Scalable Synthesis of 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane-2-Carboxylic Acid for Antiviral APIs
Introduction to Advanced Antiviral Intermediate Synthesis
The global pharmaceutical landscape has witnessed an unprecedented demand for potent antiviral agents, particularly those targeting the 3CL protease of coronaviruses such as SARS-CoV-2. At the heart of these therapeutic candidates lies a critical structural motif: the 6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid scaffold. This chiral bicyclic amino acid serves as the foundational core for next-generation covalent inhibitors, including derivatives of Rupintrivir and PF-00835231. Recognizing the strategic importance of this building block, recent intellectual property developments, specifically patent CN114394927A, have unveiled a groundbreaking manufacturing methodology. This new technical disclosure addresses long-standing bottlenecks in the production of this high-value pharmaceutical intermediate by introducing a safer, more efficient, and environmentally benign synthetic pathway. For R&D directors and supply chain strategists, understanding the nuances of this patented process is essential for securing a reliable supply of high-purity antiviral intermediates.
The significance of this innovation extends beyond mere academic interest; it represents a pivotal shift in process chemistry that directly impacts commercial viability. Traditional methods for constructing this complex bicyclic system have been plagued by severe safety hazards, toxic reagent usage, and difficult purification challenges. The new approach detailed in CN114394927A leverages modern catalytic strategies, including enzymatic resolution and transition metal catalysis, to streamline the synthesis. By integrating these advanced techniques, the process not only enhances the stereochemical integrity of the final product but also aligns with the rigorous environmental and safety standards demanded by modern regulatory bodies. This report provides a deep technical and commercial analysis of this novel route, offering actionable insights for procurement and technical teams aiming to optimize their antiviral API supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advent of the technology described in CN114394927A, the industrial synthesis of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid relied heavily on legacy routes that presented substantial operational risks. The most prominent conventional pathway initiates from caronic anhydride, a starting material that necessitates a series of perilous transformations. The initial step involves a high-temperature dehydration reaction with urea, typically conducted at temperatures around 170°C. This thermal process is inherently unstable, generating significant volumes of gas that can lead to the dangerous clogging of reflux condensers, posing a direct threat to reactor integrity and operator safety. Furthermore, subsequent steps in this traditional route require the use of lithium aluminum hydride (LiAlH4) for reduction purposes.
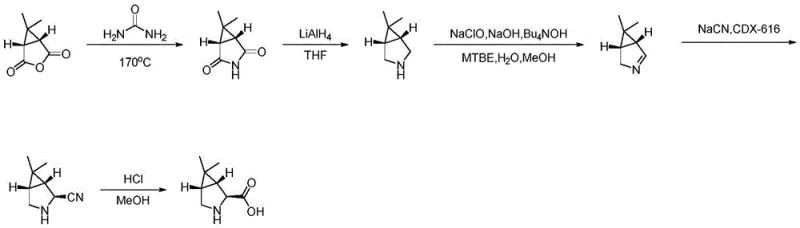
The utilization of LiAlH4 introduces severe fire hazards due to its pyrophoric nature and the potential for exothermic runaway reactions if heat accumulation is not meticulously managed. Perhaps most critically, the construction of the nitrile functionality in the old route depends on the addition of sodium cyanide (NaCN). The handling of cyanide salts on a commercial scale requires extensive safety infrastructure, specialized waste treatment protocols, and rigorous personnel training, all of which drive up operational costs and complicate regulatory compliance. These cumulative risks make the conventional caronic anhydride route increasingly untenable for modern, sustainable pharmaceutical manufacturing, creating an urgent need for a safer alternative.
The Novel Approach
In stark contrast to the hazardous legacy methods, the novel process disclosed in patent CN114394927A offers a streamlined and significantly safer synthetic trajectory. This innovative route begins with N-Boc-3-pyrroline, a readily available and stable starting material, thereby bypassing the dangerous high-temperature urea condensation entirely. The synthesis proceeds through a logical sequence of five key steps that prioritize safety and selectivity. Instead of using pyrophoric hydrides, the new method employs mild esterification conditions using lithium diisopropylamide (LDA) and dimethyl carbonate. Crucially, the introduction of chirality is achieved through biocatalysis rather than harsh chemical resolution.
The use of Novozym435, an immobilized lipase, allows for highly selective enzymatic hydrolysis under mild aqueous conditions, effectively resolving the racemic mixture to yield the desired chiral acid intermediate with high purity. Furthermore, the construction of the bicyclic core is accomplished via a sophisticated cobalt-catalyzed cyclopropanation using 2,2-dichloropropane and zinc reagents. This transformation elegantly installs the gem-dimethyl cyclopropane ring without the need for toxic cyanide sources. By eliminating the most dangerous reagents from the workflow—specifically LiAlH4 and NaCN—this novel approach drastically reduces the safety burden on manufacturing facilities while improving the overall efficiency and environmental profile of the production process.
Mechanistic Insights into Cobalt-Catalyzed Cyclopropanation and Enzymatic Resolution
The technical brilliance of the new synthesis lies in its strategic application of catalytic mechanisms to solve complex structural problems. A focal point of this chemistry is the cobalt-catalyzed cyclopropanation step (Step S4), which constructs the strained 3-azabicyclo[3.1.0]hexane core. In this transformation, the methyl ester intermediate is treated with a specialized cobalt catalyst, specifically [2-t-BuPDI]CoBr2, in the presence of zinc powder and zinc bromide. The 2,2-dichloropropane serves as the carbene precursor, which, upon reductive activation by the zinc/cobalt system, generates a reactive metal-carbenoid species. This species undergoes a concerted addition across the double bond of the pyrroline ring, simultaneously forming the two new carbon-carbon bonds required for the cyclopropane fusion. This mechanism is superior to traditional diazo-based cyclopropanations as it avoids the generation of explosive diazo compounds and utilizes stable, liquid chloroalkanes instead.
Equally critical to the success of this route is the enzymatic resolution step (Step S2), which dictates the stereochemical outcome of the synthesis. The substrate, a racemic methyl ester, is subjected to hydrolysis by Novozym435 in the presence of sodium bicarbonate. The enzyme exhibits exquisite stereoselectivity, preferentially hydrolyzing one enantiomer of the ester to the corresponding carboxylic acid while leaving the other enantiomer untouched or reacting at a negligible rate. This kinetic resolution allows for the isolation of the chiral acid intermediate with exceptional optical purity, reported to reach 96.5% in experimental examples. The ability to achieve such high stereocontrol under mild, near-neutral pH conditions (using bicarbonate buffer) preserves the integrity of sensitive functional groups and prevents racemization, a common pitfall in chemical resolution methods. This biocatalytic precision ensures that the downstream cyclopropanation proceeds with a homochiral substrate, ultimately delivering the final API intermediate with the rigorous enantiomeric excess required for antiviral efficacy.
How to Synthesize 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane-2-Carboxylic Acid Efficiently
The implementation of this novel synthetic route requires careful attention to reaction parameters to maximize yield and purity. The process is divided into distinct operational stages, beginning with the low-temperature lithiation and esterification of the pyrroline starting material, followed by the critical enzymatic workup. The subsequent methylation and cobalt-catalyzed ring closure demand anhydrous conditions and inert atmosphere handling to ensure catalyst longevity. Finally, the global deprotection and hydrolysis step utilizes aqueous acid to reveal the free amino acid. For process chemists looking to adopt this methodology, adherence to the specific stoichiometry and temperature profiles outlined in the patent is essential to replicate the high yields and purity levels demonstrated in the examples. The detailed standardized synthesis steps are provided below to facilitate immediate technology transfer and pilot scale evaluation.
- Perform substitution reaction of N-Boc-3-pyrroline with dimethyl carbonate using LDA base to form the methyl ester intermediate.
- Conduct enzymatic hydrolysis using Novozym435 and sodium bicarbonate to resolve the chiral center and obtain the carboxylic acid.
- Execute cobalt-catalyzed cyclopropanation using 2,2-dichloropropane and zinc reagents to construct the bicyclic core, followed by acid deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in CN114394927A translates into tangible strategic advantages that extend far beyond the laboratory bench. The primary value proposition lies in the drastic simplification of the safety infrastructure required for production. By completely eliminating the need for sodium cyanide and lithium aluminum hydride, manufacturers can avoid the substantial capital expenditures associated with specialized containment systems, cyanide detoxification units, and pyrophoric handling equipment. This reduction in hazard profile directly correlates to lower operational overheads and reduced insurance liabilities, making the cost of goods sold (COGS) for this intermediate significantly more competitive compared to material produced via legacy routes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of expensive and hazardous reagents with more economical and manageable alternatives. The removal of NaCN eliminates the costly waste treatment processes mandated for cyanide-containing effluents, while the replacement of LiAlH4 with milder reducing systems reduces the risk of batch loss due to thermal runaways. Furthermore, the use of enzymatic resolution, while requiring specific biocatalysts, often results in higher effective yields of the desired enantiomer compared to classical crystallization-based resolutions, thereby improving overall material throughput and reducing raw material consumption per kilogram of final product.
- Enhanced Supply Chain Reliability: Supply continuity is a critical concern for pharmaceutical buyers, and this new route offers superior robustness. The starting material, N-Boc-3-pyrroline, is a commodity chemical with a stable supply chain, unlike some specialized anhydrides used in older methods. Additionally, the avoidance of high-risk steps means that production schedules are less likely to be disrupted by safety incidents or regulatory inspections triggered by the storage of extreme hazards. The modular nature of the synthesis, with distinct isolation points for intermediates, allows for flexible inventory management, ensuring that key precursors can be stocked to buffer against demand fluctuations for the final antiviral API.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety risks, but this methodology is inherently designed for scale-up. The exothermic profiles of the reactions are manageable, and the absence of gas-generating high-temperature steps prevents the engineering bottlenecks associated with condenser clogging. From an environmental perspective, the process aligns with Green Chemistry principles by utilizing biocatalysis and reducing toxic waste generation. This compliance facilitates smoother regulatory approvals in stringent markets such as the US and EU, where environmental, social, and governance (ESG) criteria are increasingly influencing supplier selection and procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid. These answers are derived directly from the technical specifications and advantageous effects disclosed in the patent literature, providing clarity on the feasibility and benefits of this advanced manufacturing route. Understanding these details is crucial for stakeholders evaluating the integration of this intermediate into their broader antiviral drug development pipelines.
Q: How does the new process improve safety compared to traditional routes?
A: The novel route eliminates the use of highly toxic sodium cyanide (NaCN) and pyrophoric lithium aluminum hydride (LiAlH4). It also avoids high-temperature urea dehydration steps that generate excessive gas and pose clogging risks, significantly enhancing operational safety.
Q: What is the role of Novozym435 in this synthesis?
A: Novozym435 is utilized for kinetic resolution during the hydrolysis step. This biocatalytic approach ensures high stereochemical purity (up to 96.5% purity reported) of the chiral intermediate under mild aqueous conditions, replacing harsher chemical resolution methods.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. By removing hazardous reagents like NaCN and LiAlH4 and utilizing robust cobalt catalysis for ring construction, the method reduces the need for specialized containment infrastructure, facilitating easier scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane-2-Carboxylic Acid Supplier
As the demand for antiviral therapeutics continues to evolve, securing a supply of high-quality intermediates is paramount for drug developers. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in process chemistry to deliver complex pharmaceutical building blocks. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our partners receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid meets the exacting standards required for clinical and commercial API synthesis.
We invite procurement leaders and R&D teams to collaborate with us to optimize their supply chains. By leveraging the advanced synthetic routes discussed in this report, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to accelerate the development of life-saving antiviral medications through reliable, safe, and cost-effective chemical manufacturing solutions.
