Advanced Manufacturing of Pyrazolopyrimidinone Derivatives for Erectile Dysfunction Therapy
The pharmaceutical landscape for treating erectile dysfunction continues to evolve, driven by the demand for more potent and selective phosphodiesterase-5 (PDE5) inhibitors. Patent CN1441802A introduces a significant breakthrough in the preparation of pyrazolopyrimidinone derivatives, which serve as critical active pharmaceutical ingredients (APIs) or intermediates in this therapeutic class. Unlike traditional synthetic routes that rely on harsh conditions and expensive coupling agents, this invention proposes a streamlined methodology involving the chlorosulfonation of pyrazole amides followed by amination and intramolecular cyclization. This approach not only enhances the overall yield but also significantly improves the economic feasibility of producing these complex heterocyclic compounds. For global procurement teams and R&D directors, understanding this shift in synthetic strategy is vital for securing a reliable supply chain of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazolopyrimidinone scaffolds, such as those found in sildenafil analogues, has relied on a sequence that begins with the functionalization of alkoxybenzoic acids. As illustrated in the reaction scheme below, conventional methods typically involve the chlorosulfonation of an alkoxybenzoic acid, followed by amidation with a primary amine to form a sulfonamide-substituted benzoic acid. The critical bottleneck in this traditional pathway occurs in the subsequent step, where the carboxylic acid group must be activated to couple with a pyrazole amine. This activation often necessitates the use of costly and hazardous reagents such as trichlorobenzoyl chloride or N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline (EEDQ). Furthermore, the yield of the initial sulfonamide formation from chlorosulfonated alkoxybenzoic acid is frequently suboptimal, leading to cumulative yield losses that negatively impact the overall process efficiency and cost structure.
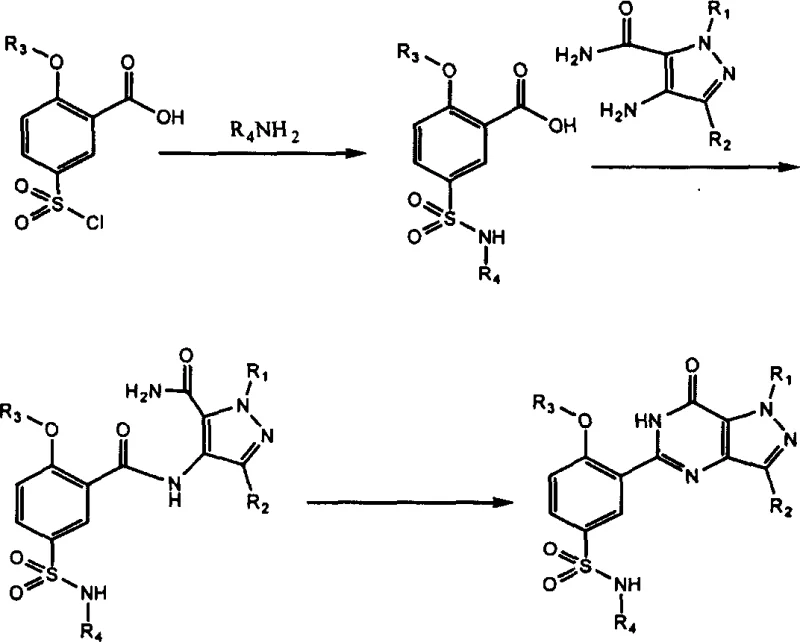
The Novel Approach
In stark contrast to the legacy processes, the method disclosed in CN1441802A fundamentally reorders the synthetic sequence to bypass these inefficiencies. The innovation lies in starting with a pre-formed pyrazole amide compound, which is then subjected to chlorosulfonation. By shifting the chlorosulfonation step to occur on the pyrazole amide rather than the benzoic acid, the process eliminates the need for the problematic carboxyl activation step entirely. The resulting chlorosulfonyl-pyrazole intermediate is then reacted with a primary amine to install the necessary sulfonamide moiety. Finally, a mild intramolecular cyclization closes the pyrimidinone ring. This strategic reversal of steps not only removes the dependency on expensive coupling reagents but also operates under milder conditions, thereby preserving the integrity of sensitive functional groups and ensuring a cleaner reaction profile suitable for large-scale manufacturing.
Mechanistic Insights into Chlorosulfonation and Cyclization
The core of this technological advancement rests on the precise control of the chlorosulfonation mechanism. In the novel pathway, the pyrazole amide substrate is treated with chlorosulfonic acid, optionally in the presence of thionyl chloride, at temperatures maintained below 20°C. This low-temperature regime is crucial for preventing over-sulfonation or degradation of the pyrazole ring, which can occur under more vigorous conditions. The electrophilic aromatic substitution selectively targets the position ortho to the alkoxy group on the benzene ring (if present in the specific substrate design) or activates the sulfonyl chloride handle efficiently. Following this, the nucleophilic attack by the primary amine on the sulfonyl chloride group proceeds rapidly to form the sulfonamide bond. This step is highly exothermic but manageable, allowing for the use of stoichiometric amounts of amine or slight excesses to drive the reaction to completion without generating significant byproducts.
The final stage involves an intramolecular cyclization that constructs the fused pyrazolo[4,3-d]pyrimidin-7-one core. This transformation is facilitated by strong bases such as potassium tert-butoxide or DBU in alcoholic or aprotic solvents. The mechanism involves the deprotonation of the amide nitrogen or the adjacent carbon, followed by nucleophilic attack on the carbonyl carbon of the amide side chain, leading to ring closure and elimination of ammonia or an amine fragment depending on the specific substituents. This cyclization is remarkably efficient, often proceeding in a one-pot manner directly from the crude sulfonamide intermediate. The ability to telescope these steps reduces solvent usage and waste generation, aligning with green chemistry principles while maintaining high stereochemical integrity for chiral variants of the molecule.
How to Synthesize Pyrazolopyrimidinone Efficiently
The synthesis of these high-value intermediates requires strict adherence to the optimized reaction parameters outlined in the patent to ensure maximum yield and purity. The process is designed to be robust, utilizing readily available starting materials and standard laboratory equipment, which facilitates easy technology transfer to pilot and commercial plants. Operators must pay close attention to temperature control during the chlorosulfonation phase to avoid exotherms, and careful selection of the base for the cyclization step is essential to prevent hydrolysis of the sensitive sulfonamide linkage. The detailed standardized synthesis steps see the guide below.
- Perform chlorosulfonation on the pyrazole amide precursor using chlorosulfonic acid at low temperatures (below 20°C) to generate the chlorosulfonyl intermediate.
- React the chlorosulfonyl intermediate with a primary amine (such as substituted pyrrolidines) in a suitable solvent like dichloromethane to form the sulfonamide compound.
- Execute intramolecular cyclization using a base such as potassium tert-butoxide in tert-butanol under reflux conditions to close the pyrimidinone ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible benefits that extend beyond simple chemical yield. The primary advantage is the drastic simplification of the raw material portfolio. By eliminating the need for specialized coupling reagents like EEDQ or trichlorobenzoyl chloride, the process reduces exposure to volatile pricing markets for these niche chemicals. Furthermore, the removal of these reagents simplifies the downstream purification process, as there are fewer urea or acyl chloride byproducts to remove. This translates directly into reduced processing time and lower consumption of chromatography media or recrystallization solvents, driving down the overall cost of goods sold (COGS) for the final API intermediate.
- Cost Reduction in Manufacturing: The most significant financial impact comes from the complete avoidance of expensive carboxyl activating agents. In conventional synthesis, these reagents can account for a substantial portion of the raw material cost, and their stoichiometric use generates equimolar waste that requires disposal. By restructuring the synthesis to start with a pyrazole amide, the new method bypasses this cost center entirely. Additionally, the higher yields reported in the patent examples mean that less starting material is required to produce the same amount of final product, effectively lowering the material cost per kilogram. The use of common solvents like dichloromethane and tert-butanol further ensures that solvent recovery and recycling systems can be easily implemented to maximize economic efficiency.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on commodity chemicals rather than specialized reagents. Chlorosulfonic acid, thionyl chloride, and common primary amines are produced globally in massive quantities, ensuring stable availability and minimizing the risk of supply disruptions. In contrast, specialized coupling reagents often have fewer suppliers and longer lead times. The robustness of the new process also means that batch-to-batch variability is minimized, reducing the likelihood of failed batches that could delay shipments to downstream API manufacturers. This reliability is critical for maintaining continuous production schedules in the highly regulated pharmaceutical sector.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is highly favorable. The reduction in the number of distinct chemical steps and the potential for one-pot operations significantly decrease the volume of waste solvent generated per unit of product. This aligns with increasingly stringent environmental regulations regarding waste disposal and emissions. The mild reaction conditions, particularly the low-temperature chlorosulfonation, reduce energy consumption for heating and cooling compared to processes requiring prolonged reflux or cryogenic conditions. These factors combined make the technology highly scalable from kilogram to multi-ton production without the need for exotic reactor configurations or complex safety mitigation systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrazolopyrimidinone synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear basis for technical evaluation and feasibility studies.
Q: How does this new method reduce production costs compared to conventional routes?
A: The novel process eliminates the need for expensive carboxyl activating reagents like trichlorobenzoyl chloride or EEDQ, which are required in traditional methods to couple the benzoic acid and pyrazole amine. By reversing the sequence and starting with a pre-formed pyrazole amide, the synthesis avoids these costly additives and simplifies purification.
Q: What are the key purity advantages of this chlorosulfonation approach?
A: Conducting chlorosulfonation on the pyrazole amide rather than the benzoic acid allows for milder reaction conditions and better control over side reactions. This results in high-purity sulfonamide intermediates without the complex impurity profiles often associated with aggressive activation steps in older methodologies.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the process utilizes common industrial solvents like dichloromethane and tert-butanol and avoids sensitive transition metal catalysts. The one-pot potential for the amination and cyclization steps further enhances scalability by reducing unit operations and solvent exchanges.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolopyrimidinone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced patent technologies like CN1441802A into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of pyrazolopyrimidinone intermediate meets the exacting standards required for global pharmaceutical registration. We understand that consistency is key in the supply of erectile dysfunction therapeutics, and our infrastructure is designed to deliver that consistency reliably.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and high-yield synthetic route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive landscape of PDE5 inhibitor manufacturing.
