Scalable One-Pot Synthesis of 1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
The landscape of heterocyclic chemistry is continually evolving, driven by the demand for safer, more efficient synthetic routes to pharmacologically active scaffolds. Among these, the 1,2,3-triazole ring stands out as a privileged structure in medicinal chemistry, renowned for its metabolic stability and ability to act as a bioisostere for amide bonds. A significant breakthrough in this domain is detailed in Chinese Patent CN109776435B, which discloses a novel one-pot method for preparing 1,2,3-triazoles using dibromovinylbenzene derivatives. This technology represents a paradigm shift from traditional multi-step sequences, offering a streamlined approach that integrates azide generation and cycloaddition into a single operational unit. For R&D directors and process chemists, this methodology addresses critical pain points regarding safety and operational complexity, while providing supply chain leaders with a robust pathway for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3-triazole derivatives has relied heavily on the classic Huisgen 1,3-dipolar cycloaddition between organic azides and terminal alkynes. While effective, the conventional preparation of the requisite organic azide precursors often involves the reduction of nitro compounds to amines followed by a diazotization reaction. This diazotization step is notoriously hazardous due to the potential formation of unstable diazonium salts and the subsequent isolation of energetic organic azides, which pose severe safety risks on an industrial scale. Furthermore, when the starting material contains sensitive functional groups such as aldehydes, the reduction process can lead to undesirable self-condensation side reactions, significantly compromising yield and purity. Additionally, the reliance on terminal alkynes as coupling partners limits the structural diversity, as specialized alkyne sources are not always commercially available or are prohibitively expensive, creating bottlenecks in the procurement of high-purity pharmaceutical intermediates.
The Novel Approach
The methodology described in Patent CN109776435B circumvents these inherent dangers and inefficiencies by employing a direct one-pot strategy that utilizes 2,2-dibromovinylbenzene compounds and nitrobenzene derivatives as the primary building blocks. Instead of pre-forming dangerous organic azides, the reaction generates the azide species in situ from sodium azide and the nitro compound under mild reducing conditions facilitated by the catalyst system. This eliminates the need for isolating hazardous intermediates and avoids the harsh conditions associated with diazotization. The use of dibromovinylbenzene serves as a versatile alkyne equivalent, expanding the scope of accessible substituents beyond what is possible with standard terminal alkynes. As illustrated in the general reaction scheme below, this approach merges multiple transformation steps into a single vessel, drastically simplifying the workflow and enhancing the overall safety profile of the manufacturing process.
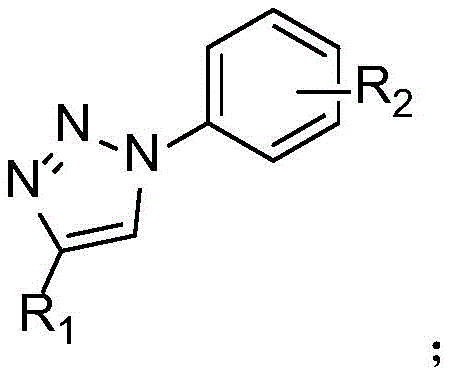
Mechanistic Insights into Copper-Catalyzed In Situ Cycloaddition
The core of this innovative synthesis lies in the intricate interplay between the copper catalyst, the base, and the reducing agent within the reaction medium. The mechanism initiates with the in situ reduction of the nitrobenzene compound to an intermediate capable of forming an azide species upon reaction with sodium azide, a process mediated by the copper catalyst and sodium ascorbate. Sodium ascorbate plays a pivotal role by continuously reducing Cu(II) salts to the catalytically active Cu(I) species, which is essential for facilitating the cycloaddition. The 2,2-dibromovinylbenzene compound acts as the dipolarophile, undergoing elimination of hydrogen bromide to generate the reactive alkyne species or participating directly in the metallacycle formation. This catalytic cycle ensures high regioselectivity, typically favoring the 1,4-disubstituted triazole isomer, which is the desired pharmacophore in most drug discovery applications. The presence of a strong base, such as 1,5-diazabicyclo[5.4.0]undec-5-ene (DBU), further promotes the elimination steps and neutralizes acidic byproducts, driving the equilibrium towards the formation of the triazole ring.
From an impurity control perspective, this mechanism offers distinct advantages over traditional routes. By avoiding the free amine intermediate, the process prevents the formation of azo-coupling byproducts that often plague diazotization reactions. Moreover, the mild reaction conditions, ranging from 25°C to 100°C, minimize thermal degradation of sensitive functional groups like aldehydes and esters, which are prone to decomposition or polymerization under harsher acidic or basic conditions found in classical synthesis. The use of polar aprotic solvents such as hexamethylphosphoric triamide (HMPA) or dimethyl sulfoxide (DMSO) ensures excellent solubility of the ionic reagents like sodium azide, facilitating homogeneous reaction kinetics and consistent batch-to-batch reproducibility. This level of control is critical for meeting the stringent purity specifications required for regulatory submission of new drug candidates.
How to Synthesize 1,2,3-Triazole Derivatives Efficiently
Implementing this one-pot protocol requires precise attention to reagent stoichiometry and atmospheric control to maximize yield and safety. The process begins by charging a reaction vessel with the organic solvent, followed by the sequential addition of the nitrobenzene compound, sodium azide, the 2,2-dibromovinylbenzene derivative, the base, the copper catalyst, and sodium ascorbate. Maintaining an inert nitrogen atmosphere is crucial throughout the reaction to prevent oxidation of the copper catalyst and ensure operator safety when handling azide salts. The reaction mixture is then heated to the specified temperature, typically between 40°C and 80°C depending on the substrate reactivity, and stirred for a period of 2 to 8 hours. Following the reaction, a straightforward workup involving filtration to remove inorganic salts, extraction with ethyl acetate, and purification via recrystallization or silica gel chromatography yields the target triazole compound in high purity. The detailed standardized synthesis steps for this procedure are outlined below.
- Charge a reaction vessel with organic solvent (e.g., HMPA or DMSO), 2,2-dibromovinylbenzene compound, sodium azide, nitrobenzene compound, base (e.g., DBU), copper catalyst, and sodium ascorbate.
- Stir the mixture under nitrogen protection at a controlled temperature between 25°C and 100°C for a duration of 2 to 8 hours to facilitate the cycloaddition.
- Upon completion, remove insolubles via suction filtration, extract with ethyl acetate, wash with saturated brine, dry the organic phase, and purify via recrystallization or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot synthesis method translates into tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By utilizing readily available nitrobenzene compounds and dibromovinylbenzene derivatives instead of specialized, often expensive terminal alkynes or unstable organic azides, manufacturers can leverage commodity chemical markets to secure raw materials at significantly lower costs. This shift reduces dependency on niche suppliers and mitigates the risk of supply disruptions caused by the limited availability of exotic alkyne precursors. Furthermore, the elimination of the diazotization step removes the need for specialized equipment designed to handle explosive intermediates, thereby lowering capital expenditure requirements for facility upgrades and reducing insurance premiums associated with hazardous operations.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the consolidation of multiple reaction steps into a single pot. Traditional multi-step syntheses incur substantial costs related to solvent usage, energy consumption for heating and cooling between steps, and labor for intermediate isolation and purification. By telescoping the azide formation and cycloaddition into one operation, the process significantly reduces solvent waste and energy demand. Additionally, the avoidance of expensive transition metal catalysts that require complex removal protocols, or the use of simple copper salts that are easily managed, contributes to a leaner cost structure. The high yields reported in the patent examples, often exceeding 80%, further enhance the cost-effectiveness by maximizing the output per unit of input material, ensuring that cost reduction in pharmaceutical intermediate manufacturing is achieved without compromising quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and flexibility of the reaction conditions. The method tolerates a wide range of functional groups and substituents, meaning that a single manufacturing platform can be adapted to produce a diverse library of triazole derivatives simply by swapping the starting nitro or dibromovinyl components. This modularity allows for rapid response to changing market demands or clinical trial requirements without the need for extensive process re-validation. The use of stable, shelf-stable reagents like sodium azide and copper salts ensures that inventory management is straightforward, reducing the risk of raw material degradation and spoilage. Consequently, lead times for high-purity pharmaceutical intermediates can be shortened, enabling faster delivery to downstream API manufacturers and accelerating the overall drug development timeline.
- Scalability and Environmental Compliance: Scaling this process from laboratory benchtop to commercial production is facilitated by the absence of hazardous exothermic events typically associated with diazotization. The mild thermal profile allows for the use of standard stainless steel reactors without the need for exotic containment systems. From an environmental standpoint, the one-pot nature of the reaction minimizes the generation of aqueous waste streams associated with multiple workup stages. The ability to use common organic solvents that can be recovered and recycled further aligns the process with green chemistry principles and stringent environmental regulations. This ease of scale-up ensures that commercial production of complex heterocycles can be ramped up efficiently to meet global demand, securing a continuous supply of critical intermediates for the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this one-pot triazole synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, aiming to clarify the practical aspects of adopting this method for industrial applications. Understanding these nuances is essential for process engineers and technical buyers evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this one-pot method improve safety compared to traditional triazole synthesis?
A: Traditional methods often require the isolation of unstable organic azides via diazotization, which poses significant explosion risks. This patented one-pot method generates the azide in situ from sodium azide and nitro compounds, avoiding the handling of hazardous intermediates and eliminating the need for dangerous diazotization steps.
Q: What is the substrate scope for the nitrobenzene and dibromovinyl components?
A: The method demonstrates broad compatibility. Nitrobenzene compounds can bear electron-withdrawing groups such as aldehyde, cyano, carboxyl, or ketone functionalities. Similarly, the dibromovinyl component accepts various substitutions including alkyl, alkoxy, halogen, and heterocyclic groups like pyridine, allowing for diverse functionalization of the final triazole scaffold.
Q: Why is sodium ascorbate included in the reaction mixture?
A: Sodium ascorbate acts as a reducing agent to maintain the copper catalyst in its active +1 oxidation state (Cu(I)). Since the reaction may utilize stable Cu(II) salts like copper sulfate pentahydrate as the precursor, sodium ascorbate ensures the continuous generation of the active Cu(I) species required for the catalytic cycle, thereby sustaining high reaction efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3-Triazole Supplier
The technological advancements presented in Patent CN109776435B underscore the immense potential of 1,2,3-triazole derivatives in modern drug discovery, yet realizing this potential requires a manufacturing partner with deep technical expertise and scalable infrastructure. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that ensure every batch meets the highest international standards. We understand that the transition from a patented laboratory method to a robust commercial process involves nuanced optimization, and our team of expert chemists is dedicated to navigating these challenges to deliver consistent, high-quality results.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this efficient synthesis route for your specific project needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your target molecule, identifying opportunities to optimize raw material usage and processing time. We encourage you to contact our technical procurement team today to request specific COA data for our triazole portfolio and discuss route feasibility assessments that can accelerate your path to market. Let us be your trusted ally in transforming innovative chemistry into commercial success.
