Scalable Electrochemical Production of Chloroethyl Sulfoxide Intermediates for Global Pharmaceutical Supply Chains
Introduction to Advanced Electrochemical Oxidation Technology
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift towards greener, more sustainable synthetic methodologies, driven by the urgent need to reduce environmental footprints and enhance process safety. A pivotal development in this domain is disclosed in patent CN110846676B, which details a novel electrochemical synthesis method for chloroethyl sulfoxide compounds. These compounds serve as critical building blocks in the manufacture of high-value active pharmaceutical ingredients (APIs), including quinolone antibiotics and proton pump inhibitors. Unlike traditional routes that rely heavily on stoichiometric oxidants and transition metal catalysts, this innovative approach utilizes electrical energy to drive the oxidative coupling reaction directly. By operating in an undivided electrolytic cell, the process achieves high chemical selectivity while completely bypassing the use of hazardous reagents. This technological breakthrough not only aligns with modern green chemistry principles but also offers a robust pathway for the reliable pharmaceutical intermediate supplier networks to secure high-purity materials with improved operational efficiency.
The significance of this patent extends beyond mere academic interest; it addresses fundamental bottlenecks in the supply chain of complex organic intermediates. Sulfoxides are notoriously difficult to synthesize without over-oxidation to sulfones or the formation of disulfide byproducts. The electrochemical method described herein provides precise control over the oxidation state through the regulation of current density and potential. This level of control is essential for maintaining the stringent purity specifications required by global regulatory bodies. Furthermore, the versatility of the method allows for the functionalization of various aryl thiophenols, enabling the production of a diverse library of sulfoxide derivatives from a single, streamlined platform. For R&D directors and procurement managers alike, understanding the mechanics and commercial implications of this technology is vital for optimizing future manufacturing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of sulfoxide derivatives has been fraught with challenges related to reagent toxicity, waste generation, and process complexity. Traditional synthetic routes typically involve the oxidation of sulfides or thiols using chemical oxidants such as hydrogen peroxide, often in the presence of transition metal catalysts like manganese or copper complexes. For instance, prior art describes methods utilizing Mn(III) or Cu(II) complexes which, while effective, introduce significant downstream processing burdens. The necessity to remove trace metal residues from the final product is a major concern for pharmaceutical applications, requiring additional purification steps such as chelation or extensive chromatography. Moreover, these chemical oxidants are often used in stoichiometric excess to drive the reaction to completion, leading to substantial amounts of chemical waste that must be treated and disposed of safely. The multi-step nature of some conventional protocols, such as the two-step synthesis involving 1-chloro-2-bromoethane followed by oxidation, further exacerbates cost and time inefficiencies. These factors collectively contribute to a higher cost of goods sold (COGS) and increased environmental liability for manufacturers relying on legacy technologies.
The Novel Approach
In stark contrast, the electrochemical synthesis method outlined in the patent data presents a streamlined, one-pot solution that fundamentally reimagines the oxidation process. By employing electricity as the primary reagent, the method eliminates the need for external chemical oxidants and metal catalysts entirely. The reaction proceeds in an undivided cell where aryl thiophenols and 1,2-dichloroethane are dissolved in a solvent system containing a supporting electrolyte. Upon application of a constant current, the thiophenol undergoes anodic oxidation, facilitating a direct coupling with the dichloroethane to form the desired chloroethyl sulfoxide. This direct transformation is not only atom-economical but also significantly reduces the E-factor of the process by minimizing waste generation. The simplicity of the setup, requiring only standard electrode materials such as platinum or reticulated vitreous carbon (RVC), makes it highly adaptable for both laboratory-scale optimization and industrial-scale production. This novel approach effectively decouples the synthesis efficiency from the availability of expensive catalytic metals, thereby enhancing the resilience of the supply chain against market fluctuations in raw material costs.
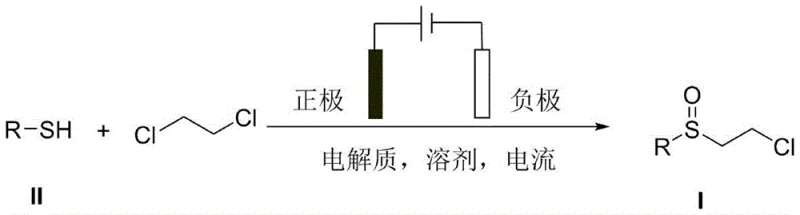
Mechanistic Insights into Electrochemical Oxidative Coupling
The core of this technology lies in the precise electrochemical activation of the sulfur center within the thiophenol substrate. Under the applied constant current, typically ranging from 10 mA to 60 mA, the thiol group at the anode surface loses electrons to generate a reactive sulfur-centered radical or cationic species. This activated intermediate then nucleophilically attacks the 1,2-dichloroethane molecule, displacing a chloride ion and forming a new carbon-sulfur bond. Simultaneously, the electrochemical potential is carefully tuned to ensure that the oxidation stops at the sulfoxide stage, preventing the thermodynamic sink of over-oxidation to the corresponding sulfone. This selectivity is a hallmark of electrochemical synthesis, where the reaction progress can be modulated simply by adjusting the charge passed through the system. The use of supporting electrolytes, such as tetrabutylammonium tetrafluoroborate, ensures sufficient conductivity in the organic solvent medium, facilitating efficient electron transfer without participating directly in the redox chemistry. This mechanism avoids the formation of metal-thiolate complexes that often plague traditional catalytic cycles, resulting in a cleaner reaction profile with fewer side products.
Furthermore, the reaction environment plays a crucial role in stabilizing the intermediates and driving the equilibrium towards the product. The patent specifies the use of solvents like 1,2-dichloroethane, which acts both as a reactant and a solvent, simplifying the reaction mixture composition. The presence of water or co-solvents like acetonitrile can be tolerated, adding flexibility to the process conditions. Temperature control, maintained optimally between 30°C and 70°C, is another critical parameter that influences the kinetics of the electron transfer and the stability of the generated sulfoxide. By avoiding harsh acidic or basic conditions often required in chemical oxidation, this method preserves sensitive functional groups on the aromatic ring, such as methoxy or fluoro substituents. This functional group tolerance is paramount for R&D teams designing complex drug molecules, as it allows for late-stage functionalization without the need for extensive protecting group strategies. The result is a highly chemoselective process that delivers high-purity intermediates suitable for immediate use in subsequent synthetic steps.
How to Synthesize Chloroethyl Sulfoxide Efficiently
Implementing this electrochemical protocol requires careful attention to the configuration of the electrolytic cell and the selection of operational parameters to maximize yield and purity. The process begins with the preparation of the reaction solution, where the molar ratios of the thiophenol substrate, 1,2-dichloroethane, and electrolyte are optimized to ensure complete conversion. The choice of electrode material is also significant; while platinum offers excellent stability and conductivity, cost-effective alternatives like graphite or RVC have demonstrated comparable performance in specific examples, providing options for cost-sensitive applications. Once the cell is assembled and the inert gas atmosphere (such as nitrogen or argon) is established to prevent unwanted side reactions with oxygen, the constant current power supply is engaged. Monitoring the reaction progress via thin-layer chromatography or HPLC allows operators to determine the optimal endpoint, typically achieved within 3 to 12 hours depending on the specific substrate and current density. Following the reaction, the workup procedure is remarkably straightforward, involving simple extraction and column chromatography to isolate the pure chloroethyl sulfoxide product, free from metal contaminants.
- Dissolve aryl thiophenol compounds, 1,2-dichloroethane, and a supporting electrolyte such as tetrabutylammonium tetrafluoroborate in a suitable organic solvent to form the reaction solution.
- Insert electrodes into the undivided electrolytic cell containing the reaction solution and apply a constant current between 10 mA and 60 mA while maintaining the temperature between 30°C and 70°C.
- After the reaction completes within 3 to 12 hours, isolate the target chloroethyl sulfoxide product through standard silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical synthesis method translates into tangible strategic advantages that extend far beyond the laboratory bench. The most immediate impact is observed in the realm of cost reduction in pharmaceutical intermediate manufacturing. By removing the dependency on precious metal catalysts such as palladium, platinum complexes, or specialized nano-materials, the raw material costs are significantly lowered. Furthermore, the elimination of these metals removes the need for expensive and time-consuming metal scavenging steps during purification, which traditionally account for a substantial portion of processing time and consumable costs. The simplified workflow also reduces the consumption of auxiliary chemicals and solvents associated with catalyst removal, leading to a leaner and more cost-effective production cycle. These savings can be passed down the supply chain, offering competitive pricing for high-quality intermediates without compromising on specification standards.
Enhanced supply chain reliability is another critical benefit derived from this technology. Traditional methods often face disruptions due to the volatility in the supply of specialized catalysts or oxidants, which may be subject to geopolitical constraints or manufacturing bottlenecks. In contrast, the reagents required for this electrochemical process—aryl thiophenols, 1,2-dichloroethane, and common electrolytes—are commodity chemicals with robust and diversified global supply bases. This abundance ensures a steady flow of materials, reducing the risk of production stoppages due to raw material shortages. Additionally, the scalability of electrochemical reactors is well-established, allowing for seamless transition from pilot scale to commercial tonnage production. The modular nature of electrochemical cells means that capacity can be increased by adding more units rather than building massive new infrastructure, providing agility in responding to market demand fluctuations. This flexibility is invaluable for maintaining continuity of supply in the fast-paced pharmaceutical sector.
Scalability and environmental compliance are increasingly becoming deciding factors in vendor selection, and this method excels in both areas. The absence of toxic heavy metals and stoichiometric oxidants drastically reduces the hazardous waste load, simplifying wastewater treatment and disposal procedures. This aligns perfectly with tightening environmental regulations globally, mitigating the risk of compliance penalties and enhancing the corporate sustainability profile of the manufacturer. The energy efficiency of the process, driven by direct electron transfer, further contributes to a lower carbon footprint compared to thermal oxidation methods. For supply chain leaders tasked with meeting corporate sustainability goals, sourcing intermediates produced via this green electrochemical route offers a clear pathway to reducing Scope 3 emissions. The combination of operational simplicity, regulatory compliance, and environmental stewardship makes this technology a superior choice for long-term strategic partnerships in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of chloroethyl sulfoxides, based on the detailed findings within the patent literature. These insights are intended to clarify the operational feasibility and strategic benefits of adopting this methodology for large-scale production. Understanding these nuances helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios. The answers reflect the specific advantages highlighted in the experimental data, focusing on purity, yield, and process robustness.
Q: What are the primary advantages of this electrochemical method over traditional oxidation?
A: This method eliminates the need for stoichiometric metal catalysts and toxic chemical oxidants like hydrogen peroxide, significantly reducing heavy metal contamination risks and simplifying downstream purification processes.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the use of an undivided cell with simple electrode materials like platinum or RVC allows for straightforward scale-up, avoiding the complex reactor engineering often required for heterogeneous catalytic systems.
Q: What is the substrate scope for this chloroethyl sulfoxide synthesis?
A: The protocol demonstrates high compatibility with various aryl thiophenols, including those with electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like fluoro and chloro, yielding diverse sulfoxide derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chloroethyl Sulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced electrochemical synthesis in delivering superior pharmaceutical intermediates to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN110846676B are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of chloroethyl sulfoxide meets the exacting standards required for API synthesis. We understand that consistency and quality are non-negotiable in the pharmaceutical supply chain, and our commitment to technical excellence ensures that our clients receive materials that facilitate smooth downstream processing and regulatory approval.
We invite forward-thinking pharmaceutical companies and chemical manufacturers to collaborate with us to leverage this cutting-edge technology for their specific project needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage you to reach out today to request specific COA data and route feasibility assessments for chloroethyl sulfoxide derivatives. Whether you are looking to optimize an existing supply chain or develop a new synthetic route for a complex drug candidate, our expertise in electrochemical manufacturing positions us as the ideal partner to drive your project forward with efficiency and reliability.
