Advanced Copper-Catalyzed Synthesis of Pyrrolo[1,2-a]quinoxaline Derivatives for Pharmaceutical Applications
Advanced Copper-Catalyzed Synthesis of Pyrrolo[1,2-a]quinoxaline Derivatives for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex heterocyclic scaffolds that serve as critical backbones for bioactive molecules. Patent CN112457339B introduces a transformative approach to synthesizing pyrrolo[1,2-a]quinoxaline derivatives, a structural motif prevalent in numerous natural products and therapeutic agents. This innovation centers on the utilization of a novel N,N-coordinated cuprous complex containing a meta-carborane ligand, which acts as a highly efficient catalyst for the coupling of 2-bromoaniline with pyrrole formaldehyde compounds. Unlike conventional methods that often struggle with harsh reaction conditions and limited substrate scope, this patented technology operates under remarkably mild parameters, typically between 50°C and 65°C, while achieving exceptional yields. For R&D directors and procurement specialists alike, this represents a significant leap forward in process chemistry, offering a pathway to high-purity intermediates with reduced operational complexity and enhanced economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrrolo[1,2-a]quinoxaline derivatives has been plagued by significant technical and economic hurdles that hinder large-scale manufacturing. Prior art methodologies frequently rely on the use of expensive aryl iodides rather than more abundant aryl bromides, drastically inflating raw material costs. Furthermore, traditional copper-catalyzed systems often necessitate the use of complex, air-sensitive phosphine ligands that are not only costly but also difficult to handle on an industrial scale. Many existing protocols require elevated reaction temperatures exceeding 110°C or even 130°C, which poses safety risks and increases energy consumption. Additionally, some routes involve multi-step sequences including the hydrolysis of trifluoroacetyl groups or the formation of intermolecular amino compounds, leading to lower overall atom economy and increased waste generation. These factors collectively create a bottleneck for supply chain managers aiming to secure reliable sources of these valuable intermediates at a competitive price point.
The Novel Approach
The methodology disclosed in CN112457339B effectively dismantles these barriers through the deployment of a specialized cuprous complex catalyst. This new approach allows for the direct use of cheap and easily obtained 2-bromoaniline compounds, bypassing the need for premium-priced iodinated substrates. The reaction proceeds efficiently in a one-pot fashion using simple inorganic bases like potassium carbonate or sodium carbonate in toluene, eliminating the requirement for exotic ligands. The mild reaction conditions, ranging from 50°C to 65°C over a period of 6 to 8 hours, ensure high substrate universality and minimize the formation of thermal degradation byproducts. This streamlined process not only simplifies the workflow but also enhances the purity profile of the final product, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Carborane-Ligand Stabilized Copper Catalysis
The core of this technological breakthrough lies in the unique structure and stability of the N,N-coordinated cuprous complex containing the meta-carborane ligand. The meta-carborane cage provides a rigid, electron-deficient framework that stabilizes the copper center against oxidation and aggregation, which are common failure modes in traditional copper catalysis. This stabilization allows the catalyst to remain active and selective even under prolonged heating, as evidenced by thermogravimetric analysis showing stability up to 300°C. The catalyst facilitates the oxidative addition of the aryl bromide to the copper center, followed by coordination with the pyrrole aldehyde and subsequent cyclization. The robust nature of the carborane ligand ensures that the catalytic cycle proceeds with high turnover numbers, minimizing the required catalyst loading to a molar ratio of merely 0.01 to 0.03 relative to the substrate.
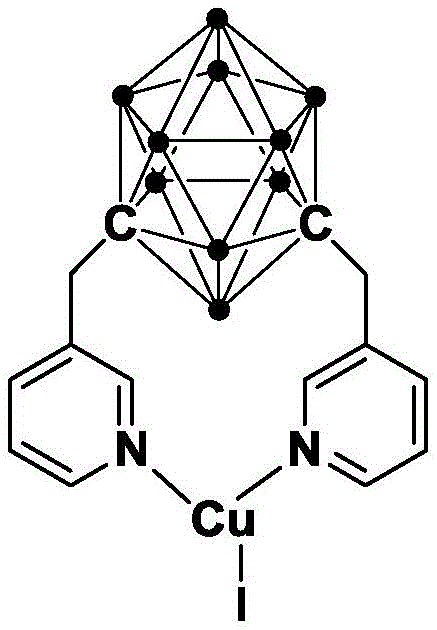
Furthermore, the mechanistic pathway promotes high selectivity, effectively suppressing side reactions that typically lead to difficult-to-remove impurities. The electronic properties of the carborane ligand modulate the Lewis acidity of the copper ion, optimizing it for the specific activation of the C-Br bond in 2-bromoaniline without promoting non-specific decomposition of the sensitive pyrrole aldehyde moiety. This precise control over the reaction environment is crucial for maintaining a clean impurity profile, which is a primary concern for regulatory compliance in API manufacturing. The catalyst's insensitivity to air and water further simplifies the operational protocol, removing the need for stringent inert atmosphere techniques that often complicate scale-up efforts in standard manufacturing facilities.
How to Synthesize Pyrrolo[1,2-a]quinoxaline Derivatives Efficiently
The practical implementation of this synthesis is straightforward and designed for immediate adoption in process development laboratories. The procedure involves dissolving the specific cuprous complex catalyst, the 2-bromoaniline substrate, the chosen pyrrole formaldehyde compound, and an inorganic base in toluene. The mixture is then heated to the specified mild temperature range, allowing the cyclization to proceed to completion within a standard work shift. Following the reaction, the product can be isolated through standard silica gel column chromatography, yielding the target pyrrolo[1,2-a]quinoxaline derivative in high purity. The detailed standardized synthesis steps, including precise reagent quantities and workup procedures, are outlined in the guide below to facilitate rapid technology transfer.
- Dissolve the cuprous complex catalyst, 2-bromoaniline, pyrrole formaldehyde compound, and alkali base (K2CO3 or Na2CO3) in an organic solvent such as toluene.
- Maintain the reaction mixture at a mild temperature between 50°C and 65°C for a duration of 6 to 8 hours to ensure complete conversion.
- Upon completion, separate and purify the resulting pyrrolo[1,2-a]quinoxaline derivative via silica gel column chromatography to obtain the high-purity target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthetic route offers compelling strategic benefits that extend beyond mere technical performance. The shift from expensive aryl iodides to commodity-grade 2-bromoanilines represents a fundamental reduction in raw material expenditure, directly impacting the cost of goods sold (COGS). Moreover, the elimination of complex ligand synthesis and the use of a stable, reusable catalyst system simplifies the supply chain, reducing dependency on niche chemical suppliers. The mild reaction conditions also translate to lower energy costs and reduced wear on reactor equipment, contributing to long-term operational savings. These factors combine to create a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the substitution of high-cost inputs with readily available commodities. By utilizing 2-bromoaniline instead of 2-iodoaniline, manufacturers can achieve substantial cost savings on starting materials, which often constitute a significant portion of total production costs. Additionally, the catalyst system does not require expensive phosphine ligands, further lowering the reagent bill. The high yields reported, often exceeding 90%, mean that less raw material is wasted, maximizing the output per batch and improving overall process efficiency without compromising quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the use of widely available raw materials. 2-Bromoaniline and simple pyrrole aldehydes are produced by multiple global vendors, mitigating the risk of supply disruptions associated with specialized or proprietary reagents. The robustness of the catalyst, which is insensitive to air and moisture, reduces the logistical complexities of storage and transport. This reliability ensures that production schedules can be maintained consistently, reducing lead times for high-purity intermediates and allowing for more agile responses to market demand fluctuations.
- Scalability and Environmental Compliance: From a scale-up perspective, the one-pot nature of the reaction and the absence of hazardous high-temperature conditions make this process inherently safer and easier to implement in large-scale reactors. The reduction in waste generation aligns with increasingly stringent environmental regulations, minimizing the costs associated with waste disposal and treatment. The simplified workup procedure, which avoids complex extraction or protection-deprotection sequences, reduces solvent consumption and processing time. This green chemistry profile not only lowers the environmental footprint but also streamlines the path to regulatory approval for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic methodology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential partners and licensees.
Q: What are the primary advantages of the carborane-ligand cuprous complex over traditional copper catalysts?
A: The novel cuprous complex featuring a meta-carborane ligand exhibits exceptional thermal stability up to 300°C and insensitivity to air and moisture. Unlike traditional systems requiring expensive phosphine ligands or harsh conditions, this catalyst enables the use of cost-effective 2-bromoaniline substrates under mild temperatures (50-65°C), significantly reducing operational costs and safety risks.
Q: Does this synthetic method support a broad range of substrate variations?
A: Yes, the methodology demonstrates high substrate universality. It successfully accommodates various pyrrole aldehyde derivatives, including those with phenyl, methyl, chloro, and fused ring substitutions (such as indole and benzimidazole aldehydes), consistently delivering high yields ranging from 85% to 96% without the need for substrate-specific optimization.
Q: How does this process impact environmental compliance and waste generation?
A: The process is designed for green chemistry principles, utilizing a one-pot reaction strategy that minimizes solvent usage and intermediate isolation steps. By avoiding complex protection-deprotection sequences and operating at lower temperatures, the method generates significantly less waste residue compared to prior art, facilitating easier downstream purification and reduced environmental burden.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolo[1,2-a]quinoxaline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable synthesis routes in the modern pharmaceutical landscape. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the carborane-copper catalyzed synthesis are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of pyrrolo[1,2-a]quinoxaline derivative meets the highest quality standards required for downstream API synthesis. We are committed to leveraging our technical expertise to optimize these processes further, ensuring maximum yield and minimal environmental impact for our global clientele.
We invite you to collaborate with us to explore the full potential of this advanced synthetic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this methodology can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, cost-effective supply of these vital pharmaceutical intermediates.
