Advanced One-Step Synthesis of Pyrrolo Carbazole Diones for Commercial Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic pathways for privileged scaffolds like carbazole derivatives, which are pivotal in developing kinase inhibitors such as Midostaurin. Patent CN110862396A introduces a groundbreaking methodology for synthesizing pyrrolo[3,4-c]carbazole-1,3(2H,6H)-dione compounds, addressing critical bottlenecks in existing manufacturing protocols. This innovation leverages a palladium-catalyzed tandem reaction coupled with C-H bond activation, enabling the direct construction of the complex fused ring system in a single operational step. By eliminating the need for multiple isolation stages and hazardous oxidants, this technology offers a streamlined route that aligns perfectly with modern green chemistry principles and industrial efficiency standards. For R&D teams focused on oncology therapeutics, this represents a significant leap forward in accessing high-value intermediates with improved speed and reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrrolo[3,4-c]carbazole core has relied on cumbersome multi-step sequences that pose substantial challenges for process chemists and supply chain managers alike. Traditional routes often involve Wittig condensation to generate vinyl indole derivatives, followed by Diels-Alder cycloadditions and subsequent aromatization using stoichiometric amounts of toxic oxidants like manganese dioxide or DDQ. These legacy processes are characterized by poor atom economy, generating significant quantities of chemical waste that require costly disposal and environmental mitigation strategies. Furthermore, the involvement of multiple discrete reaction steps inherently increases the risk of yield loss at each stage, compounding material costs and extending the overall production timeline. The reliance on hazardous reagents also introduces safety concerns that complicate regulatory compliance and increase the operational burden on manufacturing facilities.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a sophisticated palladium-catalyzed system to achieve the target scaffold through a direct tandem cyclization process. This novel approach employs 3-chloro-4-indolylmaleimide compounds and alkynes as readily available starting materials, reacting them in the presence of a phosphine ligand and base under nitrogen protection. 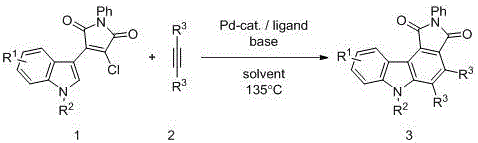 The reaction proceeds efficiently at elevated temperatures, typically around 135°C, facilitating the simultaneous formation of multiple bonds without the need for intermediate isolation. This convergence of steps not only drastically simplifies the operational workflow but also enhances the overall mass balance of the process. By avoiding the use of stoichiometric oxidants and reducing the number of unit operations, this method offers a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing while maintaining high standards of chemical safety and environmental stewardship.
The reaction proceeds efficiently at elevated temperatures, typically around 135°C, facilitating the simultaneous formation of multiple bonds without the need for intermediate isolation. This convergence of steps not only drastically simplifies the operational workflow but also enhances the overall mass balance of the process. By avoiding the use of stoichiometric oxidants and reducing the number of unit operations, this method offers a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing while maintaining high standards of chemical safety and environmental stewardship.
Mechanistic Insights into Pd-Catalyzed Tandem Cyclization
The core of this technological advancement lies in the intricate mechanistic pathway driven by the palladium catalyst, which orchestrates a series of bond-forming events with remarkable precision. The reaction initiates with the oxidative addition of the palladium species to the carbon-chlorine bond of the indolylmaleimide substrate, generating a reactive organopalladium intermediate. This species then undergoes insertion into the alkyne triple bond, followed by a crucial intramolecular C-H activation step that closes the carbazole ring system.  The presence of the specific phosphine ligand is critical in stabilizing the active catalytic species and promoting the reductive elimination step that releases the final product and regenerates the catalyst. This catalytic cycle ensures that only substoichiometric amounts of the expensive metal are required, contributing to the economic viability of the process. Understanding this mechanism allows process engineers to fine-tune reaction parameters such as temperature and ligand loading to maximize turnover numbers and minimize catalyst deactivation.
The presence of the specific phosphine ligand is critical in stabilizing the active catalytic species and promoting the reductive elimination step that releases the final product and regenerates the catalyst. This catalytic cycle ensures that only substoichiometric amounts of the expensive metal are required, contributing to the economic viability of the process. Understanding this mechanism allows process engineers to fine-tune reaction parameters such as temperature and ligand loading to maximize turnover numbers and minimize catalyst deactivation.
Beyond mere efficiency, this mechanism provides superior control over the impurity profile, a key concern for R&D directors overseeing quality assurance. The high regioselectivity of the C-H activation step ensures that the cyclization occurs exclusively at the desired position, preventing the formation of structural isomers that are difficult to separate. Additionally, the mild nature of the catalytic conditions compared to harsh oxidative aromatization preserves sensitive functional groups on the substrate, allowing for greater diversity in the final library of compounds. This selectivity translates directly into a cleaner crude reaction mixture, reducing the load on downstream purification units like chromatography columns. Consequently, the final product exhibits high purity specifications with minimal effort, ensuring that the material meets the stringent requirements for subsequent biological evaluation and clinical development.
How to Synthesize Pyrrolo Carbazole Dione Efficiently
Implementing this synthesis route requires careful attention to reaction conditions to replicate the high yields reported in the patent literature. The process begins with the precise weighing of the 3-chloro-4-indolylmaleimide substrate and the selected alkyne, ensuring a molar ratio that favors complete consumption of the limiting reagent. These solids are suspended in a polar aprotic solvent such as dimethyl sulfoxide, which has been identified as optimal for solubilizing the reactants and stabilizing the transition states. The addition of the palladium catalyst, phosphine ligand, and organic base must be performed under an inert atmosphere to prevent oxidation of the sensitive catalytic species. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility across different laboratory scales.
- Combine 3-chloro-4-indolylmaleimide, alkyne, palladium catalyst, phosphine ligand, and base in an organic solvent under nitrogen protection.
- Heat the reaction mixture to 135°C and stir magnetically for 40 to 72 hours to ensure complete cyclization.
- Quench with water, extract with ethyl acetate, dry over sodium sulfate, and purify the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond the laboratory bench into the realm of strategic sourcing and operational logistics. The elimination of multiple synthetic steps inherently reduces the consumption of raw materials, solvents, and energy, leading to substantial cost savings in the overall manufacturing budget. By consolidating what was previously a multi-day sequence into a single pot reaction, facilities can significantly increase their throughput capacity without requiring additional capital investment in reactor hardware. This efficiency gain allows suppliers to respond more agilely to market demand fluctuations, ensuring a steady flow of critical intermediates to downstream API manufacturers. The robustness of the process also mitigates the risk of batch failures, enhancing supply chain reliability and reducing the need for safety stock inventories.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the removal of expensive and toxic oxidizing agents such as DDQ and manganese dioxide from the process bill of materials. These reagents not only carry a high purchase price but also generate hazardous waste streams that incur significant disposal fees and regulatory compliance costs. By replacing these stoichiometric reagents with a catalytic system, the process achieves a drastic simplification of the waste profile, allowing for more efficient resource utilization. Furthermore, the high yields obtained reduce the amount of starting material required per kilogram of final product, directly lowering the variable cost of goods sold. This structural cost advantage provides a competitive edge in pricing negotiations while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of sourcing specialized reagents required for niche synthetic transformations. This new method relies on commodity chemicals such as alkynes and substituted indoles, which are widely available from multiple global vendors, thereby reducing single-source dependency risks. The simplified workflow also shortens the manufacturing lead time, as there are fewer intermediate quality control checkpoints and isolation steps that can cause bottlenecks. This agility enables the supply chain to recover more quickly from disruptions and adapt to urgent procurement requests from pharmaceutical clients. The consistency of the reaction performance ensures that delivery schedules can be met with high confidence, fostering stronger long-term partnerships between suppliers and buyers.
- Scalability and Environmental Compliance: Scaling chemical processes from the gram scale to metric tons often reveals hidden complexities, but this catalytic method is designed with scalability in mind. The reaction conditions utilize standard heating and stirring equipment found in most multipurpose chemical plants, avoiding the need for specialized high-pressure or cryogenic infrastructure. From an environmental perspective, the reduction in solvent usage and the elimination of heavy metal oxidants align with increasingly strict global environmental regulations regarding emissions and effluent treatment. This compliance reduces the administrative burden on EHS teams and minimizes the risk of regulatory penalties. The ability to scale up complex pharmaceutical intermediates safely and sustainably makes this technology a future-proof choice for long-term production planning.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this route for potential adoption. Understanding these nuances is essential for making informed decisions about process integration and supplier selection. The information below serves as a foundational reference for further technical discussions between our engineering teams and your procurement specialists.
Q: What are the advantages of this Pd-catalyzed method over conventional Wittig reactions?
A: Unlike conventional multi-step Wittig and Diels-Alder sequences that suffer from poor atom economy and toxic reagents, this novel approach utilizes a one-step tandem C-H activation cyclization. This significantly reduces waste generation and simplifies the purification process, leading to higher overall efficiency.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process demonstrates excellent substrate universality and operates under relatively standard heating conditions (135°C) without requiring specialized high-pressure equipment. The use of common organic solvents and straightforward workup procedures makes it highly amenable to commercial scale-up of complex pharmaceutical intermediates.
Q: How does this method impact impurity profiles in the final API intermediate?
A: The high selectivity of the palladium-catalyzed cyclization minimizes the formation of side products often associated with harsh oxidation steps found in older methods. This results in a cleaner crude reaction mixture, facilitating easier purification and ensuring high-purity carbazole derivatives suitable for sensitive drug applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolo Carbazole Dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has thoroughly evaluated the patent CN110862396A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to translate this laboratory innovation into industrial reality. We are committed to delivering high-purity carbazole derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical instrumentation. Our facility is equipped to handle the specific solvent systems and thermal requirements of this palladium-catalyzed process, ensuring consistent quality across every batch we produce for our valued clients.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next drug development project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain. Let us be your partner in transforming complex chemical challenges into reliable commercial solutions, ensuring your pipeline moves forward without interruption.
