Scalable Synthesis of 1-(3-Methoxypropyl)-4-Piperidinamine for High-Volume API Production
The pharmaceutical industry constantly seeks robust pathways for synthesizing complex heterocyclic amines, particularly those serving as critical building blocks for gastrointestinal therapeutics. Patent CN102898356A introduces a significant advancement in the preparation of 1-(3-methoxypropyl)-4-piperidinamine, a pivotal intermediate for the synthesis of Prucalopride, a selective 5-HT4 receptor agonist. This technology addresses the growing demand for high-purity intermediates by offering a streamlined, two-step synthetic route that bypasses traditional complexities. By leveraging direct N-alkylation followed by catalytic reductive amination, the process ensures high efficiency and operational simplicity. For R&D directors and procurement specialists, understanding this methodology is crucial for securing a stable supply chain for next-generation constipation treatments. The following analysis dissects the technical merits and commercial viability of this patented approach.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted piperidine amines has been plagued by multi-step sequences requiring rigorous protection and deprotection strategies. Conventional routes often involve the use of expensive reagents or harsh conditions that generate significant hazardous waste, complicating downstream purification. Many prior art methods rely on less efficient coupling reactions that suffer from poor regioselectivity, leading to difficult-to-remove impurities that compromise the final API quality. Furthermore, the reliance on specialized starting materials that are not readily available on the bulk chemical market creates bottlenecks in the supply chain. These factors collectively drive up manufacturing costs and extend lead times, making it challenging for producers to meet the rigorous demands of global regulatory bodies. The lack of a direct, scalable route has long been a pain point for manufacturers aiming to produce Prucalopride intermediates competitively.
The Novel Approach
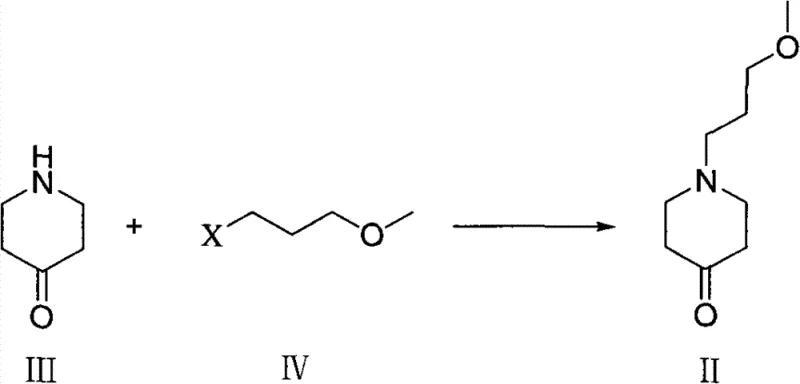
The methodology disclosed in the patent revolutionizes this landscape by introducing a direct alkylation strategy followed by a highly efficient hydrogenation step. As illustrated in the reaction scheme below, the process begins with the reaction of 4-piperidone (Formula III) with a 3-halo-propyl methyl ether derivative (Formula IV) in the presence of a base. This straightforward N-alkylation yields the key ketone intermediate (Formula II) with high fidelity. Unlike older methods, this approach utilizes common, inexpensive solvents like acetonitrile and bases such as potassium carbonate, significantly lowering the barrier to entry for production. The subsequent conversion to the amine is achieved through reductive amination in an ammonia-rich environment, eliminating the need for complex nitrogen source introductions. This streamlined workflow not only reduces the number of unit operations but also enhances the overall atom economy of the synthesis.
Mechanistic Insights into Catalytic Reductive Amination
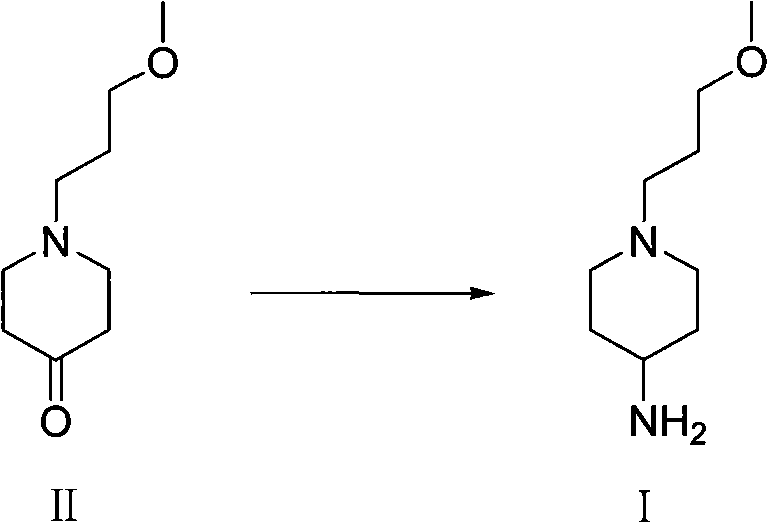
The core of this synthetic innovation lies in the reductive amination step, where the ketone intermediate (Formula II) is converted into the target amine (Formula I). This transformation occurs in an organic solution saturated with ammonia gas, under the influence of hydrogen and a hydrogenation catalyst. The mechanism involves the initial formation of an imine or iminium ion intermediate between the ketone carbonyl group and ammonia, which is subsequently reduced by hydrogen on the catalyst surface. The patent highlights the versatility of catalysts, noting that both Raney Nickel and Palladium on Carbon (Pd/C) can be effectively employed. This flexibility allows manufacturers to optimize based on cost and availability, with Pd/C often offering superior activity at lower pressures. The reaction conditions are remarkably broad, tolerating temperatures from 0°C to 160°C and hydrogen pressures up to 20 MPa, providing substantial operational window for process engineers to fine-tune kinetics and safety parameters.
Impurity control is intrinsically built into this mechanistic pathway through the use of excess ammonia and optimized solvent systems. By maintaining a high concentration of ammonia in solvents like methanol or ethanol, the equilibrium is driven towards the desired primary amine, minimizing the formation of secondary or tertiary amine byproducts that typically arise from over-alkylation. The choice of solvent plays a critical role here; polar protic solvents facilitate the solubility of ammonia and stabilize the transition states involved in the reduction. Furthermore, the post-reaction workup involving salt formation with acids like hydrochloric acid allows for the crystallization of the product, effectively purifying it from non-basic organic impurities. This dual mechanism of kinetic control during reaction and thermodynamic control during isolation ensures that the final product meets the stringent purity specifications required for pharmaceutical intermediates.
How to Synthesize 1-(3-Methoxypropyl)-4-Piperidinamine Efficiently
Implementing this synthesis requires precise control over reaction stoichiometry and environmental conditions to maximize yield and safety. The process is divided into two distinct phases: the initial alkylation to form the ketone precursor and the subsequent hydrogenation to generate the amine. Operators must ensure that the alkylation step is driven to completion, typically monitored by HPLC or TLC, before proceeding to the high-pressure hydrogenation stage. The use of standardized equipment such as high-pressure autoclaves is essential for the second step, along with strict adherence to safety protocols regarding hydrogen handling. Detailed standard operating procedures regarding catalyst loading, ammonia saturation levels, and temperature ramping are critical for reproducibility. For a comprehensive guide on the specific experimental parameters and workup procedures, please refer to the standardized synthesis steps outlined below.
- Perform N-alkylation of 4-piperidone hydrochloride with 3-halo-propyl methyl ether using a base like potassium carbonate in acetonitrile to form the ketone intermediate.
- Conduct reductive amination of the resulting ketone in an organic ammonia solution using hydrogen gas and a catalyst such as Pd/C or Raney Nickel.
- Isolate the final amine product either as a free base or convert it into a stable salt form like the hydrochloride through acid treatment.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented route offers profound advantages that directly impact the bottom line and supply chain resilience. The primary driver for cost reduction is the utilization of commodity chemicals as starting materials. 4-Piperidone and simple alkyl halides are produced in massive volumes globally, ensuring price stability and preventing supply shocks that often plague specialty chemical markets. By eliminating the need for exotic reagents or complex chiral catalysts in the early stages, the overall material cost is drastically simplified. Additionally, the ability to use common solvents like acetonitrile and methanol means that solvent recovery and recycling systems can be easily integrated into existing infrastructure, further driving down operational expenditures. This economic efficiency makes the process highly attractive for large-scale manufacturing where margin compression is a constant challenge.
- Cost Reduction in Manufacturing: The elimination of expensive protecting groups and the use of heterogeneous catalysts like Pd/C which can potentially be recovered and reused, leads to substantial cost savings. The process avoids the generation of heavy metal waste streams associated with stoichiometric reducing agents, thereby reducing waste disposal costs. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, improving the overall mass balance and yield efficiency. These factors combine to create a lean manufacturing process that is economically superior to legacy synthetic routes.
- Enhanced Supply Chain Reliability: Sourcing risk is significantly mitigated because all key reagents are widely available from multiple global suppliers. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in raw material quality or environmental variables. This reliability ensures consistent output volumes, allowing downstream API manufacturers to plan their production schedules with greater confidence. The scalability of the process from kilogram to metric ton scales without significant re-engineering further strengthens the supply chain continuity for long-term contracts.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations familiar to chemical engineers. The use of hydrogenation, while requiring safety measures, is a well-established technology in the fine chemical industry with mature safety protocols. Environmentally, the route generates fewer byproducts and utilizes solvents that are easier to treat or recycle compared to chlorinated alternatives often found in older methodologies. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on process capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this technology for their specific production needs. The answers reflect the practical realities of scaling this chemistry in an industrial setting.
Q: What catalysts are suitable for the reductive amination step?
A: The patent specifies that conventional hydrogenation catalysts such as Raney Nickel and Palladium on Carbon (Pd/C) are highly effective. The choice depends on specific pressure and temperature constraints, with Pd/C often preferred for milder conditions.
Q: How is the purity of the intermediate controlled during synthesis?
A: Purity is managed through careful selection of solvents like acetonitrile or DMF during alkylation and by optimizing the ammonia concentration during hydrogenation. Post-reaction salt formation and subsequent neutralization further refine the product quality.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method utilizes commercially available raw materials and standard equipment like high-pressure hydrogenation kettles. The operational simplicity and robust reaction conditions make it ideal for scaling up to metric ton levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(3-Methoxypropyl)-4-Piperidinamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-(3-methoxypropyl)-4-piperidinamine meets the highest international standards. Our commitment to excellence extends beyond mere compliance; we actively collaborate with clients to optimize processes for maximum efficiency and cost-effectiveness.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific application. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements. Let us be your partner in navigating the complexities of pharmaceutical intermediate sourcing and manufacturing.
