Revolutionizing Dihydropyrrole Synthesis: A Cost-Effective Iron-Catalyzed Route for Commercial Scale-Up
Introduction to Advanced Dihydropyrrole Manufacturing
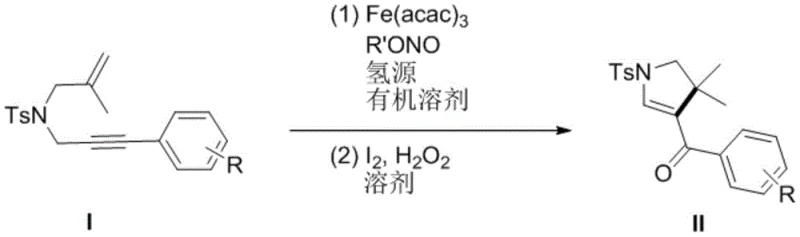
The landscape of fine chemical synthesis is undergoing a paradigm shift towards sustainability and cost-efficiency, driven by the urgent need to replace precious metal catalysts with earth-abundant alternatives. Patent CN111592481A introduces a groundbreaking methodology for the preparation of polysubstituted dihydropyrrole compounds, a critical scaffold found in numerous bioactive molecules and pharmaceutical agents. This innovation leverages a specific trivalent iron salt, Fe(acac)3, as a robust catalyst, coupled with tert-butyl nitrite as an oxidant and polymethylsiloxane (PMHS) as a hydrogen source. By operating under mild thermal conditions and eliminating the need for strong acids or bases, this technology addresses long-standing challenges in heterocyclic chemistry. For global procurement and R&D teams, this represents a strategic opportunity to secure a reliable polysubstituted dihydropyrrole supplier capable of delivering high-quality intermediates with a significantly reduced environmental footprint and optimized cost structure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted dihydropyrrole derivatives has been heavily reliant on noble metal catalysis, utilizing expensive transition metals such as Rhodium, Nickel, Silver, or Palladium. These conventional pathways often necessitate harsh reaction conditions, including elevated temperatures and the use of corrosive acids or bases, which pose significant safety risks and operational complexities in a manufacturing setting. Furthermore, the presence of heavy metal residues in the final product requires rigorous and costly purification steps to meet stringent pharmaceutical purity specifications. The economic burden of these precious metals, combined with their supply chain volatility and toxicity concerns, has severely limited the widespread application of these synthetic routes in large-scale drug production. Consequently, the industry has faced a bottleneck in achieving cost-effective and green synthesis for these valuable nitrogen-containing heterocycles.
The Novel Approach
The patented technology offers a transformative solution by substituting expensive noble metals with inexpensive and non-toxic iron catalysts. This novel approach utilizes a tandem reaction sequence where an iron-catalyzed radical cyclization is followed by an iodine-mediated oxidation, effectively constructing the dihydropyrrole core under remarkably mild conditions. The process operates efficiently at temperatures ranging from 30°C to 60°C, drastically reducing energy consumption compared to traditional high-temperature methods. Moreover, the elimination of acid-base additives simplifies the workup procedure and minimizes waste generation, aligning perfectly with modern green chemistry principles. This shift not only enhances the economic viability of producing these intermediates but also ensures a cleaner impurity profile, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Fe(acac)3-Catalyzed Cyclization
The core of this innovative synthesis lies in the unique mechanistic pathway facilitated by the Fe(acac)3 catalyst. The reaction initiates with the generation of an amidyl radical from the sulfonamide precursor through the activation of tert-butyl nitrite by the iron center. This radical species undergoes a selective intramolecular addition to the alkene moiety, followed by a cascade cyclization involving the alkyne group, ultimately forming the dihydropyrrole ring system. The use of PMHS as a hydrogen source is critical in this step, providing the necessary hydride equivalents to terminate the radical chain and stabilize the intermediate without introducing harsh reducing agents. This radical manifold allows for high functional group tolerance, accommodating various substituents on the aromatic ring, such as methoxy, fluoro, and trifluoromethyl groups, without compromising the reaction efficiency or selectivity.
Following the cyclization, the second stage involves an oxidative transformation mediated by molecular iodine and hydrogen peroxide. This step is crucial for converting the initial cyclized intermediate into the final carbonyl-functionalized dihydropyrrole product. The mild oxidative conditions prevent over-oxidation or degradation of the sensitive heterocyclic core, ensuring high product integrity. From an impurity control perspective, the specificity of the iron catalyst and the mild nature of the oxidants minimize the formation of side products commonly associated with aggressive transition metal catalysis. This results in a cleaner crude reaction mixture, which significantly reduces the burden on downstream purification processes and enhances the overall yield and purity of the final active pharmaceutical ingredient intermediate.
How to Synthesize Polysubstituted Dihydropyrrole Efficiently
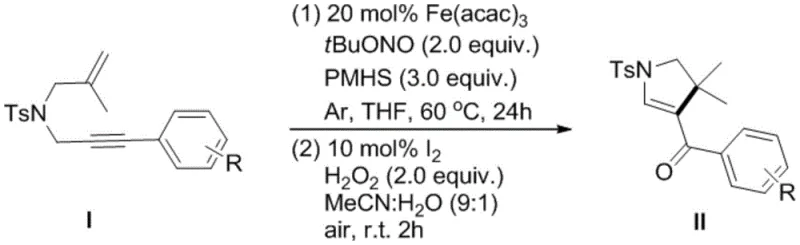
The implementation of this synthesis route is straightforward and designed for scalability, making it accessible for both laboratory optimization and industrial production. The process begins with the preparation of the reaction mixture under an inert atmosphere to prevent premature oxidation of sensitive intermediates. Precise control of stoichiometry, particularly the ratio of the hydrogen source and oxidant, is essential to maximize conversion while minimizing byproduct formation. The subsequent workup involves standard extraction and concentration techniques, followed by a mild oxidative treatment that can be performed at ambient temperature. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis protocol provided below.
- Combine N-2-methylallyl-N-3-arylpropargyl p-toluenesulfonamide with Fe(acac)3, tert-butyl nitrite, and PMHS in THF under inert atmosphere at 60°C for 24 hours.
- Quench the reaction with water, extract with ethyl acetate, and concentrate to obtain the crude intermediate product.
- Dissolve the crude product in acetonitrile/water, add catalytic I2 and H2O2, react at room temperature for 2 hours, then purify via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed technology translates into tangible strategic advantages that extend beyond simple unit cost savings. The replacement of volatile precious metals with stable, commodity-grade iron salts fundamentally alters the cost structure of the supply chain, insulating the manufacturing process from the price fluctuations typical of the noble metal market. Additionally, the use of common organic solvents like tetrahydrofuran and acetonitrile, along with commercially available reagents like PMHS and tert-butyl nitrite, ensures a robust and resilient supply chain with minimal risk of raw material shortages. This reliability is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete elimination of expensive noble metal catalysts such as Rhodium or Palladium, which traditionally account for a substantial portion of raw material costs in heterocyclic synthesis. By utilizing Fe(acac)3, a widely available and low-cost industrial chemical, manufacturers can achieve drastic reductions in direct material expenses. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, while the simplified purification process lowers solvent usage and waste disposal costs, collectively driving down the total cost of ownership for the final intermediate.
- Enhanced Supply Chain Reliability: Sourcing high-purity noble metal catalysts often involves complex logistics and long lead times due to their geopolitical concentration and limited mining output. In contrast, iron salts and the associated reagents for this process are produced globally in massive quantities, ensuring a stable and diversified supply base. This abundance mitigates the risk of supply disruptions and allows for more flexible inventory management strategies. The ability to source all key inputs from multiple vendors enhances the overall resilience of the supply chain, ensuring consistent availability of high-purity pharmaceutical intermediates even during market volatility.
- Scalability and Environmental Compliance: The mild thermal profile of this reaction, operating effectively between 30°C and 60°C, makes it inherently safer and easier to scale from kilogram to multi-ton production volumes. Lower temperatures reduce the thermal load on reactor systems and minimize the risk of runaway reactions, facilitating smoother technology transfer from R&D to commercial manufacturing. Moreover, the absence of heavy metals and corrosive acids simplifies wastewater treatment and regulatory compliance, aligning with increasingly stringent environmental regulations. This green chemistry profile not only reduces the environmental impact but also accelerates regulatory approval processes for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed synthesis route. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the practical aspects of adopting this technology for industrial applications. Understanding these nuances is essential for R&D teams evaluating process feasibility and procurement teams assessing supplier capabilities.
Q: What are the primary advantages of using Fe(acac)3 over noble metal catalysts for dihydropyrrole synthesis?
A: The use of Fe(acac)3 eliminates the reliance on expensive and toxic noble metals like Rhodium or Palladium, significantly reducing raw material costs and simplifying heavy metal removal processes in the final API.
Q: Does this synthesis method require harsh reaction conditions?
A: No, the patented method operates under mild conditions, specifically between 30°C and 60°C, avoiding the high temperatures often required by conventional transition metal catalysis, which enhances safety and energy efficiency.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of inexpensive reagents like PMHS and common solvents like THF and Acetonitrile, combined with mild thermal requirements, makes this route highly scalable for industrial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Dihydropyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed technology in reshaping the economics of pharmaceutical intermediate production. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle the specific requirements of radical chemistry and mild oxidation processes, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch. We are committed to delivering high-purity polysubstituted dihydropyrroles that meet the exacting standards of the global pharmaceutical industry.
We invite forward-thinking pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in green catalysis can drive value and efficiency in your supply chain.
