Advanced Acid-Catalyzed Synthesis of Benzopyran Compounds for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex heterocyclic scaffolds, particularly those found in bioactive natural products and advanced functional materials. Patent CN100434427C, filed in late 2008, introduces a significant advancement in the synthesis of benzopyran compounds, a class of molecules renowned for their structural similarity to anthocyanins and their potential as potassium channel activators or HIV reverse transcriptase inhibitors. This intellectual property details a novel acid-catalyzed cyclization strategy that overcomes the limitations of traditional multi-step syntheses, offering a direct route to 2-aryl-2-hydroxy-benzopyrans and their ester derivatives. By leveraging the unique reactivity of phenolic compounds with alpha,beta-unsaturated ketones or beta,gamma-unsaturated enone esters under reflux conditions, this technology provides a scalable pathway for generating high-value intermediates essential for drug discovery and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzopyran core, especially the sensitive 2-aryl-2-hydroxy variants, has been fraught with synthetic challenges that hinder large-scale production. Conventional approaches often rely on the condensation of highly reactive substrates such as diphenols or triphenols with 1,3-diketones, which limits the structural diversity of the final product due to the scarcity and cost of such specific starting materials. Furthermore, alternative routes involving Grignard reagents reacting with flavonoids introduce significant safety hazards and operational complexities, requiring strict anhydrous conditions and cryogenic temperatures that are difficult to maintain in a commercial manufacturing environment. These legacy methods frequently suffer from poor atom economy, generate substantial hazardous waste streams, and often result in low overall yields due to the instability of intermediates under the harsh basic or nucleophilic conditions required. Consequently, the supply chain for these critical intermediates has remained fragmented, with high costs and long lead times deterring broader exploration of their therapeutic potential.
The Novel Approach
In stark contrast, the methodology disclosed in CN100434427C presents a paradigm shift by utilizing a straightforward acid-catalyzed cyclization that operates under relatively mild yet effective thermal conditions. This innovative route employs readily available phenolic compounds and chalcones or unsaturated enone esters as building blocks, reacting them in an organic acid medium such as trifluoroacetic acid (TFA) or formic acid. The process eliminates the need for sensitive organometallic reagents and allows for the direct formation of the benzopyran ring system with high regioselectivity. A key feature of this approach is the ability to recover and reuse the acidic catalyst; after the reaction reaches completion, the trifluoroacetic acid can be distilled off and recycled for future batches, drastically reducing raw material consumption and waste disposal costs. This streamlined workflow not only simplifies the purification process but also enhances the overall sustainability profile of the manufacturing operation, making it an attractive option for industrial scale-up.
Mechanistic Insights into Acid-Catalyzed Cyclization
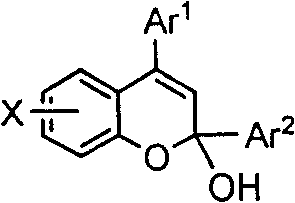
The core of this synthetic breakthrough lies in the electrophilic activation of the unsaturated carbonyl system by the strong organic acid, which facilitates a cascade of intramolecular cyclization events. When the phenolic hydroxyl group attacks the activated beta-carbon of the alpha,beta-unsaturated ketone or the gamma-carbon of the beta,gamma-unsaturated ester, a transient carbocation intermediate is formed. This intermediate subsequently undergoes dehydration and aromatization to yield the stable benzopyran framework. The versatility of this mechanism is evidenced by its tolerance to a wide range of substituents on the aromatic rings, including alkyl, aryl, methoxy, and halogen groups, as defined by the variables X, Ar1, and Ar2 in the general structure. This flexibility allows medicinal chemists to rapidly generate libraries of analogues for structure-activity relationship (SAR) studies without needing to redesign the entire synthetic route for each new derivative.
Furthermore, the reaction conditions are meticulously optimized to control impurity profiles, which is critical for pharmaceutical applications. The use of trifluoroacetic acid not only drives the equilibrium towards the product but also suppresses side reactions such as polymerization of the unsaturated precursors, which are common pitfalls in acid-catalyzed processes. The workup procedure involves a simple aqueous wash with saturated sodium bicarbonate to neutralize residual acid, followed by drying and standard column chromatography. This gentle purification strategy ensures that the final product retains its structural integrity, avoiding the decomposition often seen with base-sensitive benzopyran salts. The resulting compounds are characterized by high purity, confirmed through rigorous spectroscopic analysis including NMR, MS, and IR, ensuring they meet the stringent quality standards required for downstream biological testing and clinical development.
How to Synthesize 2-Aryl-2-hydroxy-benzopyrans Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, operators must adhere to specific thermal and stoichiometric parameters outlined in the patent examples. The process generally involves mixing the phenolic substrate and the unsaturated ketone or ester in a molar ratio of approximately 1:2 within a reactor charged with a significant excess of trifluoroacetic acid, typically 30 times the weight of the phenol. The mixture is then heated to reflux for a duration of 1 to 3 days, depending on the electronic nature of the substituents, to ensure full conversion. Following the reaction, the volatile acid is recovered via distillation, and the crude residue is processed through liquid-liquid extraction and silica gel chromatography using a petroleum ether and ethyl acetate gradient. For a comprehensive, step-by-step standard operating procedure tailored to your specific facility capabilities, please refer to the detailed guide below.
- Reflux phenolic compounds with alpha,beta-unsaturated ketones or beta,gamma-unsaturated enone esters in organic acid (TFA) for 1-3 days.
- Distill off the trifluoroacetic acid for reuse, then dissolve the residue in dichloromethane.
- Wash with saturated sodium bicarbonate, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this acid-catalyzed technology offers profound benefits for procurement managers and supply chain directors looking to optimize their vendor networks. The primary advantage lies in the drastic simplification of the raw material portfolio; by utilizing commodity chemicals like phenols and chalcones instead of specialized organometallic reagents, companies can mitigate supply risks associated with scarce or geopolitically sensitive materials. Additionally, the ability to recycle the trifluoroacetic acid solvent creates a closed-loop system that significantly lowers the total cost of ownership for the manufacturing process. This reduction in consumable usage translates directly into improved margin structures for the final API or intermediate, providing a competitive edge in price-sensitive markets without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the implementation of solvent recovery protocols lead to substantial operational expenditure savings. By avoiding the need for complex metal scavenging steps required in palladium or copper-catalyzed couplings, the downstream processing costs are minimized, and the environmental burden of heavy metal waste is completely removed. This lean manufacturing approach ensures that the production of high-purity benzopyran intermediates remains economically viable even at smaller batch sizes, facilitating agile responses to market demand fluctuations.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against variations in raw material quality ensures consistent output, reducing the risk of batch failures that can disrupt global supply chains. Since the starting materials are widely available from multiple chemical suppliers globally, procurement teams are not locked into single-source dependencies, thereby enhancing negotiation leverage and securing continuity of supply. The simplified workflow also shortens the overall production cycle time, allowing for faster turnaround from order placement to delivery, which is critical for just-in-time inventory management strategies employed by major pharmaceutical corporations.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-ton commercial production without requiring fundamental changes to the reaction engineering. The use of organic acids rather than corrosive mineral acids or toxic heavy metals aligns with modern green chemistry principles, simplifying the permitting process and reducing the costs associated with hazardous waste treatment. This environmental compatibility not only safeguards the company's reputation but also future-proofs the supply chain against increasingly stringent global regulations regarding chemical manufacturing emissions and effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzopyran synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on yield expectations, substrate scope, and process safety. Understanding these nuances is essential for R&D teams evaluating this route for inclusion in their development pipelines and for supply chain partners assessing the feasibility of long-term contracts.
Q: What are the typical yields for this benzopyran synthesis method?
A: According to patent CN100434427C, the target products obtained after separation and purification exhibit yields ranging from 50% to 85%, demonstrating robust efficiency for complex heterocycles.
Q: Can the catalyst be recycled in this process?
A: Yes, the process utilizes trifluoroacetic acid which can be distilled out after the reaction is complete and reused for subsequent batches, significantly enhancing process sustainability.
Q: What types of substrates are compatible with this cyclization?
A: The method accommodates a wide variety of phenolic compounds and unsaturated ketones or esters, allowing for the synthesis of diverse 2,2-disubstituted benzopyran derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzopyran Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics and functional materials. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and efficient. We are committed to delivering benzopyran compounds with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our dedication to technical excellence ensures that our clients receive materials that are ready for immediate use in sensitive biological assays or further synthetic transformations.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our technical procurement team is prepared to provide a Customized Cost-Saving Analysis that demonstrates how switching to this optimized route can improve your bottom line. Please contact us today to request specific COA data for our current inventory or to discuss route feasibility assessments for your proprietary targets, and let us help you accelerate your path to market with confidence and reliability.
