Advanced Six-Step Synthesis of Sildenafil: Enhancing Purity and Scalability for Global Supply Chains
Advanced Six-Step Synthesis of Sildenafil: Enhancing Purity and Scalability for Global Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways for high-demand active pharmaceutical ingredients (APIs) like Sildenafil, widely known under the trade name Virga. Patent CN102690273A introduces a sophisticated preparation method that addresses critical bottlenecks in traditional synthesis routes. This innovation outlines a streamlined six-step process initiating from readily available 2-ethoxybenzoic acid and N-hydroxysuccinimide. By leveraging an active ester strategy followed by precise cyclization and sulfonylation steps, the methodology achieves superior control over reaction conditions. For R&D directors and procurement specialists, this represents a pivotal shift towards more predictable and efficient pharmaceutical intermediates production. The technical breakthrough lies not just in the chemical transformations, but in the inherent stability of the intermediates generated throughout the sequence, which directly correlates to reduced waste and higher final purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sildenafil has been plagued by operational inefficiencies and chemical instability issues documented in earlier patents. For instance, the method disclosed in CN1057464 necessitates a protracted reaction time extending up to four days, coupled with a loaded and trivial treatment process that demands extensive crystallization purifying steps to isolate the final product. Furthermore, alternative routes such as those found in CN101429166 suffer from significant drawbacks regarding intermediate stability; specifically, the chlorosity on the pyrimidine ring is prone to facile hydrolysis, leading to complex mixtures of byproducts that are notoriously difficult to remove. These conventional approaches often involve harsh conditions that generate substantial impurities, thereby complicating the downstream purification workflow and negatively impacting the overall economic viability of the manufacturing process.
The Novel Approach
In stark contrast, the novel approach detailed in CN102690273A circumvents these historical challenges through a carefully engineered sequence that prioritizes intermediate stability and operational simplicity. By utilizing an active ester intermediate (Compound II) formed from 2-ethoxybenzoic acid, the process ensures a highly efficient condensation reaction with the pyrazole derivative. This route eliminates the need for unstable acyl chloride intermediates that typically react with difficulty to form sulphonamides, a common pain point in legacy technologies. The result is a synthesis pathway characterized by fewer side reactions, milder reaction conditions, and a significantly simplified purification treatment process. This evolution in synthetic design offers a compelling value proposition for cost reduction in pharmaceutical intermediates manufacturing, as it minimizes the resource intensity associated with separating complex impurity profiles.
Mechanistic Insights into Active Ester Mediated Cyclization
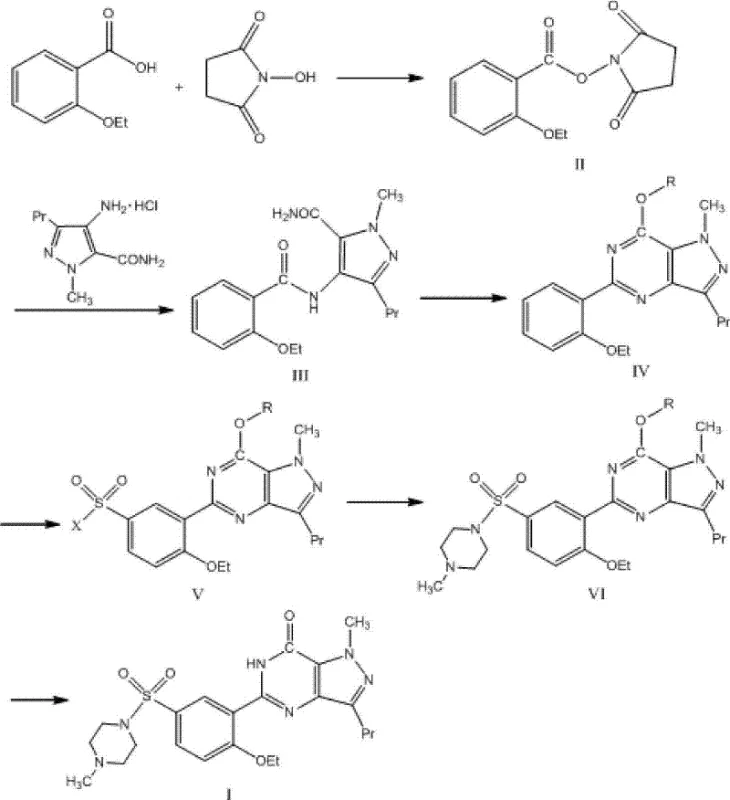
The core of this synthetic strategy relies on the formation of a stable active ester, N-(2-phenetole formyl) succinimide (Compound II), which serves as a superior acylating agent compared to traditional acid chlorides. In the initial step, 2-ethoxybenzoic acid reacts with N-hydroxysuccinimide in the presence of N,N'-dicyclohexylcarbodiimide (DCC) at room temperature. This activation step is crucial as it prevents the degradation often seen with more reactive species. Subsequently, Compound II undergoes condensation with 1-methyl-3-propyl-4-aminopyrazolyl-5-formamide hydrochloride. The use of acid binding agents like DMAP or DIEA facilitates this amide bond formation under reflux conditions, yielding Compound III with high fidelity. This mechanistic choice effectively decouples the activation energy requirements from the stability constraints, allowing for a cleaner reaction profile that is easier to monitor and control on a commercial scale.
Following the amide formation, the pathway proceeds through a critical cyclization and chlorination phase using a mixture of phosphorus oxychloride (POCl3) and phosphorus pentachloride. This step constructs the pyrazolo[4,3-d]pyrimidine core, a structural motif essential for the biological activity of Sildenafil. The subsequent nucleophilic substitution with alcohol converts the chloro-species into the alkoxy derivative (Compound IV). Crucially, the patent data indicates that the intermediates generated in this sequence possess remarkable stability, resisting the hydrolysis that plagues other methods. This stability is paramount for maintaining high-purity pharmaceutical intermediates throughout the batch cycle. The final stages involve sulphonyl halogenating with chlorosulfonic acid to introduce the sulfonyl chloride moiety, followed by nucleophilic displacement with N-methylpiperazine and final hydrolysis. Each step is optimized to minimize side products, ensuring that the impurity spectrum remains narrow and manageable.
How to Synthesize Sildenafil Efficiently
The execution of this synthesis requires precise control over stoichiometry and temperature to maximize the yields reported in the patent embodiments. The process begins with the activation of the benzoic acid derivative, followed by sequential functionalization of the pyrazole ring system. Operators must pay close attention to the exothermic nature of the chlorosulfonation step, maintaining temperatures below 5°C to prevent decomposition. The detailed standardized synthetic steps, including specific solvent volumes, reagent ratios, and workup procedures for each of the six transformations, are outlined in the technical guide below. Adhering to these protocols ensures the reproducibility of the high yields (ranging from 69% to 89% per step) demonstrated in the experimental data.
- Condense 2-ethoxybenzoic acid with N-hydroxysuccinimide using DCC to form the active ester intermediate (Compound II).
- React Compound II with 1-methyl-3-propyl-4-aminopyrazolyl-5-formamide hydrochloride to generate the amide precursor (Compound III).
- Perform cyclization and chlorination using POCl3 and phosphorus pentachloride, followed by nucleophilic substitution with alcohol to obtain Compound IV.
- Conduct sulphonyl halogenating reaction with chlorosulfonic acid to produce the sulfonyl chloride derivative (Compound V).
- React Compound V with N-methylpiperazine to form the sulfonamide intermediate (Compound VI).
- Execute final hydrolysis in alkaline aqueous solution to yield the target product Sildenafil (Compound I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates into tangible operational efficiencies and risk mitigation. The primary advantage stems from the accessibility and low cost of the starting materials, specifically 2-ethoxybenzoic acid and N-hydroxysuccinimide, which are commodity chemicals with robust global supply chains. This reduces the dependency on exotic or single-source reagents that often create bottlenecks. Furthermore, the stability of the intermediates allows for greater flexibility in production scheduling; batches can be held at intermediate stages without significant degradation, providing a buffer against demand fluctuations. This operational resilience is a key factor in reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the need for emergency re-runs due to failed stability tests.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of mild reaction conditions significantly lower the operational expenditure associated with this synthesis. By avoiding expensive heavy metal removal steps and reducing the energy consumption required for extreme temperatures, the overall cost of goods sold is optimized. Additionally, the high yields reported in the patent embodiments mean that less raw material is wasted per kilogram of final product, directly enhancing the margin profile for manufacturers seeking cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the chemical route ensures consistent output quality, which is critical for maintaining long-term contracts with downstream API producers. The simplicity of the purification process, which avoids complex chromatographic separations in favor of crystallization and washing, accelerates the throughput time. This efficiency supports the commercial scale-up of complex pharmaceutical intermediates, enabling suppliers to meet large volume orders without compromising on delivery schedules or product specifications.
- Scalability and Environmental Compliance: The process generates fewer side reactions and impurities, which simplifies waste stream management and reduces the environmental footprint of the manufacturing facility. The use of standard organic solvents like dichloromethane and ethanol, combined with aqueous workups, aligns well with existing industrial infrastructure. This compatibility facilitates rapid commercial scale-up of complex pharmaceutical intermediates from pilot plants to multi-ton production facilities, ensuring that supply can scale linearly with market demand while adhering to strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Sildenafil synthesis method. These insights are derived directly from the comparative data and experimental results presented in the patent documentation, providing clarity on why this route is preferred for modern manufacturing. Understanding these nuances helps stakeholders make informed decisions about sourcing and process adoption.
Q: How does this synthesis method improve upon previous Sildenafil production techniques?
A: Unlike prior art such as CN1057464 which requires four days of reaction time and tedious crystallization, or CN101429166 which suffers from unstable intermediates and difficult byproduct removal, this method utilizes stable intermediates and mild conditions. It significantly reduces side reactions, simplifies purification, and enhances overall yield and purity.
Q: What are the key advantages regarding raw material availability and cost?
A: The process starts with 2-ethoxybenzoic acid and N-hydroxysuccinimide, which are commercially accessible and inexpensive raw materials. The six-step route is designed for operational simplicity, avoiding complex transition metal catalysts or extreme conditions that typically drive up manufacturing costs.
Q: Is the intermediate stability sufficient for large-scale industrial application?
A: Yes, the patent explicitly highlights that the intermediates possess certain stability, addressing a critical failure point in other methods where chlorosity on the pyrimidine ring was prone to facile hydrolysis. This stability ensures consistent quality and reliable supply chain continuity during scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sildenafil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN102690273A to maintain competitiveness in the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Sildenafil intermediate meets the highest international standards. We are committed to delivering high-purity pharmaceutical intermediates that empower your drug development and manufacturing pipelines.
We invite you to engage with our technical team to explore how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic advantages tailored to your volume needs. Please contact our technical procurement team today to索取 specific COA data and route feasibility assessments, and let us demonstrate why we are the partner of choice for reliable, scalable, and cost-effective chemical solutions.
