain is documented in patent CN108864164B, which discloses a novel synthesis method for primary amine-guided 2-alkynyl indole compounds. This technology represents a paradigm shift from traditional multi-step functionalization strategies to a streamlined, direct C-H activation approach. By leveraging the coordinating ability of a primary amine group, this method achieves exceptional regioselectivity at the C2 position of the indole ring, a site that is historically challenging to functionalize due to the inherent electronic preference for the C3 position. For R&D directors and process chemists, this innovation offers a powerful tool for constructing highly functionalized indole scaffolds, which are ubiquitous in bioactive molecules and drug candidates. The methodology not only simplifies the synthetic pathway but also adheres to rigorous green chemistry standards by employing water as the reaction solvent, thereby addressing both economic and environmental concerns in modern chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 2-alkynyl indole derivatives has been fraught with synthetic challenges that hinder efficient commercial production. The classical approach typically involves a tedious two-step sequence: initial C-H halogenation at the 2-position followed by a transition metal-catalyzed cross-coupling reaction. This traditional route suffers from poor atom economy, generates significant halogenated waste, and often requires harsh reaction conditions that are incompatible with sensitive functional groups. Furthermore, direct C-H activation strategies have struggled with regioselectivity; the electron-rich C3 position of the indole ring is kinetically favored for metallization, leading to mixtures of 2- and 3-substituted isomers that are difficult and costly to separate. Previous attempts to force C2 selectivity often relied on the use of bulky substrates, expensive hypervalent iodine reagents, or complex protecting group strategies, all of which escalate the cost of goods and complicate the supply chain for high-purity intermediates.
The Novel Approach
The methodology outlined in the patent data introduces a transformative solution by utilizing a primary amine as an intramolecular directing group to guide the palladium catalyst specifically to the C2 position. This strategy bypasses the need for pre-halogenation and effectively overrides the natural electronic bias of the indole system. The reaction proceeds under mild thermal conditions (80-110°C) using readily available palladium salts and cesium bases. A standout feature of this novel approach is the use of water as the primary solvent, which drastically reduces the reliance on toxic organic volatiles and simplifies downstream processing. The broad substrate scope allows for the introduction of diverse substituents, including halogens, alkyl groups, and electron-withdrawing cyano groups, without compromising yield or selectivity. This robustness makes the process ideal for the rapid generation of compound libraries and the scalable manufacture of complex drug precursors.
Mechanistic Insights into Pd-Catalyzed C-H Alkynylation
The success of this synthesis lies in the intricate coordination chemistry between the palladium catalyst and the substrate. The reaction mechanism initiates with the coordination of the palladium species to the nitrogen atom of the primary amine directing group. This coordination facilitates the formation of a stable six-membered palladacycle intermediate, effectively positioning the metal center in close proximity to the C2-H bond of the indole ring. This geometric constraint is the key to achieving high regioselectivity, as it lowers the activation energy for C2-H cleavage relative to the C3 position. Following cyclometallation, the alkyne halide undergoes oxidative addition to the palladium center. The subsequent reductive elimination step releases the desired 2-alkynyl indole product and regenerates the active catalyst species. This catalytic cycle is highly efficient, operating with low catalyst loading (0.03-0.1 molar ratio), which is critical for minimizing residual metal impurities in the final pharmaceutical intermediate.
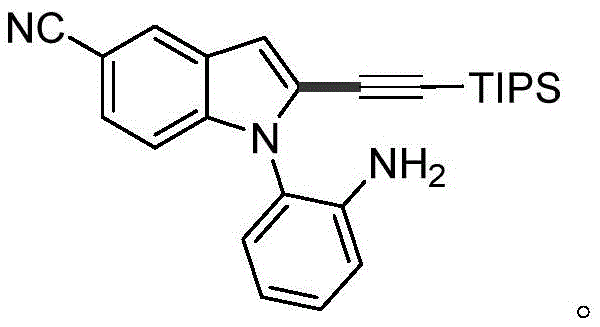
From an impurity control perspective, the mechanism inherently suppresses the formation of C3-alkynylated byproducts, which are common contaminants in non-directed reactions. The use of a bulky triisopropylsilyl (TIPS) group on the alkyne further enhances selectivity by sterically hindering attack at the more congested sites. The process demonstrates remarkable tolerance to various electronic environments on the indole backbone. For instance, electron-deficient substrates bearing cyano or fluoro substituents react smoothly, as do electron-rich variants with methoxy or methyl groups. This versatility ensures that the synthetic route remains viable even when modifying the core structure to optimize biological activity. The ability to synthesize complex derivatives, such as the cyano-substituted indole shown below, highlights the method's utility in late-stage functionalization where molecular complexity is already high.
How to Synthesize 2-Alkynyl Indole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise control over reaction parameters to maximize yield and purity. The protocol involves charging a reactor with the 2-(1H-indol-1-yl)aniline substrate, the alkyne halide coupling partner, a palladium salt catalyst such as palladium tetranitrile tetrafluoroborate, and a cesium base like cesium pivalate. Water is added as the solvent, and the mixture is heated to between 80°C and 110°C with vigorous stirring. The reaction typically proceeds to completion within 12 to 24 hours. Upon cooling, the product is isolated via liquid-liquid extraction with ethyl acetate, dried over anhydrous magnesium sulfate, and purified using standard column chromatography techniques. Detailed standardized operating procedures for scaling this reaction are provided in the technical guide below.
- Charge the reactor with 2-(1H-indol-1-yl)aniline substrate, alkyne halide, palladium salt catalyst, cesium base, and water solvent.
- Heat the reaction mixture to 80-110°C and stir vigorously for 12-24 hours to ensure complete conversion.
- Cool to room temperature, extract with ethyl acetate, dry over magnesium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers tangible strategic benefits beyond mere technical novelty. The shift towards a water-based solvent system fundamentally alters the cost structure of the manufacturing process by eliminating the need for expensive, anhydrous organic solvents and the associated recovery infrastructure. This simplification of the solvent system directly translates to reduced operational expenditures and a smaller environmental footprint, which is increasingly a prerequisite for vendor qualification by major pharmaceutical companies. Furthermore, the reliance on commodity chemicals for the catalyst and base ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or proprietary reagents that may face availability bottlenecks.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its high atom economy and the use of inexpensive, earth-abundant reagents. By avoiding multi-step sequences involving pre-functionalization, the overall material throughput is significantly improved, leading to substantial cost savings per kilogram of product. The elimination of expensive ligands and the ability to use low loadings of palladium catalyst further reduce the raw material costs. Additionally, the simplified workup procedure reduces labor hours and energy consumption associated with solvent distillation and waste treatment, driving down the total cost of ownership for the intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream API manufacturers. The starting materials, such as o-iodoaniline and indole derivatives, are widely available from multiple global suppliers, reducing dependency on single-source vendors. This diversification of the supply base enhances resilience against market fluctuations and geopolitical disruptions. The mild reaction conditions also reduce equipment wear and tear, extending the lifecycle of manufacturing assets and ensuring long-term production capacity stability.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is straightforward due to the absence of hazardous exotherms and the use of non-flammable water as the solvent. This safety profile simplifies the regulatory approval process for new manufacturing facilities and reduces insurance premiums. The green nature of the process aligns with stringent environmental regulations regarding VOC emissions and hazardous waste disposal. By adopting this technology, manufacturers can future-proof their operations against tightening environmental laws, ensuring continuous compliance and avoiding potential fines or shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this primary amine-guided synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: Why is regioselectivity critical in indole alkynylation?
A: Indole naturally favors electrophilic substitution at the C3 position due to higher electron cloud density. Achieving C2 selectivity traditionally requires harsh pre-functionalization or bulky directing groups. This novel primary amine-guided method overcomes this intrinsic bias, enabling direct access to the valuable 2-position without protecting groups.
Q: What are the environmental advantages of this synthesis route?
A: Unlike traditional methods relying on volatile organic solvents, this process utilizes water as the primary reaction medium. This significantly reduces VOC emissions, simplifies waste treatment protocols, and aligns with modern green chemistry principles required by global regulatory bodies.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the process employs commercially available palladium salts and simple inorganic bases like cesium pivalate. The operational simplicity, mild temperature range (80-110°C), and robust substrate tolerance make it highly scalable for industrial manufacturing of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkynyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert process chemists has extensively evaluated the primary amine-guided alkylation route and confirmed its potential for delivering high-purity 2-alkynyl indoles at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from benchtop to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner committed to innovation, quality, and sustainable growth in the fine chemical sector.
