Advanced One-Step Synthesis of Pyrrolo[3,4-c]carbazole Derivatives for Commercial Pharmaceutical Production
Introduction to Next-Generation Carbazole Synthesis
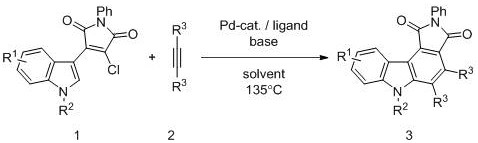
The pharmaceutical industry continuously seeks efficient pathways to construct complex heterocyclic scaffolds, particularly those exhibiting potent biological activities such as kinase inhibition. Patent CN110862396B discloses a groundbreaking synthetic methodology for producing pyrrolo[3,4-c]carbazole-1,3(2H,6H)-diketone compounds, a privileged structure found in numerous anticancer agents like midostaurin. This innovation represents a significant leap forward in process chemistry, utilizing a palladium-catalyzed cascade reaction that merges C-H bond activation with cyclization in a single operational step. By transforming readily available 3-chloro-4-indolylmaleimide compounds and alkynes directly into the target fused ring system, this technology addresses critical bottlenecks in the manufacturing of high-value oncology intermediates. The method operates under relatively mild thermal conditions compared to traditional photochemical approaches, offering a robust platform for industrial application.
![General reaction scheme showing the transformation of 3-chloro-4-indolylmaleimide and alkyne into pyrrolo[3,4-c]carbazole derivative](/insights/img/pyrrolo-carbazole-synthesis-pd-catalysis-pharma-supplier-20260304120911-01.png)
For R&D directors and process chemists, the ability to access these rigid planar structures through a direct annulation strategy is transformative. The patent details a protocol that achieves high yields while maintaining excellent functional group tolerance, allowing for the introduction of diverse substituents such as halogens, alkoxy groups, and alkyl chains on the indole core. This versatility is crucial for medicinal chemistry campaigns where structure-activity relationship (SAR) studies require rapid access to analog libraries. Furthermore, the reliance on a palladium catalyst system, specifically optimized with phosphine ligands, ensures that the reaction proceeds with high selectivity, minimizing the formation of difficult-to-remove impurities that often plague multi-step syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrrolo[3,4-c]carbazole skeleton has been fraught with synthetic challenges that hinder commercial viability. Traditional routes often rely on a Wittig condensation to generate a 2-vinyl indole derivative, followed by a Diels-Alder reaction with maleimide. This sequence is not only lengthy but also suffers from poor atom economy and the necessity for stoichiometric amounts of toxic oxidizing agents such as manganese dioxide (MnO2) or 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) to achieve aromatization. Alternatively, other reported methods involve a Mizoroki-Heck coupling between bromo-indolylmaleimides and olefins, necessitating subsequent oxidative cyclization under irradiation from a mercury lamp or catalysis by Pd/C. These photochemical steps introduce significant safety hazards and scalability issues, as mercury lamps are energy-intensive and difficult to manage in large-scale reactors, while the use of toxic reagents complicates waste disposal and regulatory compliance.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a direct cascade reaction that bypasses the need for pre-functionalized vinyl intermediates or harsh photochemical conditions. By employing a 3-chloro-4-indolylmaleimide starting material and reacting it directly with an alkyne in the presence of a palladium catalyst and base, the synthesis achieves the formation of two new carbon-carbon bonds and the final aromatic system in one pot. This telescoped process drastically reduces the number of unit operations, solvent usage, and overall processing time. The elimination of external oxidants like DDQ and the replacement of photochemical steps with thermal heating at 135°C significantly enhances the safety profile and environmental footprint of the manufacturing process. This streamlined workflow translates directly into lower production costs and a more reliable supply chain for downstream drug manufacturers.
Mechanistic Insights into Pd-Catalyzed C-H Activation Cyclization
The core of this technological advancement lies in the sophisticated palladium-catalyzed mechanism that drives the cascade transformation. The reaction initiates with the oxidative addition of the palladium catalyst to the carbon-chlorine bond of the 3-chloro-4-indolylmaleimide substrate. Following this activation, the alkyne component undergoes insertion into the palladium-carbon bond, setting the stage for the subsequent cyclization events. Crucially, the process involves an intramolecular C-H bond activation at the indole moiety, which closes the second ring to form the carbazole core. This C-H functionalization step is facilitated by the specific choice of ligands and bases, which modulate the electronic properties of the metal center to favor the desired reactivity over competing side reactions. The use of tricyclohexylphosphine tetrafluoroborate as a ligand is particularly effective in stabilizing the active catalytic species and promoting the reductive elimination step that releases the final product.
From an impurity control perspective, the mechanistic pathway offers distinct advantages over radical-based or photochemical alternatives. The concerted nature of the organometallic cycle minimizes the generation of random polymeric byproducts or regioisomers that are common in free-radical processes. The patent specifies that the reaction can be conducted in polar aprotic solvents like dimethyl sulfoxide (DMSO) or N,N-dimethylformamide (DMF), which solubilize the polar intermediates and facilitate heat transfer. The careful optimization of the base, such as diisopropylamine, ensures that the deprotonation steps required for C-H activation occur efficiently without degrading the sensitive maleimide functionality. This precise control over the reaction environment results in a cleaner crude reaction profile, simplifying the downstream purification requirements and ensuring high-quality output suitable for pharmaceutical applications.
How to Synthesize Pyrrolo[3,4-c]carbazole Derivatives Efficiently
To implement this synthesis effectively, operators must adhere to strict parameter controls regarding temperature and stoichiometry. The patent outlines a preferred embodiment where the reaction is conducted at 135°C for approximately 52 hours, although variations between 90°C and 150°C are permissible depending on the specific substrate reactivity. The molar ratio of the alkyne to the indole precursor is typically maintained between 1:1 and 1:3, with a 1:2 ratio often providing the optimal balance between conversion and cost efficiency. The catalyst loading is kept low, typically around 0.05 equivalents of palladium acetate relative to the substrate, demonstrating the high turnover number of the system. Detailed standardized synthesis steps see the guide below.
- Combine 3-chloro-4-indolylmaleimide, alkyne, palladium acetate, phosphine ligand, and base in an organic solvent like DMSO under nitrogen protection.
- Heat the reaction mixture to 135°C and stir magnetically for approximately 52 hours to ensure complete conversion via cascade cyclization.
- Quench with water, extract with ethyl acetate, dry over sodium sulfate, and purify the residue using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for specialized oxidants like DDQ, toxic heavy metal salts like manganese dioxide, and energy-intensive mercury lamps, the bill of materials becomes significantly leaner and less hazardous to handle. This reduction in hazardous reagents directly correlates to lower costs associated with safety equipment, waste treatment, and regulatory compliance. Furthermore, the use of commodity chemicals such as simple alkynes and substituted indoles ensures a stable and competitive supply base, reducing the risk of raw material shortages that can disrupt production schedules.
- Cost Reduction in Manufacturing: The one-pot nature of this cascade reaction eliminates multiple isolation and purification steps that are inherent in traditional multi-step syntheses. Each skipped step saves substantial amounts of solvent, labor, and time, leading to a significantly reduced cost of goods sold (COGS). Additionally, the high atom economy of the reaction means that a larger proportion of the starting mass ends up in the final product, minimizing waste disposal fees. The avoidance of expensive and toxic oxidizing agents further contributes to substantial cost savings, making the process economically attractive for large-scale commercial production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions allows for flexible manufacturing strategies. Since the process does not rely on specialized photochemical reactors or cryogenic conditions, it can be executed in standard stainless steel reactors available in most contract manufacturing organizations (CMOs). This compatibility with existing infrastructure reduces the lead time for technology transfer and scale-up. Moreover, the broad substrate scope means that a single manufacturing line can be adapted to produce a variety of analogs by simply switching the alkyne or indole starting material, providing agility in responding to market demands for different drug candidates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to pilot plant is straightforward due to the absence of exothermic hazards associated with strong oxidants or the safety risks of UV irradiation. The reaction operates at atmospheric pressure and moderate temperatures, simplifying engineering controls. From an environmental standpoint, the reduction in toxic waste streams aligns with green chemistry principles, facilitating easier permitting and reducing the environmental liability of the manufacturing site. The use of recyclable solvents and the potential for catalyst recovery further enhance the sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical teams evaluating this route for their specific projects. Understanding these nuances is essential for successful process adoption.
Q: What are the key advantages of this Pd-catalyzed method over traditional Wittig or Heck reactions?
A: This method eliminates the need for toxic oxidants like DDQ or MnO2 and avoids complex multi-step sequences involving mercury lamp irradiation, resulting in higher atom economy and simpler operation.
Q: What is the substrate scope for the alkyne component in this synthesis?
A: The process demonstrates excellent universality, accommodating both symmetrical diarylalkynes (like diphenylacetylene) and dialkylalkynes (like 4-octyne) to produce diverse substituted carbazole derivatives.
Q: How is the reaction purity maintained during scale-up?
A: High purity is achieved through the use of specific phosphine ligands like tricyclohexylphosphine tetrafluoroborate which minimize side reactions, followed by standard silica gel chromatography purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolo[3,4-c]carbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for complex pharmaceutical intermediates. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN110862396B and is fully equipped to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are designed to handle palladium-catalyzed reactions safely and efficiently, with rigorous QC labs dedicated to maintaining stringent purity specifications for every batch produced.
We invite you to collaborate with us to leverage this advanced synthesis for your drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to discuss your project needs, obtain specific COA data for related intermediates, and receive comprehensive route feasibility assessments that will accelerate your path to market.
