Advanced Manufacturing of Rebamipide Intermediates via Optimized Glycine Ester Route
The pharmaceutical landscape for gastric mucosal protective agents continues to evolve, driven by the demand for more efficient and cost-effective manufacturing processes. A pivotal development in this sector is detailed in patent CN108069901B, which discloses a novel synthesis process for Rebamipide, a critical active pharmaceutical ingredient (API). This technology represents a significant departure from traditional methodologies by utilizing glycine methyl ester as a foundational building block. By shifting the synthetic strategy away from expensive malonate derivatives, this innovation addresses long-standing challenges regarding raw material availability and process complexity. For R&D directors and procurement specialists alike, understanding this pathway is essential for optimizing supply chains and reducing the overall cost of goods sold (COGS) for this high-value therapeutic agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Rebamipide has relied on routes that are both economically and technically burdensome. As illustrated in the prior art, such as the method disclosed in Japanese patent JP2008-143794a, the synthesis typically begins with the bromination and ring-closure of bisacetanilide to form bromomethyl quinolone. This intermediate is then reacted with diethyl acetamidomalonate, a reagent that is not only costly but also necessitates a multi-step sequence to install the amino acid side chain. The subsequent removal of acetyl and carboxyl groups under acidic conditions introduces further complications, often leading to lower overall yields and increased waste generation. Furthermore, the decarboxylation step in these conventional routes is prone to generating significant amounts of gas, which can cause foaming and incomplete reactions, thereby complicating reactor design and process control on a large scale.
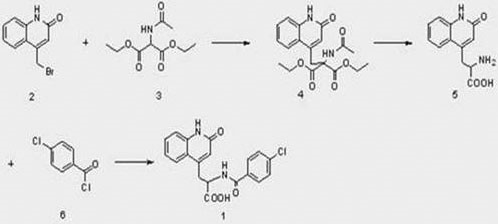
The Novel Approach
In stark contrast, the methodology presented in CN108069901B offers a streamlined alternative that fundamentally simplifies the molecular construction. By initiating the synthesis with glycine methyl ester, the process bypasses the need for complex malonate chemistry entirely. The new route involves a direct amidation followed by a highly efficient chlorination to create a reactive chloroimine intermediate. This intermediate then undergoes a substitution reaction with the quinolone core, followed by a straightforward hydrolysis to yield the final product. This approach not only reduces the number of unit operations but also utilizes starting materials that are commodity chemicals, thereby securing a more stable and affordable supply chain. The elimination of the problematic decarboxylation step ensures a smoother reaction profile, making this method exceptionally suitable for reliable rebamipide intermediate supplier operations aiming for high-volume production.
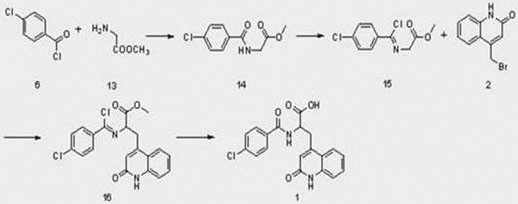
Mechanistic Insights into Amidation and Chlorination Cascade
The core of this technological breakthrough lies in the precise manipulation of the glycine backbone through a controlled amidation and chlorination sequence. In the first stage, glycine methyl ester hydrochloride reacts with p-chlorobenzoyl chloride under alkaline conditions, typically using triethylamine or sodium bicarbonate in solvents like dichloromethane. This step is critical for establishing the amide bond with high fidelity, achieving yields as high as 95% when optimized at temperatures between 0°C and 30°C. The resulting p-chlorobenzoyl glycine methyl ester (compound 14) serves as the precursor for the subsequent activation. The mechanistic elegance continues in the chlorination step, where phosphorus pentachloride acts as a potent chlorinating agent in toluene at elevated temperatures (80-110°C). This transformation converts the amide carbonyl into a highly electrophilic chloroimine species (compound 15), which is primed for nucleophilic attack.
From an impurity control perspective, this route offers distinct advantages over the malonate-based alternatives. The avoidance of harsh acidic decarboxylation conditions minimizes the formation of degradation byproducts that are often difficult to separate from the final API. The substitution reaction between the chloroimine intermediate and bromomethyl quinolone proceeds cleanly under basic conditions, with sodium hydroxide in methanol proving to be the optimal system, delivering yields around 88%. Finally, the hydrolysis of the ester moiety to the free acid is conducted under mild conditions, preserving the integrity of the quinolone ring and the newly formed peptide bond. This careful orchestration of reaction conditions ensures that the impurity profile remains manageable, facilitating easier purification and higher final purity for the commercial product.
How to Synthesize Rebamipide Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters outlined in the patent to ensure maximum efficiency and safety. The process is divided into four distinct operational stages: the initial amidation to form the protected glycine derivative, the activation via chlorination, the coupling with the quinolone scaffold, and the final hydrolysis. Each step has been rigorously tested with various solvents and bases to identify the most robust conditions for industrial application. For process chemists looking to adopt this technology, it is crucial to note that the choice of solvent in the chlorination step significantly impacts the yield, with toluene outperforming dichloromethane. Detailed standard operating procedures and specific stoichiometric ratios are essential for replicating the high success rates reported in the intellectual property.
- Perform amidation reaction on methyl glycinate with p-chlorobenzoyl chloride under alkaline conditions to obtain compound 14.
- Carry out chlorination reaction on compound 14 using phosphorus pentachloride to generate the chloroimine intermediate (compound 15).
- Execute substitution reaction between the chloroimine intermediate and bromomethyl quinolone (compound 2) under basic conditions to form compound 16.
- Hydrolyze compound 16 under acidic or basic conditions to finalize the synthesis of Rebamipide (compound 1).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis pathway translates into tangible strategic benefits that extend beyond simple chemistry. The primary driver of value is the substantial reduction in raw material costs achieved by substituting expensive specialty reagents with commodity chemicals. By eliminating the dependency on diethyl acetamidomalonate, manufacturers can insulate their production costs from the volatility associated with complex fine chemical intermediates. Furthermore, the simplified process flow reduces the requirement for specialized equipment capable of handling vigorous gas evolution, thereby lowering capital expenditure requirements for new production lines. This operational simplicity also contributes to enhanced supply chain reliability, as the risk of batch failure due to process upsets is significantly mitigated.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling, primarily driven by the replacement of high-cost starting materials with inexpensive glycine methyl ester. The elimination of multiple protection and deprotection steps inherent in the malonate route results in a drastic simplification of the manufacturing workflow. This reduction in synthetic steps directly correlates to lower labor costs, reduced solvent consumption, and decreased waste disposal fees. Consequently, the overall cost of production is significantly lowered, providing a competitive edge in the global market for generic pharmaceuticals without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on niche intermediates that have limited suppliers. This new method leverages glycine methyl ester and p-chlorobenzoyl chloride, which are widely produced bulk chemicals with established global supply networks. This shift ensures that production schedules are less likely to be disrupted by raw material shortages. Additionally, the robustness of the reaction conditions means that manufacturing can be scaled up or down with greater flexibility, allowing suppliers to respond more agilely to fluctuating market demands for Rebamipide.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, the new process offers a cleaner profile that aligns with modern green chemistry principles. The avoidance of violent decarboxylation reactions reduces the generation of gaseous emissions and the associated safety risks in the plant. Moreover, the higher yields achieved at each step mean that less raw material is wasted, leading to a reduced environmental footprint per kilogram of product. This efficiency makes the process easier to scale from pilot plant to full commercial production, ensuring that regulatory compliance regarding waste and emissions is more easily maintained.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is vital for stakeholders evaluating the feasibility of technology transfer or licensing agreements.
Q: What are the primary cost advantages of this new Rebamipide synthesis route?
A: The process utilizes glycine methyl ester as a starting material, which is significantly cheaper and more readily available than the diethyl acetamidomalonate used in conventional methods. Additionally, the streamlined pathway eliminates the need for complex acetyl removal and decarboxylation steps, drastically reducing raw material consumption and processing time.
Q: How does the new process improve industrial scalability compared to prior art?
A: Unlike traditional methods that suffer from incomplete reactions due to gas generation during decarboxylation, this novel route employs mild reaction conditions. The substitution and hydrolysis steps are optimized to avoid violent bubbling, ensuring consistent reaction kinetics that are far easier to control and scale up for commercial production.
Q: What yields can be expected from the key intermediate steps?
A: The patent data indicates high efficiency across the board. The initial amidation step achieves yields up to 95%, while the critical chlorination step reaches 91% in toluene. The final hydrolysis step consistently delivers yields above 92%, ensuring a robust overall process mass intensity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rebamipide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of optimized synthetic routes like the one described in CN108069901B for the production of high-quality gastric therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest international standards. We understand that in the pharmaceutical industry, consistency is key, and our advanced manufacturing facilities are designed to deliver exactly that.
We invite potential partners to engage with our technical team to explore how this cost-effective synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of scientific rigor and commercial viability.
