Advanced Salt-Formation Purification Strategy for Commercial Scale Rebamipide Production
Introduction to Advanced Rebamipide Purification Technologies
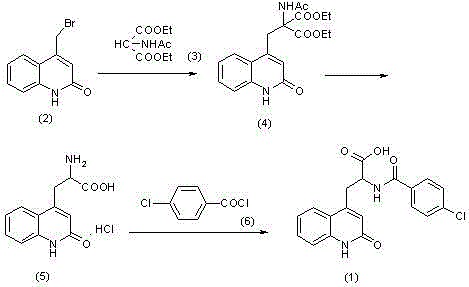
The pharmaceutical industry constantly demands higher purity standards for Active Pharmaceutical Ingredients (APIs) to ensure patient safety and regulatory compliance. Patent CN102174015B introduces a groundbreaking refining method for Rebamipide, a quinolone compound widely used for treating gastric mucosal injuries and gastritis. Traditional synthesis routes, such as the one depicted in the reaction scheme below, often yield crude products contaminated with specific structural analogs and byproducts that are notoriously difficult to separate. This novel technology addresses these challenges by employing a strategic salt-formation and recrystallization protocol that fundamentally alters the physicochemical properties of the target molecule, enabling the removal of persistent impurities that conventional methods fail to eliminate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Rebamipide has relied heavily on direct recrystallization from various solvent systems or simple pH adjustments. However, these conventional techniques suffer from significant inefficiencies when dealing with the complex impurity profile generated during the acylation and cyclization steps of the synthesis. As illustrated in the synthetic pathway, intermediates such as unreacted amines or partially hydrolyzed esters can co-crystallize with the final product, leading to purity levels that often stagnate below the stringent requirements for modern pharmaceutical applications. Furthermore, traditional methods frequently necessitate the use of high-boiling point solvents to achieve adequate solubility, which complicates the drying process, increases energy consumption, and poses risks of residual solvent contamination in the final API, thereby creating bottlenecks in both quality control and production throughput.
The Novel Approach
In stark contrast, the methodology disclosed in CN102174015B leverages the acid-base properties of the Rebamipide molecule to create a transient salt intermediate, typically using alkali metal hydroxides like potassium hydroxide or sodium hydroxide. This chemical transformation converts the neutral organic acid into a highly soluble ionic species, which exhibits markedly different crystallization kinetics compared to its neutral counterpart and the associated neutral impurities. By recrystallizing this salt form from optimized solvent mixtures such as isopropanol or ethyl acetate blends, the process achieves a highly effective fractionation where the pure salt precipitates while impurities remain dissolved in the mother liquor. Subsequent acidification regenerates the free acid form of Rebamipide with exceptional purity, effectively breaking the cycle of impurity entrapment that plagues direct recrystallization techniques.
Mechanistic Insights into Salt-Formation Purification
The core mechanism driving this purification success lies in the differential solubility and lattice energy of the Rebamipide salt versus the impurity molecules. When the crude Rebamipide reacts with an alkali, the carboxylic acid moiety is deprotonated to form a carboxylate anion, which interacts strongly with the counter-cation (e.g., K+ or Na+). This ionic interaction enhances solubility in polar protic solvents like methanol and water, allowing for a homogeneous solution where activated carbon can effectively adsorb colored polymeric byproducts. Upon cooling or anti-solvent addition, the Rebamipide salt crystallizes in a highly ordered lattice that energetically excludes structurally dissimilar impurities, a phenomenon known as lattice rejection. This step is critical because it separates the target molecule from non-ionic or differently charged contaminants that would otherwise co-precipitate in a neutral crystallization environment.
Furthermore, the final acidification step is meticulously controlled to ensure the regeneration of the free acid without re-introducing impurities. By adjusting the pH to a specific range between 2 and 5 using dilute mineral acids, the carboxylate anion is reprotonated, causing the Rebamipide to precipitate out of the aqueous-alcoholic solution. The choice of solvent ratios, particularly the volume-to-mass ratio of lower alcohols to water, is optimized to maximize the yield while maintaining the exclusion of water-soluble salts and residual organic byproducts. This precise control over the protonation equilibrium ensures that the final crystal habit is uniform and free from occluded mother liquor, resulting in a product that meets the rigorous HPLC purity specifications of greater than 99.8% with total impurities suppressed below 0.20%.
How to Synthesize High Purity Rebamipide Efficiently
The implementation of this refining process requires a systematic approach to reaction conditions and solvent management to ensure reproducibility at scale. The procedure begins with the conversion of the crude material into a stable salt form, followed by a rigorous recrystallization cycle, and concludes with a controlled precipitation of the final API. Each stage is designed to incrementally increase purity while managing the physical form of the solid to facilitate filtration and drying. For detailed operational parameters, including specific temperature ramps, stirring rates, and solvent volumes required for GMP-compliant manufacturing, please refer to the standardized synthesis guide provided below.
- React crude Rebamipide with an alkali (such as potassium hydroxide) in a solvent like methanol to form a crude Rebamipide salt, followed by activated carbon decolorization.
- Recrystallize the crude salt using isopropanol or a mixed solvent system to obtain refined Rebamipide salt with significantly reduced impurity levels.
- Dissolve the refined salt in a lower alcohol-water mixture and adjust the pH to 2-5 using acid to precipitate high-purity Rebamipide crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this refining technology offers substantial benefits that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. By shifting the purification paradigm from complex chromatographic separations or multi-step solvent swaps to a streamlined salt-recrystallization workflow, the process significantly reduces the operational complexity and equipment footprint required for production. The elimination of high-boiling solvents not only lowers the energy costs associated with solvent recovery and product drying but also accelerates the batch cycle time, allowing for increased throughput without the need for additional capital investment in specialized vacuum or distillation infrastructure.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substitution of expensive or hazardous solvents with commodity chemicals such as methanol, ethanol, and isopropanol, which are readily available in the global market at stable prices. Additionally, the high efficiency of the salt recrystallization step minimizes product loss, leading to improved overall yields compared to traditional purification methods that often sacrifice yield for purity. The removal of the need for extensive chromatographic purification columns further reduces consumable costs and waste disposal fees, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on common alkali reagents like potassium hydroxide and sodium hydroxide ensures that the supply chain is not vulnerable to the shortages of exotic catalysts or specialized reagents. This robustness is critical for maintaining continuous production schedules, especially in a volatile global market where raw material availability can fluctuate. The simplicity of the process also means that it can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without requiring extensive requalification of unique equipment, thereby diversifying supply sources and mitigating risk.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory bench scales to multi-ton commercial production due to the use of standard unit operations like dissolution, filtration, and crystallization. The reduced solvent load and the ability to recycle low-boiling solvents align with green chemistry principles, lowering the environmental footprint of the manufacturing process. This compliance with environmental regulations reduces the burden of waste treatment and facilitates smoother regulatory approvals in jurisdictions with strict environmental oversight, ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of adopting this refining method, we have compiled answers to common inquiries regarding the process mechanics and quality outcomes. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of how this technology resolves specific purification bottlenecks. Understanding these details is essential for R&D directors and process engineers planning the scale-up of Rebamipide production.
Q: What is the primary advantage of the salt-based purification method for Rebamipide?
A: The primary advantage is the ability to remove structurally similar impurities that persist during direct recrystallization. By converting Rebamipide into a salt, its solubility profile changes drastically, allowing for selective crystallization that leaves impurities in the mother liquor, achieving purity levels above 99.8%.
Q: Does this refining process require expensive or high-boiling solvents?
A: No, the process is designed to avoid high-boiling solvents which are difficult to remove and energy-intensive to recover. It utilizes common, low-cost solvents such as methanol, ethanol, isopropanol, and ethyl acetate, which simplifies the downstream solvent recovery and reduces overall operational costs.
Q: How does this method address the issue of product color and aesthetic quality?
A: The method incorporates activated carbon decolorization at multiple stages, specifically during the initial salt formation, the salt recrystallization, and the final acidification step. This multi-stage filtration ensures that colored impurities and organic byproducts are effectively adsorbed, resulting in a white crystalline product with excellent aesthetic appearance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rebamipide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced purification technologies like the salt-formation method are fully realized in practice. We operate with stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Rebamipide meets the highest global pharmacopoeia standards, guaranteeing consistency and safety for your downstream formulations.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior refining technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing processes can deliver high-purity Rebamipide with unmatched reliability and efficiency.
