Scalable Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Drug Discovery
The strategic incorporation of fluorine atoms, particularly the trifluoromethyl (CF3) group, into heterocyclic scaffolds has become a cornerstone of modern medicinal chemistry and agrochemical design. Patent CN113121462A discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing critical bottlenecks in the synthesis of these high-value intermediates. The 1,2,3-triazole ring is renowned for its metabolic stability and ability to act as a bioisostere for amide bonds, while the trifluoromethyl group enhances lipophilicity and membrane permeability. Traditionally, accessing these specific substitution patterns has required harsh conditions or hazardous reagents. This new methodology offers a transformative approach by utilizing readily available trifluoroacetimidoyl chlorides and diazo compounds under mild, metal-free conditions. For R&D directors and procurement specialists alike, this represents a significant opportunity to streamline the supply chain for complex fluorinated building blocks, ensuring both cost efficiency and regulatory compliance in the production of active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted triazoles has relied heavily on copper-catalyzed azide-alkyne cycloaddition (CuAAC) followed by subsequent trifluoromethylation steps, or the direct reaction of organic azides with trifluoromethyl ketones. As illustrated in the prior art pathways, these conventional routes suffer from severe safety and operational drawbacks. The use of organic azides poses significant explosion hazards, necessitating specialized equipment and strict safety protocols that drive up operational costs. Furthermore, copper-catalyzed processes introduce transition metal contaminants that are notoriously difficult to remove to the parts-per-million (ppm) levels required by regulatory bodies like the FDA and EMA. The reliance on expensive trifluoromethylating reagents and the need for rigorous post-reaction purification to eliminate heavy metals create a bottleneck in the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel method described in the patent utilizes a base-promoted cyclization between trifluoroacetimidoyl chloride and diazo compounds, completely bypassing the need for azides or transition metal catalysts. 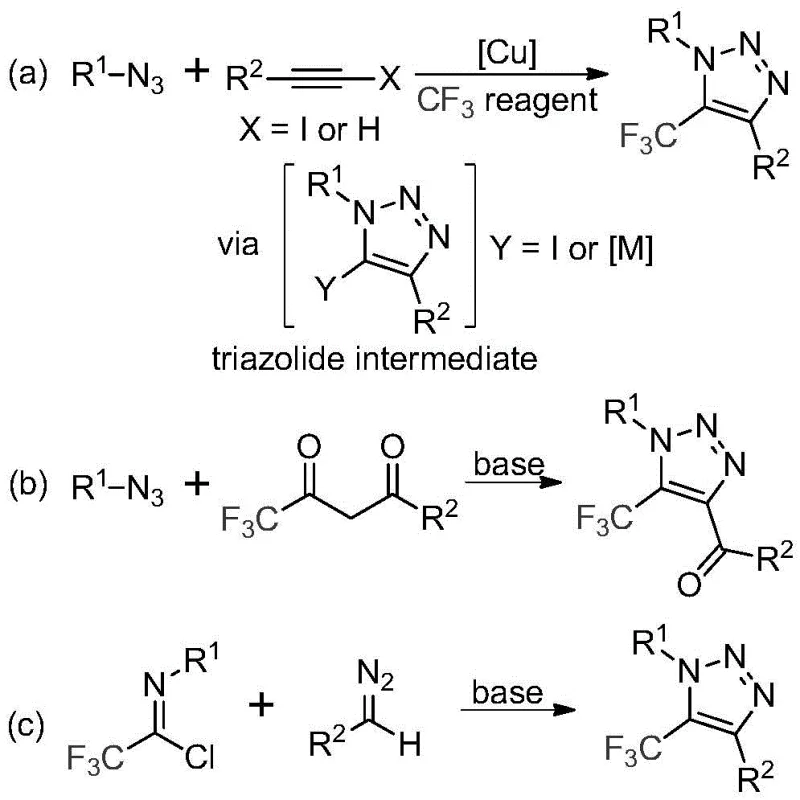 This shift in synthetic strategy fundamentally alters the risk profile and cost structure of the manufacturing process. By employing cesium carbonate as a promoter in common organic solvents like acetonitrile, the reaction proceeds smoothly at moderate temperatures ranging from 50 to 70 °C. The absence of explosive azides and toxic copper catalysts not only enhances laboratory safety but also drastically simplifies the downstream processing. This metal-free approach ensures that the resulting 5-trifluoromethyl-1,2,3-triazoles are generated with high purity, reducing the burden on quality control laboratories and accelerating the time-to-market for new drug candidates.
This shift in synthetic strategy fundamentally alters the risk profile and cost structure of the manufacturing process. By employing cesium carbonate as a promoter in common organic solvents like acetonitrile, the reaction proceeds smoothly at moderate temperatures ranging from 50 to 70 °C. The absence of explosive azides and toxic copper catalysts not only enhances laboratory safety but also drastically simplifies the downstream processing. This metal-free approach ensures that the resulting 5-trifluoromethyl-1,2,3-triazoles are generated with high purity, reducing the burden on quality control laboratories and accelerating the time-to-market for new drug candidates.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic elegance of this transformation lies in the intermolecular nucleophilic addition-elimination process facilitated by the base. The reaction initiates with the deprotonation of the diazo compound by cesium carbonate, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroacetimidoyl chloride. This is followed by the elimination of a chloride ion and a subsequent intramolecular 5-endo-dig cyclization to form the triazole ring. 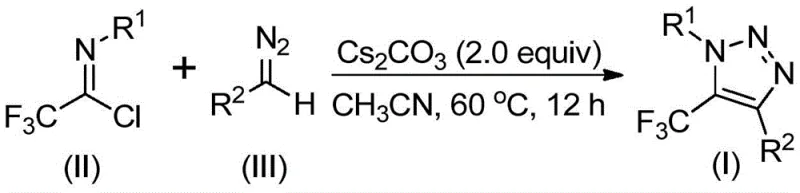 This pathway is highly efficient because it avoids the formation of stable metal-carbene intermediates often seen in rhodium or copper catalysis, which can lead to unpredictable side reactions. The use of 4 Å molecular sieves in the reaction mixture further drives the equilibrium forward by sequestering trace moisture, which could otherwise hydrolyze the sensitive imidoyl chloride starting material. For process chemists, understanding this mechanism is vital for optimizing reaction parameters; the data suggests that maintaining a slight excess of the diazo compound (1.5 equivalents) relative to the imidoyl chloride ensures complete conversion while minimizing the formation of unreacted starting materials.
This pathway is highly efficient because it avoids the formation of stable metal-carbene intermediates often seen in rhodium or copper catalysis, which can lead to unpredictable side reactions. The use of 4 Å molecular sieves in the reaction mixture further drives the equilibrium forward by sequestering trace moisture, which could otherwise hydrolyze the sensitive imidoyl chloride starting material. For process chemists, understanding this mechanism is vital for optimizing reaction parameters; the data suggests that maintaining a slight excess of the diazo compound (1.5 equivalents) relative to the imidoyl chloride ensures complete conversion while minimizing the formation of unreacted starting materials.
From an impurity control perspective, this mechanism offers distinct advantages over traditional methods. The mild basic conditions prevent the decomposition of sensitive functional groups that might be present on the aromatic rings of the substrates. Unlike acid-catalyzed cyclizations which can promote polymerization or rearrangement, the cesium carbonate system is sufficiently gentle to tolerate esters, ketones, and phosphonates, as evidenced by the successful synthesis of diverse derivatives in the patent examples. The reaction produces nitrogen gas as the only byproduct from the diazo moiety, which harmlessly vents from the reaction vessel, leaving behind a clean reaction profile. This cleanliness is paramount for industrial applications, as it reduces the number of crystallization or chromatography steps required to meet stringent purity specifications, thereby directly impacting the overall yield and cost of goods sold (COGS).
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
To implement this synthesis effectively, precise control over stoichiometry and reaction environment is essential. The protocol dictates the use of anhydrous conditions and specific molar ratios to maximize efficiency. While the general procedure is robust, adhering to the optimized parameters found in the patent data will ensure reproducible results across different scales of operation. The following guide outlines the standardized steps derived from the experimental examples provided in the intellectual property documentation.
- Prepare the reaction mixture by adding cesium carbonate (2.0 equivalents), 4 Å molecular sieves, trifluoroacetimidoyl chloride, and the diazo compound into an organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature range of 50-70 °C and maintain stirring for 8 to 16 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture to remove solids, concentrate the filtrate, and purify the crude product via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple chemical yield. The elimination of transition metal catalysts removes a major cost center associated with ligand procurement and, more importantly, the expensive and time-consuming heavy metal scavenging processes required before final API release. This metal-free characteristic significantly reduces the complexity of the purification train, allowing for faster batch turnover and reduced solvent consumption. Furthermore, the starting materials—trifluoroacetimidoyl chlorides and diazo compounds—are commercially available or easily synthesized from commodity chemicals, ensuring a stable and resilient supply chain that is not dependent on scarce precious metals like palladium or rhodium.
- Cost Reduction in Manufacturing: The economic impact of switching to this base-promoted method is substantial due to the drastic simplification of the bill of materials. By replacing expensive copper catalysts and specialized trifluoromethylating reagents with inexpensive cesium carbonate, the direct material costs are significantly lowered. Additionally, the operational expenditure is reduced because the process does not require inert atmosphere gloveboxes typically needed for handling sensitive organometallic catalysts, nor does it require the extensive wastewater treatment protocols associated with heavy metal disposal. The high atom economy of the reaction, where the only byproduct is nitrogen gas and salt, further contributes to a leaner, more cost-effective manufacturing process suitable for large-scale production.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of robust, shelf-stable reagents that do not require cryogenic storage or special handling permits associated with explosive azides. The ability to source trifluoroacetimidoyl chlorides and diazo precursors from multiple global suppliers mitigates the risk of single-source dependency. Moreover, the reaction's tolerance to a wide variety of functional groups means that a single platform technology can be used to synthesize a broad library of analogs without needing to requalify entirely new synthetic routes for each derivative. This flexibility allows manufacturers to respond rapidly to changing market demands or clinical trial requirements without disrupting the production schedule.
- Scalability and Environmental Compliance: Scaling this reaction from gram to multi-kilogram quantities is straightforward due to the absence of exothermic hazards commonly associated with azide chemistry. The mild temperature range of 50 to 70 °C is easily maintained in standard glass-lined or stainless steel reactors, eliminating the need for specialized high-pressure or cryogenic equipment. From an environmental standpoint, the process aligns with green chemistry principles by avoiding toxic heavy metals and generating minimal waste. The use of acetonitrile, a solvent that can be efficiently recovered and recycled, further enhances the sustainability profile of the manufacturing process, helping companies meet increasingly rigorous corporate social responsibility (CSR) and environmental, social, and governance (ESG) goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis platform. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear framework for evaluating the technology's fit within your existing manufacturing capabilities. Understanding these nuances is critical for making informed decisions about process adoption and vendor qualification.
Q: Why is this base-promoted method superior to copper-catalyzed azide cycloaddition?
A: This method eliminates the need for toxic and potentially explosive organic azides and avoids the use of transition metal catalysts like copper. This significantly simplifies the purification process by removing the requirement for rigorous heavy metal scavenging, which is critical for pharmaceutical grade intermediates.
Q: What are the optimal reaction conditions for scaling this synthesis?
A: The patent data indicates that using acetonitrile as the solvent with cesium carbonate as the base at temperatures between 60 °C provides the highest efficiency. The reaction tolerates a wide range of functional groups, making it robust for scale-up from gram to kilogram levels.
Q: Can this method produce diverse derivatives for SAR studies?
A: Yes, the method demonstrates high substrate tolerance. By varying the R1 group on the imidoyl chloride and the R2 group on the diazo compound, diverse 1,4-disubstituted triazoles containing trifluoromethyl groups can be synthesized efficiently for structure-activity relationship studies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated heterocycles play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the laboratory bench to full-scale manufacturing. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the exacting standards required for pharmaceutical and agrochemical applications. Our expertise in metal-free synthesis allows us to offer a superior purity profile, free from the persistent heavy metal contaminants that plague traditional catalytic methods.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Whether you require custom synthesis of novel analogs for structure-activity relationship (SAR) studies or bulk supply for commercial production, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to see how our optimized processes can enhance your supply chain efficiency and reduce your overall time to market.
