Advanced Electrochemical Synthesis of Chloroethyl Sulfoxides for Commercial Pharmaceutical Manufacturing
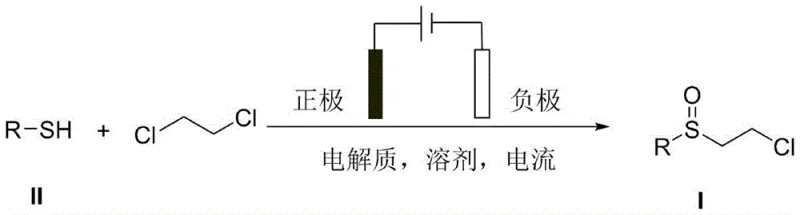
The pharmaceutical and fine chemical industries are constantly seeking sustainable and efficient pathways to synthesize critical intermediates, particularly sulfoxide derivatives which serve as key structural motifs in numerous active pharmaceutical ingredients (APIs). Patent CN110846676A introduces a groundbreaking electrochemical synthesis method for chloroethyl sulfoxide compounds, offering a transformative alternative to conventional oxidation protocols. This technology leverages constant current electrolysis in an undivided cell to directly couple aryl thiophenols with 1,2-dichloroethane, bypassing the need for stoichiometric metal catalysts or hazardous chemical oxidants. For R&D directors and procurement specialists, this represents a significant opportunity to streamline supply chains for quinolone antibiotics like sitafloxacin and proton pump inhibitors. By replacing traditional reagents with electrons, the process not only enhances atomic economy but also mitigates the environmental burden associated with heavy metal waste disposal, positioning it as a highly attractive route for modern green manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of sulfoxide derivatives has relied heavily on the oxidation of sulfides or thiols using stoichiometric amounts of transition metal complexes and strong chemical oxidants. Prominent literature, such as studies by the Brinksma and Velusamy groups, details the use of Mn(III) or Cu(II)络合物配合物 with hydrogen peroxide to achieve these transformations. While effective on a small scale, these methods suffer from inherent sustainability drawbacks, including the generation of substantial metal-containing waste streams that require complex and costly removal processes to meet pharmaceutical purity standards. Furthermore, the reliance on specific nano-mesoporous materials or gold-loaded catalysts introduces significant supply chain vulnerabilities and cost volatility. The multi-step nature of traditional routes, often involving separate sulfide formation followed by oxidation under harsh conditions, exacerbates these inefficiencies, leading to lower overall yields and increased operational expenditure for large-scale manufacturing facilities.
The Novel Approach
In stark contrast, the electrochemical methodology disclosed in the patent utilizes electrical energy as the primary driving force for the oxidative coupling reaction, effectively rendering the process catalyst-free and oxidant-free. By dissolving aryl thiophenol compounds and 1,2-dichloroethane in a solvent system containing a supporting electrolyte, the reaction proceeds smoothly under constant current conditions within a simple undivided cell setup. This approach eliminates the need for expensive and potentially toxic metal catalysts, thereby simplifying the workup procedure to a straightforward column chromatography separation. The operational simplicity is further enhanced by the mild reaction conditions, typically ranging from 30°C to 70°C, which reduce energy consumption compared to high-thermal processes. This paradigm shift from chemical reagents to electrochemical activation offers a robust, scalable, and environmentally benign pathway for producing high-purity chloroethyl sulfoxides essential for advanced drug synthesis.
Mechanistic Insights into Electrochemical Oxidative Coupling
The core of this innovation lies in the anodic oxidation mechanism where the thiol substrate undergoes electron transfer at the electrode surface to generate reactive sulfur-centered radical species. These intermediates subsequently interact with 1,2-dichloroethane, facilitating the formation of the sulfur-carbon bond and the simultaneous introduction of the oxygen atom to yield the sulfoxide functionality. The use of an undivided cell allows for efficient ion transport while maintaining the necessary potential gradient to drive the reaction forward without the complexity of membrane separation. Crucially, the electrochemical parameters, such as current density and electrolyte concentration, can be finely tuned to control the oxidation state of the sulfur atom, ensuring high selectivity for the sulfoxide over the thermodynamically more stable sulfone. This precise control is vital for pharmaceutical applications where impurity profiles must be strictly managed to comply with regulatory guidelines.
Furthermore, the choice of electrode materials, such as platinum or reticulated vitreous carbon (RVC), plays a pivotal role in minimizing side reactions and maximizing faradaic efficiency. The presence of water or mixed solvent systems can also influence the reaction kinetics, potentially participating in the oxygen transfer step or stabilizing intermediate species. By avoiding the use of external oxidants like peroxides, the risk of uncontrolled exothermic events is significantly reduced, enhancing process safety. The mechanistic pathway suggests a direct and atom-economical route where the chlorine atoms in the dichloroethane serve both as the alkylating agent and potentially as leaving groups or stabilizers, depending on the specific substitution pattern. This deep understanding of the electrochemical interface allows process chemists to optimize reaction conditions for diverse substrates, ensuring consistent quality and yield across different batches of chloroethyl sulfoxide intermediates.
How to Synthesize Chloroethyl Sulfoxide Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible framework for generating chloroethyl sulfoxide derivatives with high purity and yield. The process begins with the preparation of a homogeneous reaction mixture containing the aryl thiophenol substrate, 1,2-dichloroethane as both reactant and solvent component, and a quaternary ammonium salt electrolyte. This solution is then subjected to constant current electrolysis using inert electrodes under a controlled atmosphere, allowing the oxidative coupling to proceed over a defined period. The detailed standardized synthesis steps below outline the specific parameters for electrode selection, current settings, and workup procedures required to achieve optimal results in a laboratory or pilot plant setting.
- Dissolve aryl thiophenol compounds, 1,2-dichloroethane, and a supporting electrolyte such as tetrabutylammonium tetrafluoroborate in an organic solvent to form the reaction solution.
- Transfer the solution into an undivided electrolytic cell, insert appropriate electrodes (e.g., Platinum-RVC), and establish a constant current between 10mA and 60mA under a controlled gas atmosphere.
- Stir the mixture at a temperature range of 30°C to 70°C for 3 to 12 hours, then isolate the final chloroethyl sulfoxide product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of precious metal catalysts removes a major cost driver and supply bottleneck, as there is no longer a need to source, recover, or dispose of expensive transition metals. This simplification of the bill of materials directly translates to reduced raw material costs and lower inventory holding requirements. Additionally, the streamlined one-pot nature of the reaction reduces the number of unit operations, leading to shorter cycle times and increased throughput capacity in existing manufacturing infrastructure. These factors collectively contribute to a more resilient and cost-effective supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most significant financial advantage stems from the complete removal of stoichiometric metal catalysts and chemical oxidants from the process. Traditional methods often require costly manganese or copper complexes and hydrogen peroxide, which not only add to the direct material cost but also necessitate expensive downstream purification steps to remove metal residues to ppm levels. By utilizing electricity as the reagent, the variable cost per kilogram of product is drastically lowered, and the capital expenditure associated with metal recovery systems is eliminated. This structural cost advantage allows for more competitive pricing strategies and improved margin protection against fluctuations in metal commodity markets.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as aryl thiophenols and 1,2-dichloroethane ensures a stable and diversified supply base, reducing the risk of disruptions associated with specialized catalyst suppliers. Since the process does not depend on proprietary nano-materials or rare earth elements, sourcing is straightforward and can be easily scaled to meet increasing demand without long lead times. The robustness of the electrochemical setup also means that production can be maintained consistently even if specific reagent grades vary slightly, providing greater flexibility in vendor selection and procurement negotiations for bulk raw materials.
- Scalability and Environmental Compliance: Electrochemical processes are inherently scalable, as increasing production capacity often involves adding more electrode surface area or cells in parallel rather than redesigning the entire chemical process. This modularity facilitates a smoother transition from pilot scale to commercial tonnage production. Moreover, the green nature of the technology, characterized by the absence of heavy metal waste and reduced solvent usage, aligns perfectly with increasingly stringent environmental regulations. This compliance reduces the administrative burden and costs associated with waste treatment and environmental reporting, making the facility more sustainable and socially responsible in the eyes of stakeholders and regulators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of chloroethyl sulfoxides, based on the detailed specifications and experimental data provided in the patent documentation. These insights are designed to clarify the operational feasibility and strategic value of implementing this technology within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and supply chain integration.
Q: What are the primary advantages of this electrochemical method over traditional oxidation?
A: Unlike traditional methods requiring stoichiometric metal catalysts like Mn(III) or Cu(II) complexes and chemical oxidants, this electrochemical approach uses electricity as a clean reagent. This eliminates heavy metal contamination risks, simplifies downstream purification, and significantly reduces chemical waste, aligning with strict pharmaceutical environmental standards.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the process utilizes an undivided electrolytic cell with readily available starting materials like aryl thiophenols and 1,2-dichloroethane. The absence of sensitive metal catalysts and the use of robust electrode materials facilitate easier scale-up from laboratory to commercial tonnage production without complex catalyst recovery systems.
Q: What is the chemical selectivity of this synthesis regarding over-oxidation?
A: The method demonstrates high chemical selectivity for the sulfoxide functionality, effectively preventing over-oxidation to the corresponding sulfone. By controlling parameters such as constant current density and reaction temperature, the process ensures the formation of the desired chloroethyl sulfoxide with minimal byproduct formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chloroethyl Sulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this electrochemical synthesis route for producing high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering consistent quality and reliability for complex molecules like chloroethyl sulfoxides, supporting your drug development timelines with precision and efficiency.
We invite you to collaborate with our technical team to explore how this metal-free synthesis can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your volume needs. Please contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial viability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
