Advanced Synthesis of Boceprevir Intermediates: A Cost-Effective Route for Commercial API Manufacturing
The global demand for direct-acting antivirals (DAAs) targeting Hepatitis C Virus (HCV) has necessitated the development of robust, scalable synthetic routes for key active pharmaceutical ingredients (APIs) and their precursors. Patent CN103435532A, filed in late 2013, introduces a groundbreaking synthetic methodology for producing the critical Boceprevir intermediate, specifically (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid methyl ester hydrochloride. This molecule serves as the chiral core for Boceprevir, a protease inhibitor that revolutionized HCV genotype 1 treatment upon its FDA approval. The structural complexity of this bicyclic system requires precise stereochemical control to ensure therapeutic efficacy. 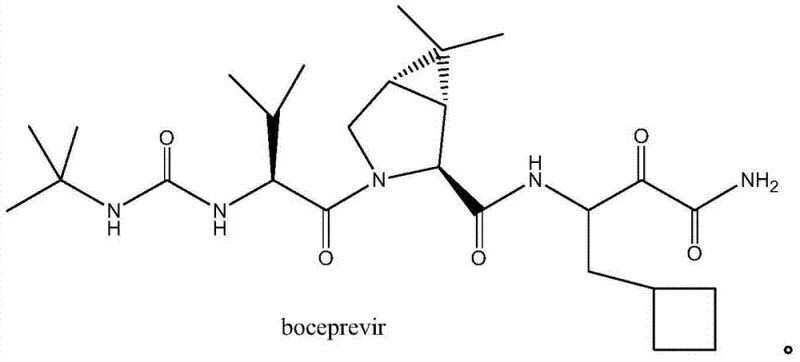
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the cumbersome and hazardous steps associated with earlier generations of synthesis. By leveraging a novel chiral induction strategy involving 4,4'-difluorobenzophenone, the inventors have established a pathway that not only enhances optical purity but also drastically simplifies the operational workflow. This report analyzes the technical merits of this approach, highlighting its potential to serve as a reliable pharmaceutical intermediate supplier standard for cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Boceprevir intermediates was plagued by significant technical and economic inefficiencies, as documented in international patent applications such as WO2004/113295 and WO2007/075790. These legacy routes heavily relied on the use of expensive transition metal catalysts, specifically palladium on carbon (Pd-C), which not only inflated raw material costs but also introduced stringent requirements for heavy metal removal to meet regulatory limits. Furthermore, these methods often utilized hazardous reducing agents like lithium aluminum hydride (LiAlH4), posing severe safety risks during scale-up due to their pyrophoric nature and sensitivity to moisture. 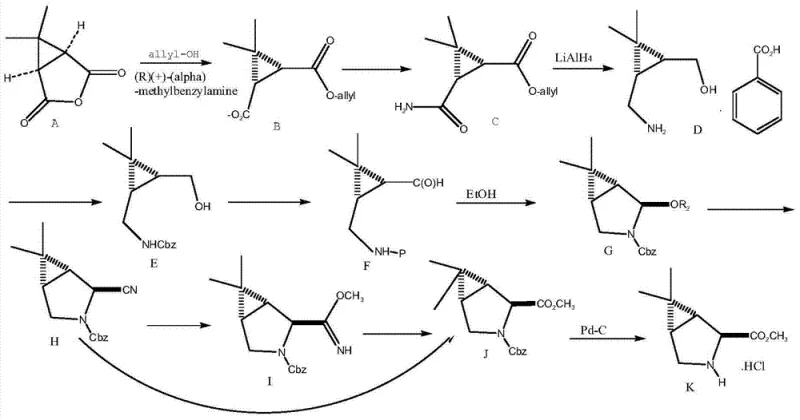
Beyond safety and cost, the operational complexity of these conventional methods was a major bottleneck for supply chain continuity. For instance, certain prior art processes required extended drying periods in ovens lasting up to three days for a single step, severely limiting throughput and increasing energy consumption. Additionally, the multi-step sequences often generated by-products with different chirality, necessitating complex purification protocols that eroded overall yield. The cumulative effect of these factors was a fragile supply chain vulnerable to delays and quality deviations, making the consistent production of high-purity intermediates a persistent challenge for manufacturers.
The Novel Approach
The methodology disclosed in CN103435532A represents a paradigm shift by eliminating the dependency on precious metal catalysts and hazardous hydrides. Instead, the process utilizes readily available, cost-effective reagents such as s-butyllithium and 4,4'-difluorobenzophenone to drive the key transformations. This strategic substitution removes the need for expensive catalyst recovery systems and mitigates the risks associated with handling pyrophoric materials, thereby enhancing the inherent safety profile of the manufacturing plant. The reaction conditions are notably milder, with key steps proceeding efficiently at temperatures between 30°C and 35°C, contrasting sharply with the extreme conditions often required in older protocols.
Moreover, the novel approach streamlines the synthetic sequence, reducing the total number of unit operations and significantly shortening reaction times. The critical resolution step, which establishes the vital stereochemistry, is completed within 3 to 4 hours, a fraction of the time required by competing technologies. This acceleration in kinetics directly translates to improved equipment utilization rates and faster batch turnover. By simplifying the workup procedures—avoiding complex chromatographic separations in favor of crystallization and extraction—the new method ensures a more robust and reproducible process, ideal for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Chiral Induction and Resolution
At the heart of this synthetic breakthrough is a sophisticated chiral induction mechanism that leverages the electronic properties of 4,4'-difluorobenzophenone. The process begins with the protection of the amino group on the starting material, 6,6-dimethyl-3-azabicyclo[3.1.0]hexane hydrochloride, typically using a carbobenzoxy (Cbz) or tert-butoxycarbonyl (Boc) group. This protection is crucial as it prevents unwanted oxidation and side reactions during the subsequent lithiation steps. The protected intermediate is then subjected to deprotonation using a strong base, such as s-butyllithium, at cryogenic temperatures ranging from -85°C to -70°C. 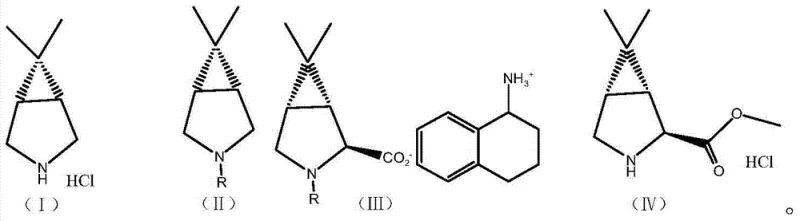
The addition of 4,4'-difluorobenzophenone acts as a chiral inductive agent, guiding the formation of the carbanion intermediate with high stereoselectivity. The patent emphasizes that lower acidity and stronger hydrogen abstraction capabilities favor the formation of the desired carbanion species. Following the generation of the carbanion, the reaction mixture is carefully quenched and the pH is adjusted to a narrow window of 2.5 to 4.0 using a buffered solution. This precise pH control is critical; it ensures that the system remains in a state conducive to the subsequent resolution step without promoting racemization or degradation of the sensitive bicyclic core.
The resolution is achieved through the introduction of 1,2,3,4-tetrahydro-1-naphthylamine, which acts as a chiral resolving agent. This amine selectively interacts with the desired stereoisomer (Formula IIa), forming a diastereomeric salt or complex that can be isolated via filtration, while the undesired isomer (Formula IIb) remains in the mother liquor. This dynamic kinetic resolution effectively enriches the optical purity of the intermediate before the final esterification. The final step involves the removal of the amino protecting group and salt formation with methanolic hydrogen chloride, yielding the target hydrochloride salt with exceptional purity, reported to reach levels as high as 99.37% in experimental embodiments.
How to Synthesize (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid methyl ester hydrochloride Efficiently
The synthesis of this high-value intermediate is executed through a logical three-stage sequence designed for maximum efficiency and yield. The process initiates with the protection of the starting amine, followed by the pivotal chiral induction and resolution phase, and concludes with deprotection and salt formation. Each stage is optimized to minimize impurity carryover and maximize throughput. For process chemists looking to implement this technology, strict adherence to temperature profiles and pH adjustments during the lithiation and resolution steps is paramount to success. The detailed standardized synthesis steps are outlined in the guide below.
- Protect the amino group of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane hydrochloride using a protecting agent like Boc-anhydride in the presence of an acid binding agent.
- Perform chiral induction using 4,4'-difluorobenzophenone and a hydrogen abstraction reagent (e.g., s-butyllithium) at low temperatures, followed by resolution with 1,2,3,4-tetrahydro-1-naphthylamine.
- Remove the amino protecting group and perform acid salt formation with methanolic hydrogen chloride to obtain the final high-purity hydrochloride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthetic route described in CN103435532A offers compelling strategic advantages that extend beyond mere technical feasibility. The primary value driver is the substantial reduction in manufacturing costs achieved by eliminating the reliance on precious metal catalysts. In traditional pharmaceutical manufacturing, the cost of palladium and the associated infrastructure for metal scavenging can represent a significant portion of the COGS (Cost of Goods Sold). By replacing Pd-C with organic chiral inducers and conventional bases, this process removes a major cost center, allowing for more competitive pricing structures in the final API market.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts like Pd-C and Pt-C, along with hazardous reagents like LiAlH4, fundamentally alters the cost structure of production. Without the need for specialized equipment to handle pyrophoric materials or complex systems to remove trace heavy metals to ppm levels, capital expenditure (CAPEX) and operational expenditure (OPEX) are significantly lowered. Furthermore, the use of common solvents like methyl tert-butyl ether (MTBE) and ethyl acetate, which are easily recovered and recycled, contributes to a leaner and more cost-effective solvent management strategy.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability and stability of raw materials. This novel route utilizes commoditized chemicals that are widely available in the global market, reducing the risk of supply disruptions caused by the scarcity of specialized catalysts. The simplified process flow, characterized by fewer unit operations and shorter reaction times, also enhances production agility. Manufacturers can respond more rapidly to fluctuations in market demand, ensuring a continuous and reliable supply of high-purity pharmaceutical intermediates to downstream API producers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a greener alternative to legacy processes. The avoidance of heavy metals reduces the burden of hazardous waste disposal and simplifies regulatory compliance regarding residual solvents and metals. The shorter reaction times and reduced energy consumption for heating and drying further diminish the carbon footprint of the manufacturing process. These factors make the technology highly scalable, facilitating the transition from pilot plant batches to multi-ton commercial production with minimal environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the practical benefits of this novel approach for industry stakeholders.
Q: How does this new synthesis method reduce production costs compared to prior art?
A: Unlike previous methods (e.g., WO2004/113295) that relied on expensive palladium-carbon (Pd-C) catalysts and hazardous lithium aluminum hydride, this novel route utilizes conventional reagents and avoids precious metals entirely, significantly lowering raw material and waste treatment costs.
Q: What is the key to achieving high optical purity in this process?
A: The process employs 4,4'-difluorobenzophenone as a chiral inductive agent combined with precise pH control (2.5–4.0) during the resolution step. This specific environment favors the formation of the desired carbanion and facilitates the selective crystallization of the target stereoisomer using 1,2,3,4-tetrahydro-1-naphthylamine.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It features fewer reaction steps, shorter reaction times (3-4 hours for the key resolution step versus days in prior art), and avoids complex drying procedures, making it highly robust for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boceprevir Intermediate Supplier
The technological advancements detailed in patent CN103435532A underscore the immense potential for optimizing the production of Hepatitis C therapeutics. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory methodologies into robust commercial realities. As a premier CDMO partner, we bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to market. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest global standards.
We invite you to collaborate with us to leverage this cost-effective synthesis for your supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can improve your margins. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the pharmaceutical marketplace.
