Advanced Catalytic Synthesis of Dextromethorphan Intermediates for Scalable Pharmaceutical Manufacturing
Introduction to Next-Generation Dextromethorphan Intermediate Synthesis
The pharmaceutical industry continuously seeks more efficient pathways for producing high-value active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is detailed in patent CN111333573A, which discloses a novel preparation process for the key dextromethorphan intermediate, (S)-1-(4-methoxy)benzyl-1,2,3,4,5,6,7,8-octahydroisoquinoline salt. This technology represents a paradigm shift from traditional resolution methods to a highly stereoselective asymmetric hydrogenation approach. By leveraging a sophisticated catalytic system comprising a metal M complex (specifically Rhodium or Iridium), a specialized chiral P-N ligand, and an iodine-containing additive, the process achieves exceptional enantiomeric excess values reaching up to 99% ee. This advancement is critical for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with minimal environmental impact.
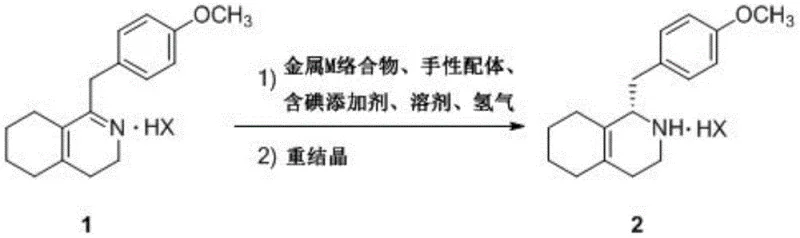
The core innovation lies in the synergistic effect of the catalyst components, which allows the reaction to proceed under relatively mild conditions ranging from 10-80°C and 1.0-6.0 MPa hydrogen pressure. Unlike older methodologies that struggled with low yields or required harsh reagents, this protocol ensures a robust conversion rate, often exceeding 90% yield after recrystallization. For R&D directors and process chemists, this translates to a streamlined workflow that reduces the complexity of downstream purification. The ability to directly synthesize the desired (S)-enantiomer with such precision eliminates the need for wasteful chiral resolution steps, thereby aligning perfectly with modern green chemistry principles and cost reduction in API manufacturing initiatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of dextromethorphan intermediates relied heavily on two primary strategies, both of which present significant bottlenecks for modern supply chains. The first method, chiral resolution of racemates, is inherently inefficient because it theoretically caps the maximum yield at 50%, generating an equimolar amount of unwanted (R)-enantiomer waste that must be disposed of or recycled at great expense. Furthermore, this approach often requires multiple crystallization cycles to achieve acceptable optical purity, leading to substantial solvent consumption and extended production timelines. The second conventional approach involves asymmetric hydrogenation using earlier generation catalysts, such as chiral ferrocene bisphosphine ligands employed by companies like Lonza. While effective, these ligands are notoriously difficult to synthesize, sensitive to air and moisture, and expensive to procure, creating a fragile supply chain vulnerable to raw material fluctuations.
The Novel Approach
The methodology outlined in CN111333573A effectively dismantles these barriers by introducing a catalytic system based on chiral phosphoramidite ligands derived from BINOL scaffolds. These P-N ligands are structurally distinct from the bulky bisphosphines of the past, offering superior stability and ease of handling without compromising catalytic performance. The process integrates an iodine-containing additive, which plays a pivotal role in modulating the electronic properties of the metal center, thereby accelerating the hydrogenation kinetics. This novel approach not only boosts the enantiomeric excess to levels comparable to or exceeding the best historical benchmarks (up to 99% ee) but also simplifies the operational procedure. By avoiding the use of air-sensitive, complex bisphosphine ligands, the new method significantly lowers the barrier to entry for commercial scale-up of complex pharmaceutical intermediates, ensuring a more resilient and cost-effective production model.
Mechanistic Insights into Rh/Ir-Catalyzed Asymmetric Hydrogenation
At the heart of this transformative synthesis is a finely tuned catalytic cycle driven by Rhodium or Iridium complexes coordinated with chiral P-N ligands. The mechanism begins with the formation of an active cationic metal-hydride species upon exposure to hydrogen gas in the presence of the iodine additive. The iodine source, whether elemental iodine or salts like potassium iodide, acts as a crucial promoter that likely facilitates the heterolytic cleavage of hydrogen or stabilizes the active catalytic species against deactivation. The substrate, 1-(4-methoxy)benzyl-3,4,5,6,7,8-hexahydroisoquinoline salt, coordinates to the metal center through its olefinic bond and potentially the nitrogen atom, positioning itself within the chiral pocket created by the bulky binaphthyl backbone of the ligand.
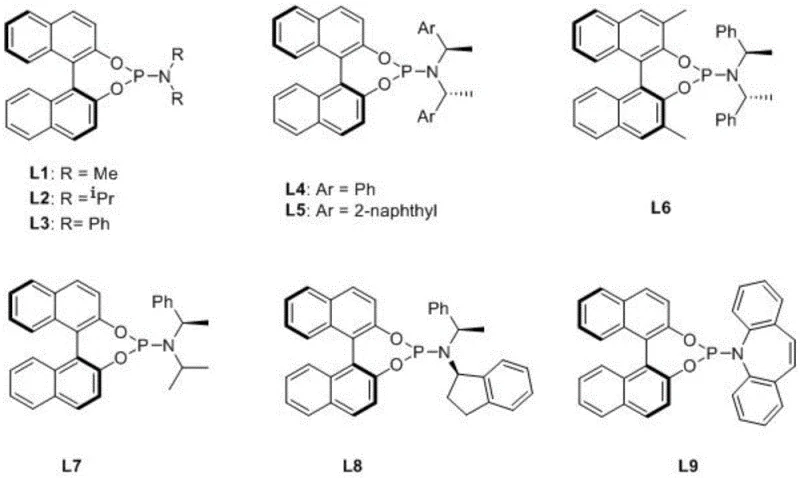
The stereochemical outcome is dictated by the specific spatial arrangement of the substituents on the ligand's nitrogen and phosphorus atoms, as illustrated by the diverse library of ligands (L1-L9) available for optimization. As the hydride transfer occurs, the chiral environment forces the hydrogen addition to occur exclusively from one face of the prochiral double bond, resulting in the formation of the (S)-configured product with high fidelity. Following the hydrogenation step, the catalyst releases the saturated octahydroisoquinoline product and regenerates to enter the next cycle. This mechanistic robustness allows the reaction to tolerate a variety of solvents, including tetrahydrofuran, dichloromethane, and alcohols, providing process chemists with the flexibility to optimize solubility and workup procedures. The subsequent recrystallization step further amplifies the optical purity, acting as a final polishing stage to ensure the final salt meets stringent pharmaceutical specifications.
How to Synthesize (S)-1-(4-methoxy)benzyl-1,2,3,4,5,6,7,8-octahydroisoquinoline Salt Efficiently
Implementing this synthesis requires careful attention to the molar ratios of the catalyst components and the control of atmospheric conditions. The patent specifies that the metal complex should be used in catalytic amounts relative to the substrate, typically ranging from 1:100 to 1:100000, demonstrating the high turnover potential of the system. The chiral ligand and iodine additive are introduced in stoichiometric proportions relative to the metal to ensure the formation of the active catalytic species. The reaction is conducted in a sealed pressure vessel where air is thoroughly purged and replaced with hydrogen to prevent catalyst oxidation. Detailed standardized synthesis steps see the guide below.
- Charge the reactor with the hexahydroisoquinoline substrate, metal catalyst (Rh/Ir), chiral ligand, iodine additive, and solvent under inert atmosphere.
- Perform asymmetric hydrogenation at 10-80°C and 1.0-6.0 MPa hydrogen pressure for 2-24 hours to achieve high conversion.
- Concentrate the reaction mixture and recrystallize the crude product from alcohol solvents at 50-60°C to obtain high-purity salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits that extend beyond simple yield improvements. The shift towards a more robust catalytic system directly addresses the volatility often associated with specialty chemical sourcing. By utilizing ligands that are easier to prepare and more stable than their predecessors, manufacturers can reduce dependency on niche suppliers who may have long lead times or inconsistent quality. This stability translates into a more predictable production schedule, allowing for better inventory management and reduced risk of stockouts for critical downstream API production. Furthermore, the simplified workflow minimizes the number of unit operations required, which inherently lowers the operational expenditure associated with labor and equipment utilization.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps removes the inherent 50% yield loss associated with racemic separation, effectively doubling the theoretical output from the same amount of starting material. Additionally, the use of stable, easily synthesized P-N ligands instead of expensive, air-sensitive bisphosphines drastically reduces the raw material cost per kilogram of product. The mild reaction conditions also contribute to energy savings, as there is no need for extreme cryogenic temperatures or excessive heating, leading to substantial cost savings in utility consumption over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The robustness of the catalyst system against impurities and its tolerance for a wide range of solvents means that production is less likely to be halted due to minor variations in raw material quality. This resilience is crucial for maintaining continuous supply lines in a global market. Moreover, the high selectivity of the reaction reduces the formation of difficult-to-remove impurities, simplifying the purification process and ensuring that the final product consistently meets high-purity pharmaceutical intermediate standards without requiring extensive reprocessing.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, operating at pressures and temperatures that are standard in industrial hydrogenation reactors. This compatibility allows for seamless technology transfer from pilot plant to full commercial production without the need for specialized, custom-built equipment. From an environmental perspective, the high atom economy of the hydrogenation reaction and the reduction in solvent usage for purification align with increasingly strict regulatory requirements for waste disposal, facilitating smoother regulatory approvals and reducing the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on performance metrics and operational parameters. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of the P-N ligand system over traditional bisphosphine ligands?
A: The P-N ligand system described in CN111333573A offers superior stability and easier preparation compared to cumbersome ferrocene or biphenyl bisphosphine ligands, while maintaining high enantioselectivity (up to 99% ee).
Q: How does the iodine additive impact the reaction efficiency?
A: The inclusion of iodine-containing additives (such as KI or I2) is critical for activating the metal complex and enhancing both the reaction rate and stereoselectivity during the hydrogenation process.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions (10-80°C, 1.0-6.0 MPa) and robust catalysts, making it highly amenable to commercial scale-up with consistent quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dextromethorphan Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic and patent research into viable commercial realities. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the CN111333573A process are executed with precision and reliability. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels that meet or exceed global pharmacopoeia standards. We understand that consistency is key in the pharmaceutical supply chain, and our quality management systems are designed to deliver batch-after-batch reproducibility.
We invite global partners to collaborate with us to leverage this cutting-edge synthesis route for their dextromethorphan supply needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to reach out today to obtain specific COA data from our recent pilot runs and comprehensive route feasibility assessments that demonstrate how we can optimize your supply chain for efficiency and cost-effectiveness.
