Optimized Industrial Synthesis of Olaparib Intermediates for High-Purity API Manufacturing
Optimized Industrial Synthesis of Olaparib Intermediates for High-Purity API Manufacturing
The pharmaceutical landscape for oncology treatments continues to evolve rapidly, with Poly (ADP-ribose) polymerase (PARP) inhibitors representing a cornerstone in the management of BRCA-mutated advanced ovarian cancers. Central to this therapeutic class is Olaparib, chemically defined as 4-[3-(4-cyclopropanecarbonyl-piperazine-1-carbonyl)-4-fluoro-benzyl]-2H-phthalazin-1-one. The efficient and scalable production of this potent active pharmaceutical ingredient (API) relies heavily on the robustness of its synthetic pathway. Patent CN106928149B discloses a groundbreaking preparation method that addresses critical bottlenecks in yield, purity, and process controllability. This technical insight report analyzes the proprietary advancements detailed in the patent, offering a comprehensive evaluation for R&D directors and supply chain stakeholders seeking reliable optimization strategies for complex heterocyclic synthesis.
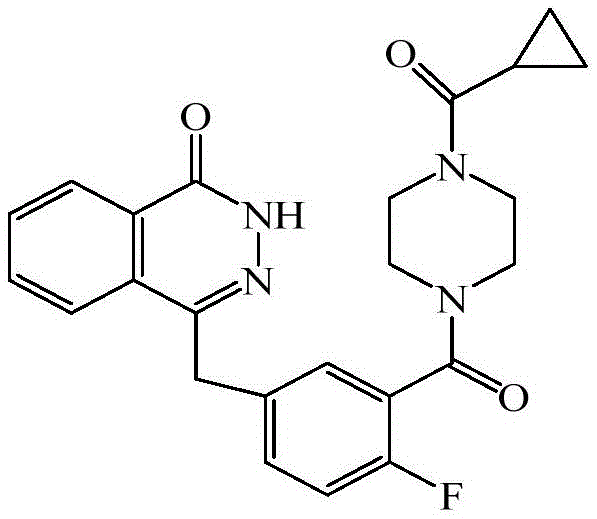
The structural complexity of Olaparib necessitates a multi-step synthesis where the integrity of the piperazine ring and the phthalazinone core must be meticulously preserved. The patent introduces a refined two-step sequence focusing on the conversion of the protected precursor (Compound C) to the key intermediate (Compound B), followed by the final acylation to yield the target API. This approach not only streamlines the operational workflow but also fundamentally alters the impurity profile, ensuring that the final drug substance meets the stringent regulatory requirements for global distribution.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in patent CN102627611A, have historically relied on harsh acidic conditions using ethanol-containing solutions and concentrated hydrochloric acid for the deprotection of the piperazine nitrogen. While chemically feasible, these legacy protocols suffer from significant drawbacks that hinder industrial viability. The reaction environment is often violent and difficult to control, leading to inconsistent batch-to-batch reproducibility. Furthermore, the post-treatment procedures are excessively cumbersome, requiring multiple extraction steps—often up to four cycles with dichloromethane—to isolate the intermediate. This not only increases solvent consumption and waste generation but also results in substantial product loss, capping the isolated yield of Intermediate B at approximately 58.5% with a purity of only 94.12%. Such low purity levels propagate through the synthesis, generating difficult-to-remove impurities in the final API stage.
The Novel Approach
In stark contrast, the methodology disclosed in CN106928149B employs a sophisticated acidolysis strategy utilizing trifluoroacetic acid (TFA) in dichloromethane. This solvent system provides a much milder and more controllable reaction environment, effectively mitigating the risks associated with exothermic runaway reactions. The innovation extends beyond the reaction conditions to the workup procedure; the novel process introduces a critical filtration step prior to solvent extraction. By adding water to the reaction residue, filtering out insoluble materials, and then extracting the filtrate with specific solvents like n-hexane or butyl acetate, the process achieves a dramatic improvement in efficiency. This streamlined workflow eliminates the need for repetitive extractions, significantly reducing processing time and solvent load while boosting the yield of Intermediate B to over 82% with purity levels exceeding 96%.
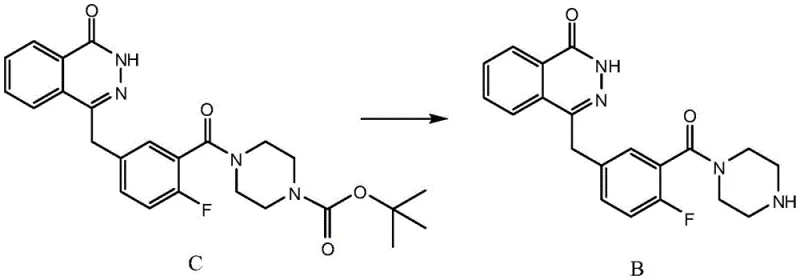
Mechanistic Insights into Acidolysis Deprotection and Impurity Control
The core of this technological advancement lies in the precise manipulation of the deprotection mechanism. The conversion of Compound C to Compound B involves the cleavage of a tert-butyl carbamate (Boc) protecting group from the piperazine ring. In the optimized protocol, trifluoroacetic acid acts as a potent proton source that facilitates the formation of a tert-butyl cation, which subsequently eliminates as isobutylene, leaving behind the free amine as a trifluoroacetate salt. The choice of dichloromethane as the co-solvent is crucial, as it maintains the solubility of the organic substrate while allowing for effective interaction with the acid. The patent specifies a molar ratio of Compound C to TFA ranging from 1:9 to 1:14, with a preferred range of 1:10 to 1:12. This stoichiometric excess ensures complete conversion without driving side reactions that could degrade the sensitive phthalazinone scaffold.
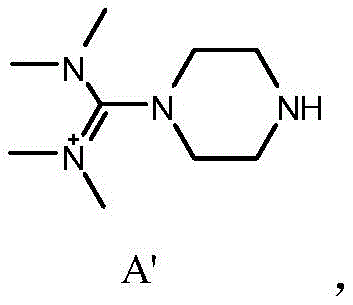
A critical aspect of this synthesis is the suppression of Impurity A', a structurally related byproduct that poses significant challenges during final purification. As illustrated in the structural analysis, Impurity A' arises from incomplete deprotection or alternative reaction pathways that are favored under the harsher conditions of conventional methods. The new process effectively minimizes the formation of this species through the optimized extraction protocol. By filtering the aqueous mixture before extraction, particulate matter and potential nucleation sites for impurity precipitation are removed. Subsequent extraction with non-polar solvents like n-hexane selectively partitions organic impurities away from the desired intermediate, which remains in the aqueous phase until basification. This orthogonal separation strategy ensures that the final Intermediate B contains negligible amounts of Impurity A', typically less than 0.2% when butyl acetate is used, thereby simplifying the downstream purification of the final Olaparib API.
How to Synthesize Olaparib Intermediate Efficiently
The implementation of this synthesis route requires strict adherence to the specified operational parameters to replicate the high yields and purity reported in the patent data. The process is designed to be robust, utilizing readily available industrial reagents while incorporating specific unit operations that are critical for success. The following overview outlines the strategic flow of the synthesis, emphasizing the critical control points identified during the development of this technology. For laboratory or pilot plant execution, precise temperature control during the acidolysis step and careful management of the pH during the basification phase are paramount to ensuring consistent quality.
- Dissolve compound C in dichloromethane and react with trifluoroacetic acid (molar ratio 1: 10-12) at 10-30°C for 3 hours to remove the protecting group.
- Evaporate solvents, add water, filter to remove insolubles, and extract the filtrate with n-hexane or butyl acetate to separate organic impurities.
- Adjust the aqueous phase pH to 8-10 with ammonia water, filter the resulting precipitate, and dry to obtain intermediate B with >96% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this optimized synthesis route offers tangible benefits that extend beyond mere technical metrics. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced throughput. By eliminating the need for four successive extraction cycles and replacing them with a single filtration and extraction sequence, the process significantly lowers solvent consumption and waste disposal costs. This reduction in material intensity not only improves the environmental footprint of the manufacturing site but also decreases the logistical burden associated with solvent procurement and storage.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substantial increase in yield and the reduction in processing steps. Achieving yields of over 80% for the key intermediate, compared to the sub-60% yields of legacy methods, means that significantly less raw material is required to produce the same amount of final API. Furthermore, the elimination of complex purification stages reduces energy consumption and labor hours. The use of cost-effective solvents like n-hexane and dichloromethane, which are standard commodities in the fine chemical industry, ensures that the variable costs remain stable and predictable, avoiding the volatility associated with specialized or exotic reagents.
- Enhanced Supply Chain Reliability: Supply continuity is a critical concern for any long-term API project. This synthesis route relies on robust, commercially available starting materials and reagents that are not subject to the supply constraints often seen with specialized catalysts or chiral auxiliaries. The high controllability of the reaction conditions reduces the risk of batch failures, which can otherwise lead to significant delays in production schedules. By stabilizing the quality of Intermediate B, the process ensures a consistent feed for the final acylation step, thereby smoothing out the overall production timeline and reducing the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The design of this process inherently supports commercial scale-up. The avoidance of violent exothermic reactions and the use of standard unit operations like filtration and liquid-liquid extraction make the technology easily transferable from kilogram to metric-ton scales. Additionally, the improved purity profile reduces the need for extensive recrystallization or chromatographic purification in later stages, which minimizes the generation of hazardous waste streams. This alignment with green chemistry principles facilitates easier regulatory compliance and supports the sustainability goals of modern pharmaceutical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Olaparib synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the practical aspects of adopting this technology for industrial production.
Q: How does the new acidolysis method improve impurity profiles compared to conventional HCl/Ethanol methods?
A: The novel method utilizes trifluoroacetic acid in dichloromethane rather than hydrochloric acid in ethanol, which significantly suppresses the formation of Impurity A'. By optimizing the extraction protocol with a filtration step prior to solvent extraction, the process achieves intermediate purities exceeding 96%, compared to roughly 94% in legacy methods.
Q: What represents the critical control point for maximizing yield in this synthesis?
A: The molar ratio of trifluoroacetic acid to the starting material is critical. Maintaining a ratio between 1:9 and 1:14 ensures complete deprotection without excessive side reactions. Additionally, the specific sequence of adding water, filtering, and then extracting with non-polar solvents like n-hexane prevents emulsification and product loss.
Q: Is this process suitable for large-scale commercial production of Olaparib?
A: Yes, the patent explicitly highlights the method's suitability for industrial mass production. The use of common industrial solvents like dichloromethane and n-hexane, combined with simplified workup procedures that avoid complex multi-stage extractions, makes the process highly scalable and cost-effective for metric-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olaparib Supplier
The technical advancements detailed in patent CN106928149B represent a significant leap forward in the manufacturing of PARP inhibitors, yet translating laboratory success to commercial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors and rigorous QC labs capable of handling the specific solvent systems and temperature controls required for this acidolysis process. We understand that maintaining stringent purity specifications is non-negotiable in the oncology sector, and our quality management systems are designed to ensure every batch meets the highest international standards.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthesis capabilities, we can help you secure a stable supply of high-quality Olaparib intermediates while driving down your overall cost of goods. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering excellence can become a competitive advantage for your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
