Revolutionizing 2-Aminothiazole Production: A Green Aqueous Copper-Catalyzed Approach for Commercial Scale-Up
Revolutionizing 2-Aminothiazole Production: A Green Aqueous Copper-Catalyzed Approach for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for key heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN114436991A, which discloses a novel synthetic method for 2-aminothiazole compounds. This technology represents a paradigm shift from traditional halogenation strategies to a direct, copper-catalyzed amination process conducted in an aqueous medium. For R&D directors and procurement managers alike, this innovation offers a compelling value proposition: it simplifies the supply chain for critical pharmaceutical intermediates by eliminating complex purification steps and hazardous organic solvents. The core of this invention lies in the efficient substitution of the bromine atom at the 2-position of the thiazole ring using ammonia water, facilitated by a monovalent copper catalyst under controlled thermal and atmospheric conditions.
This method addresses long-standing challenges in the synthesis of 2-aminothiazoles, which are pivotal building blocks for antiviral, anticonvulsant, and antidiabetic agents, as well as advanced OLED materials. By leveraging a green chemistry approach, the process achieves target yields ranging from 61.9% to 80% with purities consistently exceeding 95%. The strategic use of water as a solvent not only aligns with modern sustainability goals but also exploits the differential solubility of reactants and products to facilitate isolation. As a reliable pharmaceutical intermediate supplier, understanding such proprietary advancements is crucial for securing a competitive edge in cost reduction in API manufacturing. The following analysis dissects the technical merits and commercial implications of this patented methodology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of substituted 2-aminothiazoles often relied on the direct halogenation of the parent 2-aminothiazole nucleus. As illustrated in the comparative data within the patent, traditional routes involving reagents like N-bromosuccinimide (NBS) in organic solvents such as dichloromethane suffer from severe limitations. A primary drawback is the poor solubility of 2-aminothiazole in common organic reaction media, which creates heterogeneous reaction conditions that drastically inhibit reaction kinetics. In Comparative Example 1 of the patent, attempting to brominate 2-aminothiazole with NBS resulted in a complete failure to generate the target product, yielding 0% conversion due to these solubility constraints. Furthermore, electrophilic substitution on the thiazole ring often lacks regioselectivity, leading to mixtures of 4-bromo and 5-bromo isomers that are notoriously difficult to separate. This complexity necessitates extensive chromatographic purification, driving up production costs and generating significant chemical waste, which is untenable for commercial scale-up of complex polymer additives or drug substances.

The Novel Approach
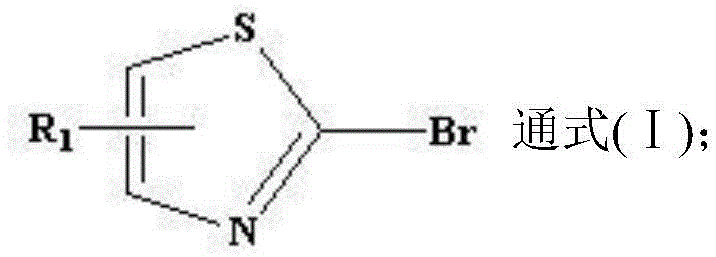
In stark contrast, the novel approach described in CN114436991A inverts the synthetic logic by starting with readily available 2-bromo-thiazole derivatives and performing a nucleophilic substitution with ammonia. This strategy bypasses the regioselectivity issues inherent in electrophilic aromatic substitution. The reaction utilizes a general formula (I) where the leaving group is a bromine atom at the 2-position, and the substituent R1 can be varied to include methyl, tert-butyl, bromo, or 4-fluorophenyl groups. By employing water as the reaction medium, the process capitalizes on the fact that the lipophilic 2-bromo-thiazole starting materials are largely insoluble, while the resulting 2-aminothiazole products exhibit enhanced polarity and water solubility due to the introduction of the amino group. This physical property difference is ingeniously exploited during workup; unreacted starting material can be removed via simple filtration, while the product remains in the aqueous phase for subsequent extraction. This streamlined workflow significantly reduces the operational burden associated with reducing lead time for high-purity pharmaceutical intermediates.
Mechanistic Insights into Cu(I)-Catalyzed Amination
The success of this transformation hinges on the precise orchestration of a copper-catalyzed cycle. The patent specifies the use of monovalent copper salts, with cuprous bromide (CuBr) demonstrating optimal catalytic activity compared to cuprous oxide or cuprous iodide. The mechanism likely involves the coordination of the ammonia nucleophile to the copper center, followed by oxidative addition into the carbon-bromine bond of the thiazole substrate. The presence of ammonia water serves a dual function: it acts as the source of the amino group and simultaneously buffers the reaction system to maintain a pH between 8 and 9. This alkaline environment is essential for promoting the deprotonation steps required for the catalytic turnover and for neutralizing the hydrobromic acid byproduct formed during the substitution. The reaction proceeds efficiently at temperatures between 100°C and 110°C, providing sufficient thermal energy to overcome the activation barrier for the C-N bond formation without degrading the sensitive heterocyclic core.
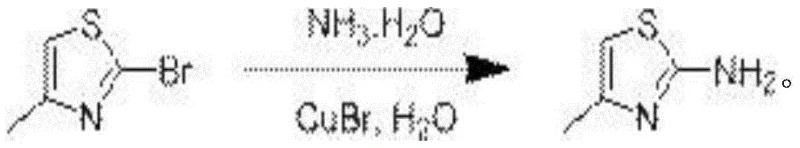
Crucially, the reaction must be conducted under a nitrogen atmosphere to ensure the integrity of the catalytic cycle. Oxygen acts as a potent inhibitor in this system; its presence interferes with the redox cycling of the copper catalyst and promotes undesirable debromination side reactions, leading to the formation of reduced thiazole byproducts. Experimental data confirms that omitting nitrogen protection causes a precipitous drop in both yield and purity. Furthermore, the choice of copper oxidation state is non-negotiable; divalent copper salts (Cu(II)) are ineffective because they cannot participate in the requisite oxidative addition/reductive elimination sequence in the same manner as Cu(I). This mechanistic understanding underscores the importance of strict process control parameters for any entity aiming to replicate this high-purity OLED material synthesis pathway.
How to Synthesize 2-Aminothiazole Derivatives Efficiently
Implementing this synthesis requires adherence to specific stoichiometric and operational parameters to maximize efficiency. The patent outlines a robust protocol where the molar ratio of the 2-bromo-thiazole substrate to ammonia water to cuprous bromide catalyst to water is optimized at approximately 1 mol : 10-15 ml : 0.10-0.20 mol : 10-15 ml. Deviating from this catalyst loading range results in suboptimal conversion; insufficient catalyst leads to incomplete reaction, while excess catalyst provides no additional benefit and complicates downstream metal removal. The reaction mixture is stirred vigorously at 500 r/min to ensure adequate mass transfer between the solid/liquid phases. Upon completion, typically monitored by TLC or LCMS after 12-15 hours, the workup procedure is designed for simplicity and scalability.
- Prepare the reaction mixture by combining 2-bromo-thiazole derivative, ammonia water, cuprous bromide catalyst, and deionized water under nitrogen protection.
- Heat the mixture to 100-110°C with stirring at 500 r/min for 12-15 hours while maintaining pH 8-9.
- Filter the cooled reaction liquid to remove solids, extract the filtrate with ethyl acetate, wash with brine, and concentrate to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous copper-catalyzed method offers tangible strategic benefits beyond mere technical feasibility. The most immediate impact is seen in the drastic simplification of the supply chain for raw materials. By replacing expensive, hazardous organic solvents like dichloromethane with commodity-grade deionized water, the process eliminates the volatility risks associated with solvent pricing and availability. Moreover, the safety profile of the operation is significantly enhanced, reducing the regulatory burden and insurance costs associated with handling large volumes of volatile organic compounds (VOCs). This shift aligns perfectly with global trends towards greener manufacturing, potentially qualifying the production facility for various environmental incentives and easing compliance with increasingly stringent waste disposal regulations.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the elimination of costly purification steps. In traditional methods, separating isomeric byproducts often requires preparative HPLC or multiple recrystallizations, which are resource-intensive. In this novel method, the insolubility of the starting material allows for its removal via simple mechanical filtration, a unit operation that is exponentially cheaper and faster than chromatography. Additionally, the high selectivity of the copper catalyst minimizes the formation of side products, meaning less material is lost to waste streams. The use of water as a solvent further reduces the cost of goods sold (COGS) by removing the need for solvent recovery systems or expensive solvent purchase contracts. These factors combine to deliver substantial cost savings without compromising on the quality of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for continuous API production. This synthesis route enhances reliability by utilizing widely available, commodity-grade reagents. Ammonia water, cuprous bromide, and water are standard industrial chemicals with stable supply chains, unlike specialized halogenating agents or exotic ligands that might be subject to geopolitical supply disruptions. The robustness of the reaction conditions—tolerating a range of substrates including those with bulky tert-butyl groups or electron-withdrawing fluorophenyl moieties—means that a single production line can be adapted to manufacture a diverse portfolio of thiazole derivatives. This flexibility allows suppliers to respond rapidly to changing market demands, ensuring consistent delivery schedules for their clients.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen thermal and mixing challenges. However, the use of water as a solvent provides excellent heat capacity, facilitating temperature control in large-scale reactors and mitigating the risk of thermal runaways. The simplified workup, involving filtration and liquid-liquid extraction, translates seamlessly to industrial equipment such as filter presses and centrifuges. From an environmental perspective, the aqueous waste stream is easier to treat than organic solvent waste, reducing the load on effluent treatment plants. The high atom economy of the substitution reaction, coupled with the ability to recover and recycle unreacted starting material from the filter cake, ensures that the process meets rigorous environmental compliance standards, making it a sustainable choice for long-term agrochemical intermediate or pharmaceutical production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on critical process parameters. Understanding these nuances is essential for process chemists tasked with technology transfer and scale-up activities.
Q: Why is cuprous bromide preferred over other copper salts for this synthesis?
A: Experimental data indicates that cuprous bromide (CuBr) exhibits superior catalytic activity compared to cuprous oxide or cuprous iodide. Furthermore, divalent copper salts like copper bromide (CuBr2) are ineffective because they cannot undergo the necessary oxidation state changes required for the catalytic cycle in this oxidative amination reaction.
Q: What are the advantages of using water as a solvent instead of organic solvents?
A: Using water significantly reduces raw material costs and environmental impact. Additionally, the starting 2-bromo-thiazole materials are generally insoluble in water, while the amino-product is soluble. This polarity difference allows for easy separation of unreacted starting materials via simple filtration, streamlining the purification process.
Q: How does the nitrogen atmosphere affect the reaction outcome?
A: The nitrogen atmosphere is critical for excluding oxygen. Oxygen interferes with the copper catalytic cycle and promotes debromination side reactions. Maintaining an inert environment ensures high selectivity for the amination product and prevents the formation of debrominated by-products, thereby securing yields above 60%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminothiazole Supplier
The technological advancements detailed in patent CN114436991A underscore the potential for high-efficiency manufacturing of 2-aminothiazole derivatives. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage such innovative pathways for our clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are translated into reliable industrial reality. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets stringent purity specifications, consistently delivering materials with purity levels exceeding 95% as demonstrated in the patent examples. Our commitment to quality assurance ensures that the impurity profiles of our intermediates are fully characterized and controlled.
We invite potential partners to engage with our technical team to explore how this green synthesis route can be integrated into your supply chain. By collaborating with us, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a sustainable and cost-effective supply of high-quality 2-aminothiazole intermediates for your next generation of therapeutic or material science applications.
