Advanced Iron-Catalyzed Synthesis of Polysubstituted Dihydropyrroles for Commercial Pharma Applications
Advanced Iron-Catalyzed Synthesis of Polysubstituted Dihydropyrroles for Commercial Pharma Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective methodologies for constructing nitrogen-containing heterocycles, which serve as critical scaffolds in bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN111592481B, which discloses a novel preparation method for polysubstituted dihydropyrrole compounds. This technology represents a paradigm shift from traditional noble metal catalysis to a more sustainable and economically viable iron-catalyzed system. By utilizing specific ferric salts as catalysts in conjunction with tert-butyl nitrite and polymethylsiloxane (PMHS), the process achieves efficient cyclization under remarkably mild conditions. For R&D directors and procurement managers alike, this innovation addresses the dual challenges of reducing raw material costs while maintaining the stringent purity standards required for active pharmaceutical ingredients (APIs) and advanced intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted dihydropyrrole derivatives has relied heavily on the use of precious transition metal catalysts such as Rhodium, Nickel, or Silver. These conventional pathways often necessitate harsh reaction conditions, including elevated temperatures and the presence of strong acids or bases, which can compromise the integrity of sensitive functional groups on the substrate. Furthermore, the reliance on noble metals introduces significant supply chain vulnerabilities and cost volatility, as the prices of these elements are subject to geopolitical fluctuations and mining constraints. From a manufacturing perspective, the removal of trace heavy metal residues from the final product to meet regulatory limits adds complex and expensive purification steps, thereby extending lead times and increasing the overall cost of goods sold for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in the patent utilizes an abundant and inexpensive iron catalyst, specifically Fe(acac)3, to drive the cyclization reaction. This approach eliminates the need for costly noble metals and avoids the use of corrosive acids or alkalis, creating a greener and safer operational environment. The reaction proceeds efficiently at moderate temperatures ranging from 30°C to 60°C, significantly reducing energy consumption compared to high-temperature alternatives. By employing a tandem strategy involving an initial iron-catalyzed reductive cyclization followed by an iodine-mediated oxidation, the process achieves high selectivity and yield. This streamlined workflow not only simplifies the operational procedure but also enhances the economic feasibility of producing complex pyrroline skeletons on a commercial scale.
Mechanistic Insights into Fe(acac)3-Catalyzed Cyclization
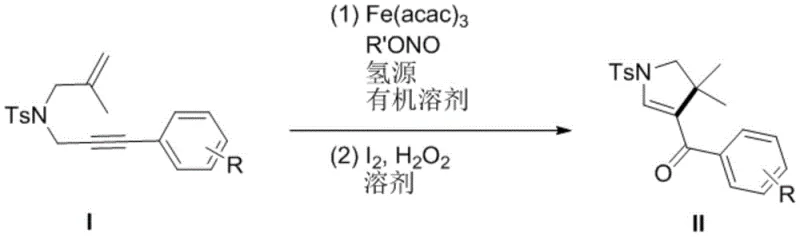
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the trivalent iron catalyst. The reaction initiates with the activation of the N-2-methallyl-N-3-arylpropargyl-p-toluenesulfonamide substrate by the Fe(acac)3 complex in the presence of tert-butyl nitrite, which acts as a radical initiator or oxidant. The polymeric siloxane (PMHS) serves as a hydride source, enabling a reductive cyclization that constructs the five-membered nitrogen-containing ring with high stereocontrol. This radical-mediated process is particularly advantageous as it tolerates a wide range of substituents on the aryl ring, including electron-donating groups like methoxy and electron-withdrawing groups such as trifluoromethyl and halogens. The robustness of this catalytic cycle ensures consistent performance across diverse substrate scopes, making it a versatile tool for medicinal chemists exploring structure-activity relationships.
Following the initial cyclization, the crude intermediate undergoes a critical oxidation step using an I2/H2O2 system. This second stage is essential for aromatizing or adjusting the oxidation state of the pyrroline ring to achieve the final desired polysubstituted dihydropyrrole structure. The use of hydrogen peroxide as a terminal oxidant is environmentally benign, producing water as the only byproduct, which aligns with modern green chemistry principles. The synergy between the iron-catalyzed reduction and the iodine-mediated oxidation creates a highly efficient cascade that minimizes the formation of unwanted byproducts. This precise control over the reaction trajectory is crucial for maintaining a clean impurity profile, thereby reducing the burden on downstream purification processes and ensuring the delivery of high-purity OLED materials or pharmaceutical intermediates.
How to Synthesize Polysubstituted Dihydropyrrole Efficiently
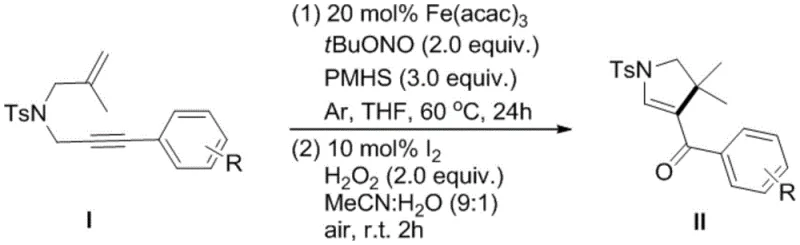
The practical implementation of this synthesis route is designed for scalability and ease of operation, making it highly attractive for contract development and manufacturing organizations (CDMOs). The process begins with the preparation of the reaction mixture in an inert atmosphere, ensuring that moisture and oxygen do not interfere with the sensitive radical intermediates generated during the iron-catalyzed step. Detailed standard operating procedures regarding stoichiometry, addition rates, and temperature ramps are critical for maximizing yield and reproducibility. For technical teams looking to adopt this methodology, understanding the nuances of the workup procedure—specifically the extraction and quenching steps—is vital for isolating the crude product with minimal loss. The comprehensive guide below outlines the standardized steps required to execute this transformation effectively in a pilot or production setting.
- React N-2-methallyl-N-3-arylpropargyl-p-toluenesulfonamide with Fe(acac)3, tert-butyl nitrite, and PMHS in THF at 60°C under inert atmosphere.
- Quench the reaction with water, extract with ethyl acetate, and concentrate to obtain the crude intermediate.
- Oxidize the crude product using I2 and H2O2 in an acetonitrile/water mixture at room temperature to yield the final polysubstituted dihydropyrrole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed technology offers transformative benefits that extend beyond simple chemical efficiency. The primary advantage lies in the drastic reduction of raw material costs associated with catalyst procurement. By replacing scarce and expensive noble metals with commodity-grade iron salts, manufacturers can insulate their production budgets from the volatility of the precious metals market. Additionally, the elimination of strong acids and bases reduces the costs associated with corrosion-resistant equipment and hazardous waste disposal. This shift towards benign reagents not only lowers operational expenditures but also simplifies regulatory compliance, as the environmental footprint of the manufacturing process is significantly diminished, facilitating smoother audits and faster time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The substitution of noble metal catalysts with iron salts results in substantial cost savings on a per-kilogram basis. Since iron is one of the most abundant elements on earth, its price stability ensures predictable long-term manufacturing costs. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility bills. The simplified purification process, necessitated by the absence of heavy metal contaminants, reduces the consumption of chromatography media and solvents, further driving down the total cost of ownership for the synthesis of complex polymer additives or agrochemical intermediates.
- Enhanced Supply Chain Reliability: Reliance on a single source for exotic catalysts poses a significant risk to supply continuity. By utilizing widely available reagents such as Fe(acac)3, tert-butyl nitrite, and PMHS, manufacturers can diversify their supplier base and mitigate the risk of shortages. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, enhancing overall process reliability. This resilience is critical for maintaining consistent delivery schedules for high-purity electronic chemicals, ensuring that downstream customers in the semiconductor or display industries experience no disruption in their own production lines.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is often hindered by safety concerns regarding exothermic reactions or toxic reagents. This iron-catalyzed method operates at moderate temperatures and uses relatively safe oxidants, making it inherently safer for large-scale reactors. The use of hydrogen peroxide in the oxidation step generates water as a byproduct, minimizing the generation of toxic waste streams. This alignment with green chemistry principles facilitates easier permitting and environmental compliance, allowing for rapid scale-up of complex specialty chemicals without the bureaucratic delays often associated with hazardous chemical processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the technology's capabilities. Whether you are evaluating the feasibility of this route for a specific API intermediate or assessing its potential for cost optimization in your current portfolio, these answers provide the necessary context for informed decision-making. We encourage technical teams to review these details closely to understand how this methodology can be integrated into existing manufacturing workflows.
Q: What are the advantages of using Iron catalysis over Noble Metals for Dihydropyrrole synthesis?
A: Iron catalysis offers significant cost reductions by eliminating expensive noble metals like Rhodium or Palladium. Furthermore, it operates under milder conditions without requiring strong acids or bases, simplifying downstream purification and waste treatment.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method utilizes readily available reagents such as Fe(acac)3 and PMHS, and operates at moderate temperatures (30-60°C). These factors, combined with the absence of sensitive transition metal complexes, make the process highly amenable to commercial scale-up.
Q: What is the purity profile of the resulting Pyrroline compounds?
A: The two-step process involving a specific I2/H2O2 oxidation system ensures high conversion and selectivity. The mild reaction conditions minimize side reactions, facilitating the isolation of high-purity products suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Dihydropyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic routes that balance performance with economic efficiency. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polysubstituted dihydropyrrole or related fine chemical intermediate meets the highest industry standards. We are committed to leveraging technologies like the iron-catalyzed cyclization described in CN111592481B to deliver superior value to our global partners in the pharmaceutical and agrochemical sectors.
We invite you to collaborate with us to explore the full potential of this cost-effective synthesis platform. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only reliable and compliant but also optimized for the future of sustainable chemical manufacturing.
