Advanced Olanzapine Synthesis and Purification via Acid Addition Salt Technology
Introduction to Novel Olanzapine Purification Technology
The pharmaceutical industry continuously seeks robust methodologies for producing high-purity antipsychotic agents, and patent CN101084222A introduces a transformative approach to synthesizing and purifying Olanzapine. This intellectual property addresses the longstanding challenge of coloration and trace impurities inherent in thiophene-based chemical processes, which often plague traditional manufacturing routes. By leveraging a novel purification strategy involving the formation of acid addition salts, the technology enables the production of pharmaceutically acceptable, decolorized final products with exceptional purity profiles. This method is particularly critical for manufacturers aiming to supply reliable pharmaceutical intermediates that meet stringent regulatory standards for mental health treatments. The core innovation lies not just in the synthesis but in the downstream processing, where converting the crude base into specific salt forms allows for the selective precipitation of the active ingredient while leaving complex impurities in the solution phase. This represents a significant leap forward in process chemistry, offering a viable pathway for the commercial scale-up of complex pharmaceutical intermediates without the need for expensive chromatographic separations.
![Chemical structure of Olanzapine (2-methyl-4-(4-methyl-1-piperazinyl)-10H-thieno[2,3-b][1,5]benzodiazepine)](/insights/img/olanzapine-purification-acid-salt-pharma-supplier-20260305092743-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
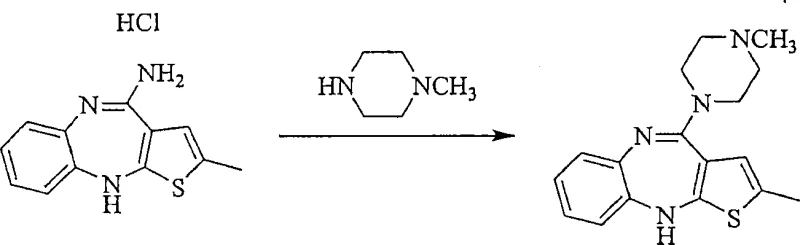
Historically, the synthesis of Olanzapine has relied on one-step alkylation methods, such as those disclosed in EP454436B1, where 4-amino-2-methyl-10H-thieno[2,3-b][1,5]benzodiazepine hydrochloride reacts directly with N-methylpiperazine. While conceptually straightforward, these conventional routes suffer from significant drawbacks, primarily the intense coloration of the final product caused by the thiophene ring chemistry. Furthermore, purification via simple recrystallization from acetonitrile or column chromatography on magnesium silicate supports is often insufficient to remove highly related impurities that possess similar physicochemical properties to the target molecule. The presence of unreacted starting materials and side products necessitates multiple purification cycles, which drastically reduces overall yield and increases manufacturing costs. Additionally, the use of harsh conditions or specific solvents in these legacy processes can lead to the formation of difficult-to-remove byproducts, complicating the supply chain for high-purity pharmaceutical intermediates. Consequently, manufacturers face challenges in consistently delivering material that meets the rigorous quality specifications required for global pharmaceutical markets.
The Novel Approach
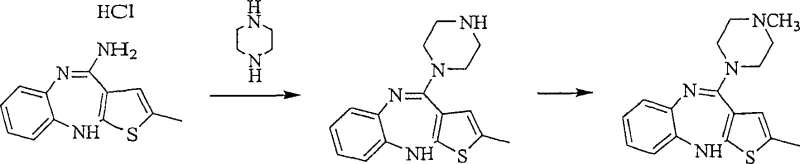
In contrast to legacy methods, the novel approach detailed in the patent utilizes a two-step synthesis pathway followed by a sophisticated acid addition salt purification protocol. This method first generates N-desmethylolanzapine as a distinct intermediate, which is subsequently methylated to form crude Olanzapine. The true breakthrough, however, occurs in the purification stage, where the crude product is converted into an acid addition salt, such as an oxalate or fumarate salt, in an organic solvent system. This conversion exploits the differential solubility and crystallization behavior of the salt form compared to impurities like N-desmethylolanzapine and dimeric byproducts. By precipitating the drug substance as a salt, manufacturers can effectively separate it from contaminants that remain dissolved in the mother liquor. This strategy not only resolves the persistent issue of product coloration, yielding a bright yellow or white crystalline powder, but also ensures that single impurity levels are driven down to below 0.1%. This robust methodology provides a scalable and efficient route for cost reduction in pharmaceutical intermediate manufacturing, bypassing the limitations of direct alkylation.
Mechanistic Insights into Acid Addition Salt Crystallization
The mechanistic foundation of this purification technology rests on the selective formation and crystallization of organic acid addition salts. When crude Olanzapine is treated with organic acids like oxalic acid, fumaric acid, or benzoic acid in solvents such as tetrahydrofuran or acetonitrile, it forms a salt that exhibits distinct crystal lattice properties. These properties differ significantly from those of the impurities, particularly the N-desmethylolanzapine and the dimeric species known as piperazine 1,4-di-4-yl-(2-methyl)-10H-thieno[2,3-b][1,5]benzodiazepine. The patent data indicates that while the target Olanzapine salt precipitates efficiently, these structurally similar impurities tend to remain in the solution phase or form salts with different solubility profiles that do not co-crystallize. This phenomenon is critical for achieving high purity, as standard recrystallization of the free base often fails to discriminate between these closely related molecules. Furthermore, the process allows for the regeneration of the free base from the purified salt by pH adjustment and extraction, ensuring the final API is free from residual acid. This chemical maneuvering provides a powerful tool for impurity control, ensuring that the final product meets the stringent requirements for pharmaceutical grade materials.
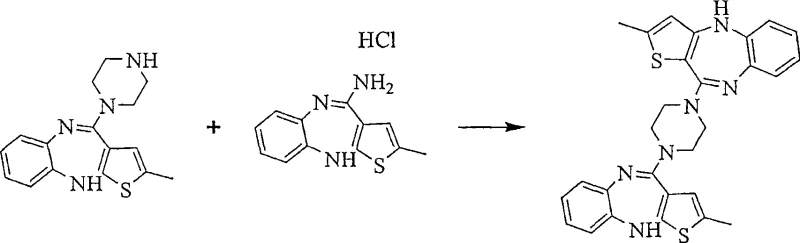
Impurity control is further enhanced by the specific reaction conditions employed during the N-methylation step, which precedes the salt formation. The patent highlights that using specific solvent mixtures, such as tetrahydrofuran combined with polar aprotic solvents like dimethylformamide or N-methylpyrrolidone, optimizes the ratio of mono-methylated product over di-methylated side products. The careful selection of bases, ranging from tertiary amines like triethylamine to strong inorganic bases like sodium hydride, allows for precise control over the reaction kinetics. This precision minimizes the formation of N,N-dimethylolanzapine, a common byproduct that is difficult to separate once formed. Additionally, the process includes a derivatization step where residual N-desmethylolanzapine can be chemically modified into a derivative, such as an acetyl or chloroacetyl species, which prevents it from precipitating with the Olanzapine acid salt. This multi-faceted approach to impurity management—combining kinetic control during synthesis with thermodynamic control during crystallization—ensures a final product with minimal coloration and exceptionally low levels of genotoxic or pharmacologically active impurities.
How to Synthesize Olanzapine Efficiently
The synthesis of high-purity Olanzapine via this patented route involves a sequence of carefully controlled chemical transformations designed to maximize yield while minimizing impurity carryover. The process begins with the preparation of the N-desmethylolanzapine intermediate, followed by its methylation and subsequent purification through acid salt formation. Detailed operational parameters, including specific solvent ratios, temperature gradients, and stoichiometric equivalents, are critical for reproducing the high purity levels described in the patent examples. For instance, the use of warm water to precipitate the intermediate and specific organic solvents for the final salt crystallization are key unit operations that define the success of the campaign. Manufacturers must adhere to these precise conditions to ensure the effective removal of the dimer impurity and residual starting materials. The following guide outlines the standardized synthesis steps derived from the patent data, providing a roadmap for technical teams to implement this advanced purification strategy in a pilot or commercial setting.
- React 4-amino-2-methyl-10H-thieno[2,3-b][1,5]benzodiazepine hydrochloride with piperazine in high-boiling aliphatic alcohols to form N-desmethylolanzapine.
- Perform N-methylation of the intermediate using methylating agents like methyl iodide in THF or polar solvent mixtures under basic conditions.
- Purify the crude olanzapine by converting it into an acid addition salt (e.g., oxalate or fumarate), separating crystals, and regenerating the free base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this acid addition salt purification technology offers substantial strategic advantages beyond mere technical compliance. By eliminating the need for complex chromatographic purification steps, which are often bottlenecks in large-scale manufacturing, the process drastically simplifies the production workflow. This simplification translates directly into enhanced supply chain reliability, as the risk of batch failure due to purification inefficiencies is significantly mitigated. The ability to consistently produce off-white or bright yellow crystalline material without extensive reprocessing means that lead times for high-purity pharmaceutical intermediates can be reduced, ensuring a steady flow of material to downstream API manufacturers. Furthermore, the use of commercially available organic acids and standard solvents ensures that raw material sourcing remains stable and cost-effective, avoiding reliance on exotic or supply-constrained reagents. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery windows of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of expensive chromatography media and the reduction in solvent consumption associated with multiple recrystallization cycles lead to significant operational cost savings. By achieving high purity through a single crystallization of the acid salt, manufacturers avoid the yield losses typically incurred during repetitive purification attempts. This efficiency lowers the overall cost of goods sold, allowing for more competitive pricing structures in the generic pharmaceutical market. Additionally, the recovery of the free base from the salt form is highly efficient, minimizing material loss and maximizing the throughput of the existing reactor infrastructure. These factors combine to create a leaner, more cost-effective manufacturing process that aligns with the financial goals of modern chemical enterprises.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent product quality, which is a cornerstone of supply chain reliability. By effectively controlling critical impurities like the dimer and N-desmethylolanzapine, the process reduces the likelihood of out-of-specification batches that could disrupt supply commitments. The use of standard, widely available reagents further insulates the supply chain from volatility in the raw material market. This reliability allows procurement teams to forecast inventory needs with greater accuracy and maintain lower safety stock levels, optimizing working capital. Ultimately, a stable and predictable manufacturing process fosters stronger partnerships between suppliers and pharmaceutical companies, ensuring uninterrupted availability of this critical antipsychotic intermediate.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as filtration, crystallization, and extraction that are easily transferred from laboratory to commercial scale. The avoidance of heavy metal catalysts or hazardous reagents simplifies waste stream management and reduces the environmental footprint of the manufacturing process. This alignment with green chemistry principles facilitates easier regulatory approval and compliance with increasingly stringent environmental regulations. The ability to scale up without compromising purity or yield makes this technology an ideal candidate for meeting the growing global demand for Olanzapine, ensuring that supply can expand in tandem with market needs without requiring prohibitive capital investment in specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Olanzapine purification technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the mechanism and benefits of the acid addition salt method. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production. The answers highlight the specific advantages of this approach over conventional methods, focusing on impurity profiles, solvent systems, and final product quality. This information serves as a foundational resource for decision-makers assessing the potential impact of this technology on their current manufacturing portfolios and supply chain strategies.
Q: How does the acid addition salt method improve Olanzapine purity compared to recrystallization?
A: The acid addition salt method effectively separates Olanzapine from highly related impurities like N-desmethylolanzapine and dimers that have similar physicochemical properties to the free base, which simple recrystallization often fails to remove.
Q: What specific impurities are targeted by this novel purification process?
A: This process specifically targets the removal of the dimer impurity (piperazine 1,4-di-4-yl derivative) and residual N-desmethylolanzapine, ensuring final product purity exceeds 99.5% with minimal coloration.
Q: Which organic acids are preferred for forming the purification salts?
A: Carboxylic acids containing at least one carboxyl group, such as oxalic acid, fumaric acid, and benzoic acid, are preferred, with oxalic acid showing particularly excellent precipitation and separation capabilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olanzapine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of psychiatric medication intermediates like Olanzapine. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated acid addition salt purification method described in patent CN101084222A can be seamlessly implemented at an industrial level. We are committed to delivering high-purity Olanzapine that meets stringent purity specifications, utilizing our rigorous QC labs to verify that impurity levels, including dimers and N-desmethyl derivatives, are maintained well below regulatory thresholds. Our facility is equipped to handle the specific solvent systems and temperature controls required for this process, guaranteeing a product that is not only chemically pure but also free from the discoloration issues that plague lesser manufacturing routes.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits associated with this purification technology. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Our goal is to provide a transparent and collaborative partnership, ensuring that you have access to the highest quality Olanzapine intermediates to support your drug development and commercialization efforts. Let us help you secure a reliable supply of this vital compound through our proven expertise in fine chemical manufacturing.
