Advanced Tetramethyl Spiroindane Phosphine-Oxazoline Ligands for Scalable Asymmetric Synthesis
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the need for more efficient and economically viable chiral ligands. Patent CN108659045B introduces a groundbreaking phosphine-oxazoline ligand compound based on a tetramethyl spiroindane skeleton, addressing critical limitations in current industrial synthesis. This innovation leverages cheap and easily obtained tetramethyl spiroindane diphenol as a starting material, bypassing the complex and costly resolutions associated with traditional SPINOL-based ligands. The technical breakthrough lies in the unique structural rigidity provided by the tetramethyl groups, which optimizes the dihedral angle for superior enantioselectivity in metal-catalyzed reactions. For R&D directors and procurement managers, this represents a pivotal shift towards high-purity pharmaceutical intermediates that are both cost-effective and scalable. The patent outlines a robust 9-step synthesis route that significantly reduces the dependency on expensive chiral pool materials, thereby enhancing the overall economic practicability of the process. This development is not merely a laboratory curiosity but a viable industrial solution for the commercial scale-up of complex polymer additives and fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing chiral phosphine-oxazoline ligands often rely on SPINOL (1,1'-spirobiindane-7,7'-diol) skeletons, which present substantial bottlenecks for large-scale manufacturing. The conventional route typically requires at least 13 steps of synthetic reaction starting from industrially available m-methoxybenzaldehyde, coupled with an additional step for chiral resolution. This extended synthetic sequence inherently accumulates impurities and drastically reduces overall yield, leading to prohibitive production costs for high-purity OLED material or API intermediate manufacturing. Furthermore, the reliance on specific chiral resolution steps introduces variability and supply chain risks, as the availability of optical enantiomers can be inconsistent. The structural limitations of these older ligands also restrict the introduction of electron-withdrawing groups on the phosphorus phenyl rings, limiting their catalytic versatility in challenging asymmetric transformations. Consequently, manufacturers face significant hurdles in achieving cost reduction in electronic chemical manufacturing when relying on these legacy ligand systems. The high dependence on reaction substrates and the lack of universality further complicate process development for diverse chemical portfolios.
The Novel Approach
The novel approach disclosed in the patent utilizes tetramethyl spiroindane diphenol (MSPINOL) as a foundational building block, which can be directly obtained from industrial bisphenol series products in high yield via acid catalysis. This strategic shift reduces the synthetic route to just 9 steps, eliminating the need for complex chiral resolution procedures that plague conventional methods. The tetramethyl spiroindane skeleton is inherently more stable and rigid due to the steric hindrance provided by the methyl groups, which locks the conformation and enhances catalytic performance without additional stabilization steps. This method allows for the preparation of phosphine-oxazoline ligands with electron-withdrawing groups, such as p-trifluoromethyl or 3,5-bis(trifluoromethyl) substituents, which were previously inaccessible or difficult to synthesize. By starting from cheap and rich raw materials like bisphenol A derivatives, the process achieves substantial cost savings and improves the practicability for industrial application. The streamlined workflow ensures a more consistent supply of reliable agrochemical intermediate supplier quality materials, mitigating the risks associated with long synthetic chains.
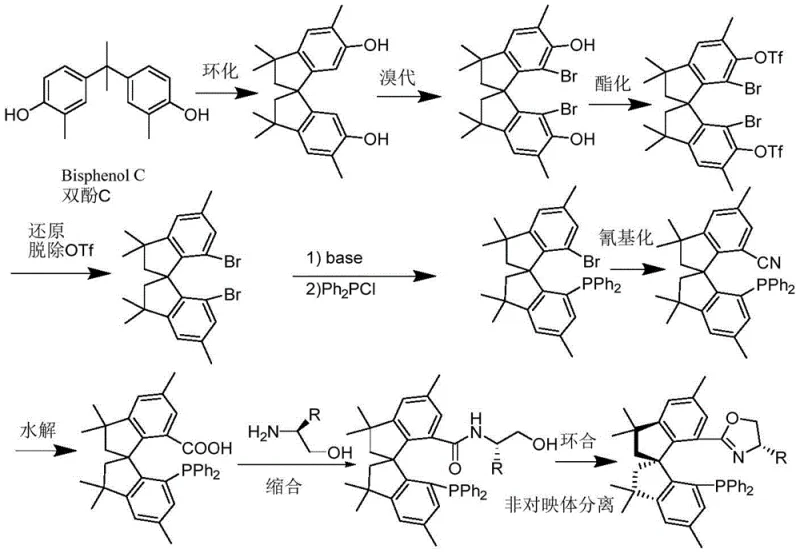
Mechanistic Insights into Tetramethyl Spiroindane Phosphine-Oxazoline Catalysis
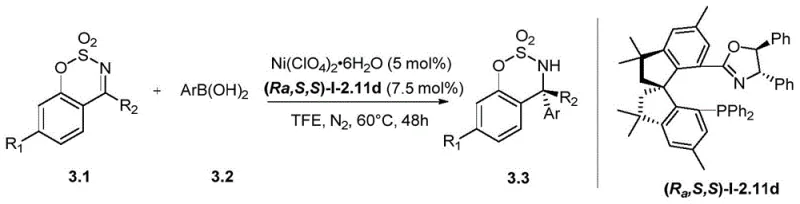
The catalytic efficacy of the new ligand system is deeply rooted in the precise electronic and structural factors engineered into the tetramethyl spiroindane backbone. The design considers the dihedral angle between the two indane rings, which has a profound influence on the enantioselectivity of asymmetric catalysis, as evidenced by extensive research in the field. The introduction of four methyl groups at the 3,3,3',3' positions creates a rigid chiral environment that effectively shields one face of the metal center during the catalytic cycle. This steric bulk forces the substrate to approach in a specific orientation, thereby maximizing the enantiomeric excess (ee) of the product, often reaching levels as high as 99% ee in nickel-catalyzed asymmetric arylation reactions. The rigidity of the skeleton prevents conformational flexibility that could lead to non-selective background reactions, ensuring that the chiral information is faithfully transferred from the ligand to the product. For R&D teams, understanding this mechanistic nuance is crucial for optimizing reaction conditions and expanding the scope to new substrates without compromising purity.
Impurity control is another critical aspect where this ligand system excels, primarily due to the stability of the spiro skeleton and the selectivity of the reaction steps. The absence of active arylmethylene groups in the spiro skeleton prevents unwanted side reactions such as polymerization or decomposition under harsh reaction conditions. The synthesis route incorporates specific purification steps, such as silica gel flash column chromatography and recrystallization, which effectively remove metal residues and organic byproducts. The ability to introduce electron-withdrawing groups on the phosphorus atom also modulates the electronic density at the metal center, reducing the likelihood of catalyst deactivation or decomposition. This results in a cleaner reaction profile with fewer byproducts, simplifying the downstream processing and reducing the burden on rigorous QC labs. The consistent structural integrity of the ligand ensures that the impurity profile remains predictable and manageable across different batches, which is essential for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Tetramethyl Spiroindane Phosphine-Oxazoline Ligand Efficiently
The synthesis of this high-performance ligand is designed for operational efficiency, leveraging standard organic transformation techniques that are well-understood in industrial settings. The process begins with the cyclization of bisphenol derivatives to form the tetramethyl spiroindane core, followed by selective bromination and phosphination to install the catalytic centers. Detailed standard operating procedures for each step, including temperature controls and reagent stoichiometry, are critical to maintaining the high enantioselectivity and yield reported in the patent. The final cyclization to form the oxazoline ring is particularly sensitive to reaction conditions, requiring precise control of base and activating agents to ensure the correct stereochemistry is established. Operators must adhere to strict anhydrous conditions during the phosphination and cyanation steps to prevent hydrolysis of sensitive intermediates. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the reaction parameters and workup procedures.
- Start with tetramethyl spiroindane diphenol derived from bisphenol condensation and perform cyclization.
- Execute bromination and esterification followed by palladium-catalyzed cyanation to introduce functional groups.
- Complete the synthesis via hydrolysis, condensation with aminoethanol, and final cyclization to form the oxazoline ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology addresses several pain points that have historically plagued the supply chain for chiral ligands and catalysts. The primary advantage is the drastic simplification of the raw material sourcing, as the process relies on commodity chemicals like bisphenols that are produced in millions of tons globally. This abundance ensures a stable supply chain and protects against price volatility that is common with specialized chiral pool materials. The reduction in synthetic steps directly translates to lower manufacturing costs, as fewer unit operations mean less energy consumption, reduced solvent usage, and lower labor requirements. For procurement managers, this means the ability to negotiate better pricing for high-purity pharmaceutical intermediates without sacrificing quality or performance. The robustness of the chemistry also implies a lower risk of batch failure, ensuring consistent delivery schedules and reducing the need for safety stock. These factors collectively contribute to a more resilient and cost-effective supply chain for fine chemical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolution steps and the use of cheap industrial bisphenols significantly lower the raw material costs associated with ligand production. By shortening the synthetic route from 13 steps to 9 steps, the process reduces the cumulative loss of material at each stage, thereby improving the overall mass balance and yield. This efficiency gain allows for substantial cost savings that can be passed down to the customer, making high-performance asymmetric catalysis more accessible for large-scale applications. The removal of transition metal catalysts in certain steps further reduces the cost burden associated with metal scavenging and waste treatment. Consequently, the total cost of ownership for this ligand system is markedly lower than that of conventional SPINOL-based ligands.
- Enhanced Supply Chain Reliability: Sourcing raw materials from established industrial supply chains, such as those for bisphenol A, ensures a high degree of reliability and continuity. Unlike specialized chiral starting materials that may have limited suppliers, commodity bisphenols are available from multiple global vendors, reducing the risk of supply disruption. The simplified synthesis route also means that production can be scaled up more rapidly to meet sudden increases in demand without the need for complex process re-engineering. This flexibility is crucial for maintaining reducing lead time for high-purity pharmaceutical intermediates in a dynamic market environment. Furthermore, the robust nature of the reaction conditions minimizes the risk of production delays caused by sensitive reagent handling or strict environmental controls.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing reactions that are amenable to large-scale batch processing without significant modification. The use of common solvents and reagents simplifies waste management and aligns with standard environmental compliance protocols in chemical manufacturing. The high selectivity of the catalyst reduces the formation of hazardous byproducts, minimizing the environmental footprint of the production process. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the costs associated with waste disposal and treatment. The ability to produce these ligands on a multi-ton scale ensures that they can support the commercial scale-up of complex polymer additives and other high-volume chemical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel ligand technology in industrial processes. These answers are derived directly from the patent data and practical experience in scaling similar catalytic systems. Understanding these details is essential for making informed decisions about adopting this technology for your specific manufacturing needs. The information provided here aims to clarify the operational benefits and technical capabilities of the tetramethyl spiroindane ligand system.
Q: What is the primary advantage of the tetramethyl spiroindane skeleton over traditional SPINOL ligands?
A: The tetramethyl spiroindane skeleton offers superior rigidity and a unique dihedral angle, enhancing enantioselectivity while utilizing cheaper, industrially available bisphenol starting materials.
Q: Can this ligand system support electron-withdrawing groups on the phosphorus atom?
A: Yes, unlike previous generations, this novel ligand design specifically accommodates electron-withdrawing substituents such as trifluoromethyl groups, expanding its catalytic scope.
Q: Is the synthesis route suitable for large-scale commercial production?
A: The 9-step route is significantly shorter than conventional 13-step methods, utilizing robust reactions that are amenable to scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphine-Oxazoline Ligand Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced patent technologies like CN108659045B into commercial reality for our global partners. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is seamless and efficient. We understand the critical importance of stringent purity specifications and rigorous QC labs in maintaining the integrity of your final products, especially in the pharmaceutical and fine chemical sectors. Our infrastructure is designed to handle the specific requirements of chiral ligand synthesis, including air-sensitive operations and precise temperature control. By partnering with us, you gain access to a wealth of technical expertise that can optimize your process for maximum yield and minimum cost.
We invite you to engage with our technical procurement team to discuss how this ligand technology can be integrated into your existing workflows. Request a Customized Cost-Saving Analysis to understand the specific economic benefits for your operation. We encourage you to contact us for specific COA data and route feasibility assessments to validate the performance of these ligands in your specific applications. Our commitment to transparency and technical excellence ensures that you receive the support needed to drive your projects forward successfully.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
