Breakthrough Purification Technology for High-Purity Valsartan and Commercial Scalability
Breakthrough Purification Technology for High-Purity Valsartan and Commercial Scalability
The global demand for antihypertensive agents continues to drive rigorous innovation in the manufacturing of Angiotensin II Receptor Blockers (ARBs), with Valsartan remaining a cornerstone therapy. However, the synthetic pathway for this critical active pharmaceutical ingredient often suffers from racemization, leading to the formation of unwanted D-type isomers that compromise drug efficacy and safety profiles. Addressing this persistent challenge, the technical disclosure in patent CN101768128B introduces a transformative refining methodology that leverages specific solvent engineering to achieve exceptional purity levels. This report analyzes the strategic implications of this technology for R&D directors and supply chain leaders seeking reliable valsartan supplier partnerships. By shifting away from traditional, yield-depleting recrystallization methods, this novel approach utilizes butanone-based systems to drastically reduce isomeric impurities while maintaining robust production throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Valsartan containing elevated levels of stereoisomeric impurities has relied heavily on repetitive recrystallization using ethyl acetate. This conventional workflow presents severe inefficiencies for large-scale manufacturing, particularly when the D-type isomer content exceeds 10% as detected by HPLC peak area analysis. To meet stringent pharmacopeial standards, manufacturers are often forced to subject the crude product to multiple cycles of dissolution and precipitation. Each cycle inevitably traps a portion of the desired L-valsartan within the mother liquor or excludes it from the crystal lattice, resulting in cumulative yield losses that severely impact the overall cost of goods sold. Furthermore, the extensive use of ethyl acetate requires significant energy input for solvent recovery and distillation, creating a bottleneck in production capacity and increasing the environmental footprint of the facility.
The Novel Approach
In stark contrast to legacy techniques, the methodology outlined in CN101768128B employs a sophisticated solvent system centered around butanone (methyl ethyl ketone), either as a standalone medium or in combination with esters and ethers. This innovative strategy capitalizes on the distinct solubility differentials between the target L-valsartan and the D-isomer impurity at controlled low temperatures. By utilizing butanone mixed with co-solvents such as isopropyl ether, ethyl acetate, or isopropyl acetate, the process achieves a highly selective crystallization environment. 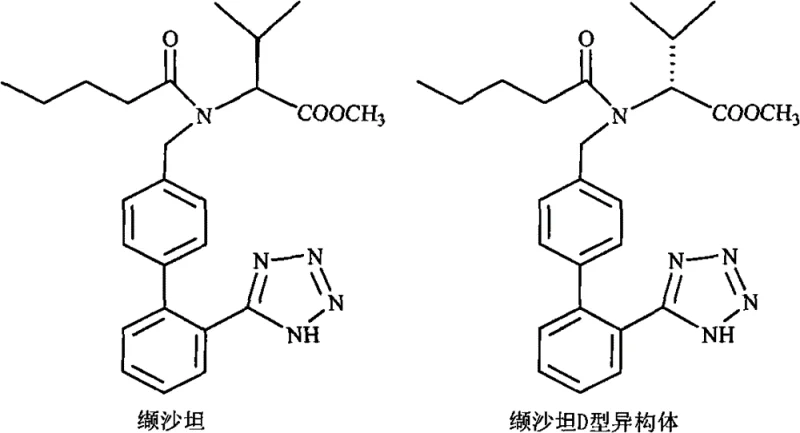 As illustrated in the structural comparison, the subtle stereochemical variance is effectively exploited by this solvent matrix, allowing the D-isomer to remain in solution while the pure L-form precipitates. This enables the reduction of isomer content from over 10% down to approximately 1.0% or lower in a single operational step, representing a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing.
As illustrated in the structural comparison, the subtle stereochemical variance is effectively exploited by this solvent matrix, allowing the D-isomer to remain in solution while the pure L-form precipitates. This enables the reduction of isomer content from over 10% down to approximately 1.0% or lower in a single operational step, representing a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Solvent-Mediated Chiral Resolution
The core mechanism driving this purification success lies in the thermodynamic and kinetic control of crystal growth within the butanone-based solvent matrix. When crude Valsartan is dissolved at elevated temperatures, typically around 40°C, both the L-enantiomer and the D-isomer impurity exist in a homogeneous solution phase. Upon cooling the system to sub-zero temperatures, specifically targeting -15°C, the solubility product of the L-valsartan is exceeded much more rapidly than that of the D-isomer due to the specific solvation shell interactions provided by the ketone and ether functional groups. The addition of non-polar or semi-polar co-solvents like isopropyl ether further depresses the solubility of the target compound, forcing rapid nucleation. Crucially, the lattice energy of the pure L-crystal form is favored under these specific conditions, effectively rejecting the sterically mismatched D-isomer molecules which remain solvated in the mother liquor. This selective exclusion is the fundamental driver behind the high purity specifications achieved without the need for chiral chromatography.
From an impurity control perspective, this mechanism offers a robust defense against batch-to-batch variability often seen in standard recrystallization. Traditional methods relying solely on ethyl acetate often struggle with occlusion, where impurity molecules get physically trapped within the growing crystal lattice. The butanone/isopropyl ether system appears to promote a cleaner crystal habit with slower, more ordered growth kinetics during the extended stirring period of approximately 5 hours. This prolonged agitation at low temperature ensures that any initially co-precipitated impurities have sufficient time to redissolve back into the solvent phase, a process known as Ostwald ripening. Consequently, the final solid product exhibits a significantly cleaner impurity profile, with HPLC data confirming D-isomer levels dropping to as low as 0.28% in optimized embodiments. This level of control is essential for producing high-purity pharmaceutical intermediates that meet the rigorous regulatory requirements of major markets.
How to Synthesize Valsartan Efficiently
Implementing this refining protocol requires precise adherence to solvent ratios and thermal profiles to maximize both purity and recovery. The process begins by charging the crude Valsartan material, which may contain greater than 10% D-isomer, into a reaction vessel equipped with efficient cooling capabilities. The choice of solvent mixture is critical; while pure butanone is effective, the patent highlights that mixing butanone with isopropyl ether or ethyl acetate in specific volume ratios (such as 1:1 or 1:4) can fine-tune the solubility curve for optimal yield. Once the material is fully dissolved at 40°C, the mixture is subjected to a controlled cooling ramp down to -15°C, followed by a sustained stirring period to ensure complete crystallization. For a comprehensive understanding of the operational parameters, the detailed standardized synthesis steps are provided in the guide below.
- Dissolve crude Valsartan containing >10% D-isomer in butanone at elevated temperature (approx. 40°C).
- Optionally add co-solvents such as isopropyl ether, ethyl acetate, or isopropyl acetate to optimize solubility profiles.
- Cool the solution to -15°C to induce crystallization, filter the solid, and dry under vacuum to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this butanone-based refining technology translates directly into enhanced operational efficiency and reduced supply risk. The primary economic driver is the drastic simplification of the purification workflow. By eliminating the need for multiple recrystallization cycles, manufacturers can significantly reduce the total processing time per batch, thereby increasing the throughput of existing production assets without requiring capital expenditure on new equipment. Furthermore, the ability to process crude materials with high impurity loads means that upstream synthesis steps do not need to be overly conservative to avoid racemization, allowing for more aggressive and cost-effective reaction conditions earlier in the value chain. This flexibility creates a more resilient supply chain capable of absorbing variations in raw material quality without compromising the final API specification.
- Cost Reduction in Manufacturing: The shift from repetitive ethyl acetate washing to a single-step butanone crystallization offers substantial cost savings through improved material utilization. In traditional processes, the cumulative loss of product across multiple purification stages can be financially devastating, especially when dealing with high-value intermediates. By achieving high purity in one pass, the overall yield of the process is preserved, meaning less starting material is required to produce the same amount of saleable API. Additionally, butanone and isopropyl ether are commodity solvents with favorable pricing structures compared to specialized chiral resolving agents, and their recovery via distillation is energy-efficient due to their relatively low boiling points, further driving down the variable cost per kilogram.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex purification bottlenecks that limit production speed. This technology mitigates such risks by streamlining the downstream processing phase. The robustness of the solvent system allows for the successful refining of 'off-spec' batches that would otherwise be scrapped or sent for expensive rework. This capability acts as an insurance policy for the supply chain, ensuring that minor deviations in the synthetic route do not lead to stockouts. Moreover, the solvents used are widely available in the global chemical market, reducing the risk of supply disruptions associated with niche or proprietary reagents, thus securing a stable production schedule for long-term contracts.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward because it relies on standard unit operations such as dissolution, cooling crystallization, and filtration, which are easily replicated in large-scale reactors. The reduction in solvent volume and the elimination of multiple wash steps also contribute to a smaller environmental footprint, aligning with modern green chemistry principles. Less solvent waste translates to lower disposal costs and reduced regulatory burden regarding volatile organic compound (VOC) emissions. This environmental efficiency is increasingly becoming a key criterion for selection by top-tier pharmaceutical companies who prioritize sustainable manufacturing partners in their vendor qualification audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this process into their current manufacturing lines. The answers below clarify the scope of applicability and the expected performance metrics based on the disclosed embodiments.
Q: What is the primary advantage of using butanone over ethyl acetate for Valsartan refinement?
A: According to patent CN101768128B, using butanone or butanone-based mixed solvents allows for the reduction of D-isomers to below 1.0% in a single crystallization step, whereas traditional ethyl acetate methods often require multiple repetitive recrystallizations, leading to significant yield loss and increased processing time.
Q: Can this purification method handle crude material with very high isomer content?
A: Yes, the technology is specifically designed to process Valsartan containing more than 10% D-type isomer impurities. The optimized solvent system effectively separates the desired L-enantiomer from the racemized byproducts, achieving high purity levels even from heavily contaminated starting materials.
Q: What yields can be expected from this refining process?
A: The patent data indicates that yields can exceed 50% even when processing difficult mother liquors or crude solids with high impurity loads. Specific embodiments demonstrate yields ranging from approximately 39% to over 65%, depending on the specific solvent mixture and cooling parameters employed.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valsartan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that advanced purification technologies like the one described in CN101768128B are implemented with precision. We understand that achieving stringent purity specifications requires not just the right chemistry, but also rigorous QC labs and state-of-the-art analytical capabilities to monitor isomer levels in real-time. Our infrastructure is designed to support the complex solvent management and temperature control required for this butanone-based crystallization, guaranteeing a consistent supply of high-quality Valsartan intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this refined manufacturing approach can optimize your specific supply chain requirements. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how yield improvements translate to bottom-line value. We encourage you to contact us to request specific COA data from our recent batches and to receive detailed route feasibility assessments. Let us collaborate to secure a stable, cost-effective, and high-purity supply of Valsartan for your global pharmaceutical portfolio.
