Advanced One-Step Synthesis of Quinazolinone Derivatives for Commercial Scale-Up
Introduction to Patent CN111718301A Technology
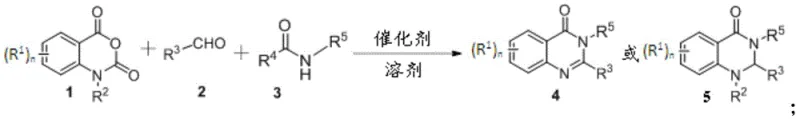
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access privileged scaffolds, and the recent disclosure in patent CN111718301A presents a transformative approach to synthesizing quinazolinone derivatives. This specific intellectual property details a robust, one-step multicomponent reaction that utilizes isatoic anhydride or its derivatives, aldehyde compounds, and inert amides as the foundational building blocks. By leveraging a simple acidic catalytic system, this methodology bypasses the cumbersome multi-step sequences traditionally associated with heterocyclic construction, offering a direct route to high-value nitrogen-containing heterocycles. The significance of this technology lies not only in its operational simplicity but also in its exceptional atom economy and the avoidance of toxic reagents, positioning it as a premier candidate for green chemistry applications in modern drug development. For R&D teams focused on accelerating lead optimization, this platform provides a versatile toolkit for generating diverse chemical space around the quinazolinone core with minimal synthetic effort.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone skeleton has relied heavily on the condensation of anthranilamide with aldehydes or their equivalents, a process that often necessitates the prior preparation of the anthranilamide starting material through separate, resource-intensive steps. Alternative strategies have emerged, such as carbonylation cyclization reactions or oxidative cyclizations of anthranilamide derivatives, but these frequently suffer from significant drawbacks including the requirement for expensive transition metal catalysts and harsh oxidative conditions. Many existing protocols demand the use of environmentally unfriendly oxidants and coupling reagents, which complicate downstream purification and generate substantial hazardous waste streams that increase disposal costs. Furthermore, the reliance on sensitive catalysts often mandates strict inert atmosphere conditions, adding complexity and equipment costs to the manufacturing process while limiting the practical scalability for industrial applications. The difficulty in recycling these catalysts and the potential for heavy metal residues in the final active pharmaceutical ingredient (API) remain persistent challenges that hinder the widespread adoption of these older methodologies in regulated environments.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in the patent utilizes a direct three-component coupling that merges isatoic anhydride, an aldehyde, and an inert amide in a single operational step. This strategy effectively streamlines the synthesis by eliminating the need for pre-functionalized precursors, thereby reducing the overall number of unit operations and significantly cutting down on solvent consumption and processing time. The reaction proceeds efficiently under mild thermal conditions ranging from 25°C to 130°C and, remarkably, can be conducted under ambient air conditions without the need for rigorous exclusion of oxygen or moisture. By employing inexpensive and commercially available acidic catalysts, including recoverable supported variants, this method ensures that the final quinazolinone derivatives are obtained with high purity and minimal metal contamination. This paradigm shift represents a major advancement in cost reduction in pharmaceutical intermediates manufacturing, offering a sustainable and economically viable pathway for producing complex heterocyclic scaffolds.
Mechanistic Insights into Acid-Catalyzed Cyclization
The underlying mechanism of this transformation is driven by the activation of electrophilic centers through Brønsted acid catalysis, which facilitates the nucleophilic attack necessary for ring closure. Initially, the acidic catalyst protonates the carbonyl oxygen of the isatoic anhydride or the aldehyde, increasing the electrophilicity of the carbonyl carbon and making it more susceptible to nucleophilic attack by the nitrogen atom of the inert amide. This initial addition is followed by a cascade of intramolecular cyclization events where the amine functionality attacks the activated carbonyl, leading to the formation of the dihydroquinazolinone intermediate. Subsequent dehydration and aromatization steps, driven by the thermodynamic stability of the aromatic quinazolinone system, result in the expulsion of carbon dioxide from the anhydride moiety and the formation of the final double bond within the heterocyclic ring. The choice of solvent, such as N-methylformamide or polar aprotic solvents, plays a crucial role in stabilizing the charged intermediates and ensuring high conversion rates throughout the reaction profile.
From an impurity control perspective, the mild nature of the acidic conditions and the absence of strong oxidants significantly reduce the formation of over-oxidized byproducts or polymerization species that often plague radical-induced tandem reactions. The high functional group tolerance of this catalytic system allows for the presence of sensitive moieties such as halogens, ethers, and heteroaromatic rings without degradation, ensuring a clean impurity profile that simplifies downstream purification. The use of supported catalysts further enhances the purity of the final product by allowing for easy filtration and removal of the catalytic species, thereby preventing metal leaching into the product stream. This level of control is critical for meeting the stringent purity specifications required for high-purity pharmaceutical intermediates, as it minimizes the burden on analytical quality control labs to identify and quantify trace metal impurities. Consequently, this mechanistic pathway offers a reliable and reproducible method for generating clinical-grade materials with consistent quality attributes.
How to Synthesize Quinazolinone Derivatives Efficiently
To implement this synthesis effectively, operators should begin by charging a reaction vessel with the isatoic anhydride derivative, the selected aldehyde, and the inert amide in a molar ratio that favors complete conversion, typically utilizing a slight excess of the aldehyde and amide components. The reaction mixture is then treated with a catalytic amount of an acidic promoter, such as p-toluenesulfonic acid or a supported variant, and heated to the optimal temperature range identified in the patent examples, which generally falls between 100°C and 130°C for optimal kinetics. Following the completion of the reaction, indicated by TLC or HPLC analysis, the solvent is removed under reduced pressure, and the crude residue is subjected to standard purification techniques such as silica gel column chromatography using a petroleum ether and ethyl acetate gradient. Detailed standardized synthesis steps see the guide below.
- Combine isatoic anhydride, aldehyde, and inert amide in a suitable solvent such as N-methylformamide or acetonitrile.
- Add an acidic catalyst, preferably a supported acid like activated carbon-supported p-toluenesulfonic acid, to the reaction mixture.
- Heat the mixture to 25-130°C under air atmosphere for 1-24 hours, then purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic benefits that extend beyond simple yield improvements, fundamentally altering the cost structure of producing quinazolinone-based intermediates. The reliance on commodity chemicals like isatoic anhydride and simple aldehydes ensures a stable and diversified supply base, mitigating the risks associated with sourcing specialized or proprietary starting materials that are subject to market volatility. Furthermore, the elimination of expensive transition metal catalysts and toxic oxidants removes significant cost drivers related to reagent procurement, specialized waste disposal, and extensive purification protocols required to meet residual metal limits. This streamlined process inherently reduces the manufacturing footprint and energy consumption, aligning with corporate sustainability goals while simultaneously driving down the cost of goods sold (COGS) for the final active ingredients.
- Cost Reduction in Manufacturing: The economic advantage of this method is primarily derived from the consolidation of multiple synthetic steps into a single pot, which drastically reduces labor costs, solvent usage, and reactor occupancy time. By avoiding the use of precious metal catalysts and harsh oxidizing agents, the process eliminates the need for costly scavenging resins and complex extraction procedures designed to remove trace metals, leading to substantial cost savings in downstream processing. The high atom economy of the reaction ensures that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste generation and maximizing the efficiency of raw material utilization. Additionally, the ability to use recoverable supported catalysts further enhances the economic viability by allowing for catalyst reuse over multiple batches, amortizing the initial catalyst cost over a larger production volume.
- Enhanced Supply Chain Reliability: The robustness of this chemistry under air conditions and its tolerance to a wide range of functional groups make it highly resilient to variations in raw material quality and environmental factors, ensuring consistent production output. Since the starting materials are bulk chemicals with established global supply chains, the risk of supply disruption due to the scarcity of exotic reagents is significantly minimized, providing greater security of supply for long-term projects. The simplified workup procedure, which often involves basic filtration and crystallization or standard chromatography, reduces the dependency on specialized equipment and highly skilled operators, thereby enhancing the overall flexibility and responsiveness of the manufacturing facility. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for key customers in the pharmaceutical sector.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic hazards associated with strong oxidants and the use of common, non-hazardous solvents. The generation of benign byproducts such as carbon dioxide and water simplifies waste treatment protocols, ensuring compliance with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs) and hazardous waste discharge. The potential for implementing continuous flow chemistry with this robust reaction system further offers opportunities for intensification, allowing for safer and more efficient production at the multi-ton scale. These factors collectively contribute to a sustainable manufacturing model that supports the commercial scale-up of complex heterocyclic scaffolds without compromising on environmental stewardship or regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinazolinone synthesis technology, providing clarity on its operational parameters and strategic value. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this pathway. Understanding these details is essential for assessing the feasibility of integrating this method into existing production workflows and for optimizing the economic outcomes of your chemical manufacturing projects.
Q: What are the key advantages of this quinazolinone synthesis method over traditional routes?
A: This method utilizes a one-step multicomponent reaction involving isatoic anhydride, aldehydes, and amides, eliminating the need for pre-synthesized anthranilamides or harsh oxidative conditions. It offers superior atom economy, avoids toxic oxidants, and operates under mild air conditions, significantly simplifying the workflow.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the process is highly scalable due to the use of readily available raw materials and the absence of sensitive transition metal catalysts. The ability to run the reaction under air atmosphere and the option to use recoverable supported catalysts make it ideal for large-scale commercial manufacturing.
Q: What types of substituents are compatible with this synthetic route?
A: The method exhibits excellent functional group tolerance, accommodating various substituents on the isatoic anhydride (alkyl, alkoxy, halogen) and diverse aldehydes (aromatic, heteroaromatic, aliphatic). This versatility allows for the rapid generation of diverse quinazolinone libraries for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to maintain a competitive edge in the global pharmaceutical market, and we are fully equipped to leverage this patented methodology for your projects. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of quinazolinone derivatives meets the highest standards of quality and consistency required for API synthesis. Our commitment to technical excellence allows us to navigate the complexities of process optimization, delivering solutions that balance performance, cost, and regulatory compliance.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific needs, offering a Customized Cost-Saving Analysis that quantifies the potential economic benefits for your supply chain. By partnering with us, you gain access to specific COA data and route feasibility assessments that empower you to make informed decisions about your sourcing strategy. Contact us today to explore how we can support your development goals with reliable, high-quality quinazolinone intermediates produced through this cutting-edge, sustainable technology.
