Advanced Crystallization Strategies for High-Purity Olanzapine Form II Manufacturing
Introduction to Advanced Olanzapine Polymorph Control
The pharmaceutical industry constantly seeks robust manufacturing processes that guarantee the highest levels of purity and stability for active pharmaceutical ingredients (APIs). In the realm of antipsychotic medications, Olanzapine stands out as a critical compound, yet its production is fraught with challenges related to polymorphism. Patent CN1122036C provides a groundbreaking solution by detailing processes for the preparation of Olanzapine and its intermediates, specifically targeting the production of the highly stable anhydrous Form II. This patent addresses the critical need to control crystalline forms, ensuring that the final drug product meets stringent regulatory standards for stability and bioavailability. The invention introduces specific dihydrate intermediates—designated as Dihydrate D, Dihydrate B, and Dihydrate E—which serve as precise precursors to the desired Form II. By leveraging these hydrated forms, manufacturers can bypass the difficulties associated with direct crystallization of the anhydrous form, thereby achieving a product that is substantially free of organic solvent residues. This approach not only enhances the safety profile of the API but also streamlines the quality control processes required for commercial scale-up. 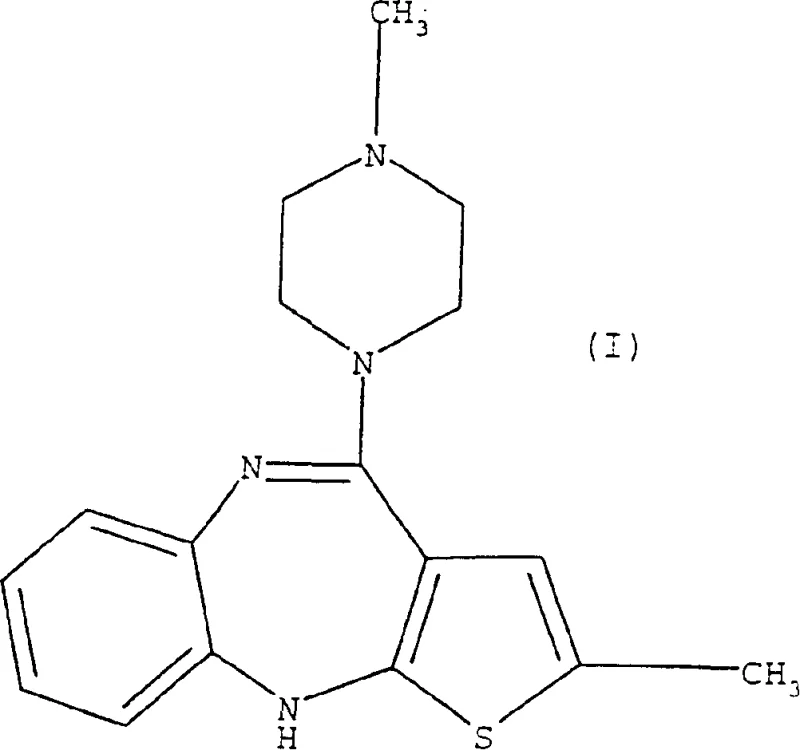
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing Olanzapine often struggle with the inconsistent formation of polymorphs, leading to batches that may contain undesirable crystalline forms such as Form I. Form I, while an anhydrate, is not as pharmaceutically desirable as Form II for certain formulations due to stability concerns. Conventional processes frequently rely on organic solvents that can become entrapped within the crystal lattice, necessitating complex and costly drying procedures to meet residual solvent limits. Furthermore, direct crystallization of the anhydrous form requires extremely careful control of conditions, which is difficult to maintain consistently on a large industrial scale. Variations in temperature, cooling rates, or solvent composition can easily result in the precipitation of the wrong polymorph or solvates, leading to significant yield losses and batch rejections. These inconsistencies pose a major risk to supply chain reliability and increase the overall cost of goods sold due to the need for extensive reprocessing or disposal of off-spec material.
The Novel Approach
The methodology outlined in Patent CN1122036C represents a paradigm shift by utilizing stable dihydrate intermediates as a gateway to Form II. Instead of attempting to crystallize the anhydrous form directly from organic media, this novel approach involves converting technical grade Olanzapine into specific dihydrate polymorphs under aqueous conditions. This strategy effectively eliminates the risk of organic solvent entrapment, as the crystallization occurs in water or water-miscible solvent systems that are easily removed. The patent identifies three distinct dihydrate forms (D, B, and E), each characterized by unique X-ray powder diffraction patterns, allowing for precise identification and quality assurance. Once the pure dihydrate is isolated, it is subjected to a controlled drying process in a vacuum oven at moderate temperatures, typically around 50°C. This gentle dehydration converts the dihydrate into the thermodynamically stable Form II without inducing degradation or forming unwanted polymorphs. This two-step process—hydration followed by controlled dehydration—offers a much wider operating window and greater reproducibility compared to direct anhydrous crystallization methods.
Mechanistic Insights into Polymorphic Transformation
The core mechanism driving this process is the controlled manipulation of the crystal lattice through hydration and dehydration cycles. The dihydrate intermediates possess a crystalline structure where water molecules are integral components of the lattice, stabilizing the molecule in a specific conformation. When technical grade Olanzapine is stirred in aqueous solvents, the system equilibrates to form the most stable hydrate under those specific conditions, whether it be Dihydrate D, B, or E. The patent provides detailed X-ray powder diffraction data for each of these forms, serving as a fingerprint for process monitoring. For instance, Dihydrate D exhibits characteristic d-spacings such as 9.4511 Å and 7.7098 Å, while Dihydrate B shows peaks at 9.9045 Å and 6.9985 Å. These distinct patterns allow R&D teams to verify the identity of the intermediate before proceeding to the final drying step, ensuring that only the correct precursor is converted to Form II. The subsequent dehydration step involves the removal of these lattice water molecules under vacuum, which triggers a rearrangement of the crystal structure into the dense, stable packing of Form II. This solid-state transformation is critical because it avoids the dissolution and recrystallization steps that often introduce impurities or generate mixed polymorphic systems.
Impurity control is inherently built into this mechanism through the use of aqueous crystallization. Organic impurities and residual solvents from the initial synthesis step are largely excluded from the growing dihydrate crystals due to the high polarity of the aqueous medium. The patent specifies that the resulting Form II contains less than 0.5% related substances, a testament to the purification power of this crystallization route. Furthermore, the process minimizes the formation of solvates, which are common contaminants in APIs crystallized from organic solvents. By ensuring the intermediate is a true dihydrate with two water molecules per drug molecule incorporated into the lattice, the process guarantees a stoichiometric and predictable conversion to the anhydrous form. This mechanistic understanding allows process chemists to fine-tune parameters such as stirring time and temperature to optimize the particle size distribution and bulk density of the final API, which are crucial for downstream tablet compression and formulation performance.
How to Synthesize Olanzapine Efficiently
The synthesis pathway described in the patent begins with the preparation of technical grade Olanzapine, which serves as the feedstock for the polymorph control process. This initial step involves the reaction of a specific intermediate, referred to as Intermediate 1, with N-methylpiperazine in a polar aprotic solvent like dimethylsulfoxide (DMSO). The reaction is conducted at elevated temperatures, typically around 120°C, to drive the substitution reaction to completion. Following the reaction, the mixture is cooled and precipitated using methanol and water, yielding the technical grade material which is then subjected to the hydration steps described previously. This foundational synthesis is robust and scalable, providing a reliable source of material for the subsequent polymorph conversion. The detailed operational parameters ensure that the technical grade material has high potency, setting the stage for the high-purity final product. 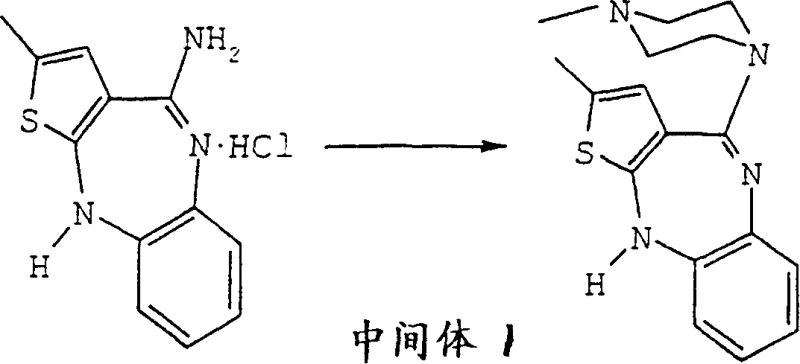
- Synthesize technical grade olanzapine by reacting Intermediate 1 with N-methylpiperazine in DMSO at 120°C.
- Convert the technical grade material into specific dihydrate polymorphs (Dihydrate D, B, or E) by stirring in aqueous solvents under controlled temperatures.
- Dry the isolated dihydrate intermediate in a vacuum oven at approximately 50°C to obtain substantially pure anhydrous Form II.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the process described in Patent CN1122036C offers significant strategic advantages beyond mere technical feasibility. The primary benefit lies in the drastic simplification of the purification train, which directly translates to cost reduction in pharmaceutical intermediate manufacturing. By utilizing aqueous crystallization to form the dihydrate intermediates, the need for expensive and hazardous organic solvent recovery systems is significantly diminished. This not only lowers the capital expenditure required for plant infrastructure but also reduces the ongoing operational costs associated with solvent purchasing, storage, and waste disposal. The elimination of complex solvent exchange steps further shortens the cycle time per batch, enhancing the overall throughput of the manufacturing facility without compromising on quality standards. Additionally, the robustness of the dihydrate-to-Form II conversion reduces the risk of batch failures, ensuring a more consistent and reliable supply of the API to downstream formulation partners.
- Cost Reduction in Manufacturing: The process eliminates the need for rigorous organic solvent removal steps typically required to meet residual solvent specifications in direct anhydrous crystallization. By crystallizing from water or aqueous mixtures, the intermediate is inherently free of toxic organic residues, reducing the burden on analytical testing and reprocessing. This streamlined workflow leads to substantial cost savings in utility consumption and waste treatment, making the production of high-purity Olanzapine more economically viable. Furthermore, the moderate drying temperatures required for the conversion (40°C to 70°C) are energy-efficient compared to high-temperature drying or lyophilization processes, contributing to a lower carbon footprint and reduced energy costs.
- Enhanced Supply Chain Reliability: The use of well-defined dihydrate intermediates provides a stable inventory buffer that can be stored and converted to the final API on demand. Since the dihydrates are stable crystalline solids, they offer better handling characteristics and longer shelf-life compared to amorphous or metastable forms. This flexibility allows manufacturers to decouple the synthesis of the technical grade material from the final polishing step, optimizing production scheduling and inventory management. The ability to produce the API from a stable intermediate mitigates the risk of supply disruptions caused by polymorphic instability, ensuring continuous availability for global pharmaceutical markets. Moreover, the starting materials such as Intermediate 1 and N-methylpiperazine are commercially available commodity chemicals, reducing dependency on specialized or scarce reagents.
- Scalability and Environmental Compliance: The aqueous nature of the crystallization process aligns perfectly with green chemistry principles, minimizing the generation of volatile organic compounds (VOCs). This makes the process highly scalable from pilot plant to commercial production without encountering the safety hazards associated with large volumes of flammable organic solvents. Regulatory agencies favor processes that minimize solvent residues, facilitating faster approval times for new drug applications or generic filings. The clear X-ray diffraction signatures for each polymorph also simplify regulatory documentation, as the physical form of the drug can be unequivocally proven and monitored throughout the product lifecycle. This compliance advantage reduces the risk of regulatory delays and ensures long-term market access for the finished drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Olanzapine Form II using the patented dihydrate method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and benefits of this approach for industry stakeholders. Understanding these details is essential for R&D teams evaluating technology transfer and for procurement professionals assessing supplier capabilities. The answers highlight the specific advantages of the dihydrate route over conventional methods, focusing on purity, stability, and process control.
Q: Why is Olanzapine Form II preferred over Form I for pharmaceutical formulations?
A: Form II is the most stable anhydrous form of olanzapine, providing stable anhydrous formulations with pharmaceutically desirable properties, whereas Form I is less desirable for certain drug products.
Q: How does the dihydrate intermediate method improve product purity?
A: Using dihydrate intermediates allows for preparation under aqueous conditions, ensuring the final Form II material is substantially free of any organic solvent residues, which is critical for safety and compliance.
Q: What are the critical processing conditions for converting dihydrates to Form II?
A: The conversion requires drying the olanzapine dihydrate in a vacuum oven at temperatures between 40°C and 70°C, typically around 50°C, until the desired crystalline Form II is formed.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olanzapine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of polymorph control in the manufacture of high-value pharmaceutical intermediates like Olanzapine. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate crystallization processes described in patents like CN1122036C are executed with precision. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced X-ray powder diffraction instruments to verify the polymorphic form of every batch. Our commitment to quality ensures that the Olanzapine we supply meets the exacting standards required for global regulatory submissions, providing our partners with peace of mind regarding product consistency and safety.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of how our optimized processes can reduce your overall cost of goods. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Whether you are developing a new generic formulation or seeking a reliable backup supplier for existing products, our expertise in complex crystallization chemistry positions us as a strategic partner dedicated to your success in the competitive pharmaceutical marketplace.
