Advanced Pd-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Pharmaceutical Applications
Advanced Pd-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. A significant breakthrough in this domain is detailed in patent CN112480015A, which discloses a highly efficient, multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. This class of compounds is renowned for its pervasive presence in bioactive molecules, exhibiting a broad spectrum of pharmacological activities including antifungal, antibacterial, antiviral, and anticancer properties. The introduction of the trifluoromethyl group is particularly strategic, as it significantly enhances the metabolic stability, lipophilicity, and bioavailability of the parent molecule, making these intermediates invaluable for modern drug discovery pipelines. The disclosed technology represents a paradigm shift from traditional, hazardous synthetic routes to a safer, more atom-economical process that leverages transition metal palladium catalysis.
For R&D directors and process chemists, the ability to access these privileged structures through a streamlined protocol is a major advantage. The patent outlines a reaction system that integrates a palladium catalyst, a specific phosphine ligand, a carbon monoxide substitute, and an additive to facilitate the transformation of readily available starting materials into high-value quinazolinone derivatives. This approach not only simplifies the operational workflow but also addresses critical safety concerns associated with handling gaseous carbon monoxide directly. By utilizing solid carbonyl sources and inexpensive nitro compounds, the method lowers the barrier to entry for producing these complex heterocycles, thereby accelerating the timeline from bench-scale discovery to pilot plant production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has been fraught with synthetic challenges that hinder efficient commercial manufacturing. Traditional pathways often rely on the use of toxic and hazardous gaseous carbon monoxide under high-pressure conditions, which necessitates specialized equipment and rigorous safety protocols that drive up capital expenditure. Furthermore, many established methods employ expensive noble metal catalysts such as ruthenium or platinum, which not only increase the raw material costs but also introduce complexities regarding residual metal removal—a critical quality attribute for pharmaceutical intermediates. Other conventional routes involve multi-step sequences requiring pre-activation of substrates or the use of unstable intermediates, leading to lower overall yields and increased waste generation. These limitations create bottlenecks in the supply chain, resulting in longer lead times and higher costs for the final active pharmaceutical ingredients (APIs).
The Novel Approach
In stark contrast, the methodology described in patent CN112480015A offers a transformative solution by employing a palladium-catalyzed carbonylation serial reaction that proceeds in a single pot. This innovative route utilizes trifluoroethylimidoyl chloride and nitro compounds as the primary building blocks, both of which are commercially abundant and cost-effective. The reaction巧妙地 replaces dangerous gaseous CO with molybdenum hexacarbonyl (Mo(CO)6), a solid surrogate that releases carbon monoxide in situ under heating, thereby eliminating the need for high-pressure reactors. This shift not only enhances operational safety but also improves the reaction's tolerance to various functional groups, allowing for the synthesis of a diverse library of substituted quinazolinones. The simplicity of the work-up procedure, involving basic filtration and chromatography, further underscores the practical utility of this method for industrial applications.
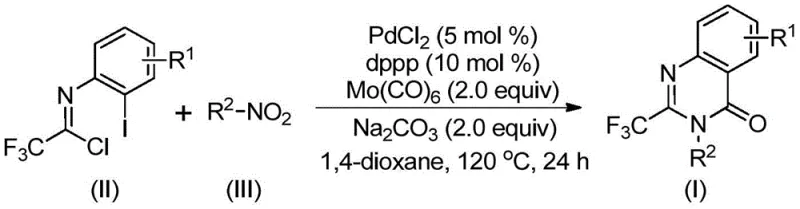
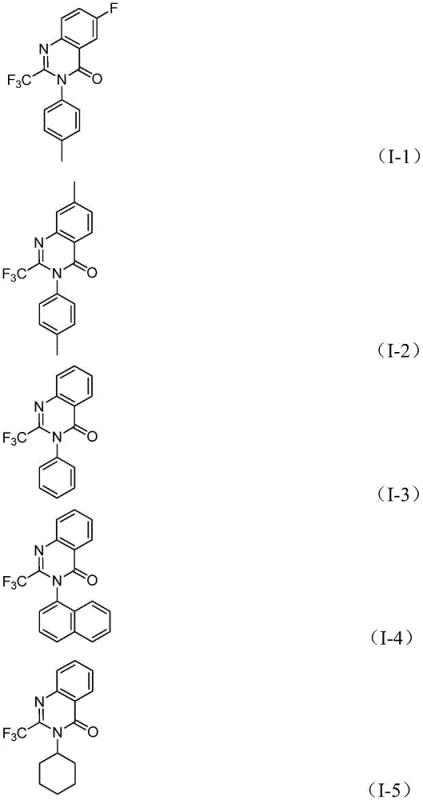
The versatility of this new approach is evident in its broad substrate scope. As demonstrated in the patent examples, the reaction accommodates a wide array of substituents on both the aromatic ring of the nitro compound and the imidoyl chloride component. Whether the substrate bears electron-withdrawing groups like halogens and trifluoromethyl moieties or electron-donating alkyl groups, the catalytic system maintains high efficiency. This flexibility is crucial for medicinal chemists who need to rapidly iterate on molecular structures to optimize biological activity. By enabling the direct conversion of nitro compounds—which are often cheaper and more stable than their amine counterparts—this method provides a distinct economic advantage over routes that require prior reduction steps.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
Understanding the mechanistic underpinnings of this transformation is essential for optimizing the process and ensuring consistent product quality. The reaction is believed to initiate with the reduction of the nitro compound to the corresponding amine by molybdenum hexacarbonyl, which serves a dual role as both a CO source and a reducing agent. Once the amine is generated in situ, it undergoes an alkali-promoted coupling with the trifluoroethylimidoyl chloride to form a trifluoroacetamidine derivative. This intermediate then enters the palladium catalytic cycle, where the Pd(0) species inserts into the carbon-iodine bond of the imidoyl chloride moiety (or the aryl iodide if present in the specific substrate design), forming a divalent palladium intermediate. Subsequently, the CO released from the thermal decomposition of Mo(CO)6 inserts into the carbon-palladium bond, generating an acyl palladium species.
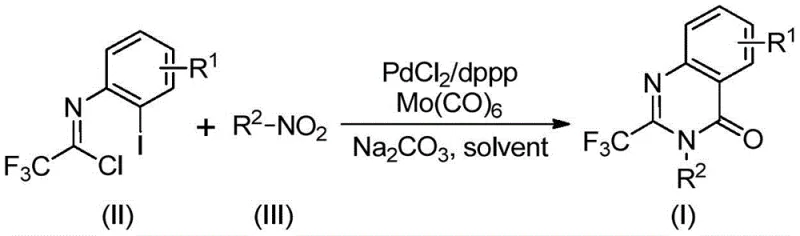
The cyclization event is driven by the formation of a palladium-nitrogen bond under basic conditions, leading to a seven-membered ring palladium intermediate. The final step involves reductive elimination, which releases the desired 2-trifluoromethyl-substituted quinazolinone product and regenerates the active Pd(0) catalyst to continue the cycle. This intricate dance of organometallic steps is finely balanced by the choice of ligand, specifically 1,3-bis(diphenylphosphino)propane (dppp), which stabilizes the palladium center and facilitates the necessary oxidative addition and reductive elimination steps. For R&D teams, controlling the stoichiometry of the base (sodium carbonate) and the temperature (optimized at 120°C) is critical to minimizing side reactions and ensuring high purity. The mechanism explains why the reaction is tolerant to various functional groups; the mild conditions prevent the degradation of sensitive moieties that might occur under harsher acidic or high-pressure environments.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The execution of this synthesis requires precise attention to reagent quality and reaction parameters to achieve the high yields reported in the patent literature. The process is designed to be user-friendly, utilizing standard laboratory glassware such as Schlenk tubes for small-scale optimization, which can be readily translated to larger reactor vessels. The key to success lies in the homogeneous mixing of the palladium catalyst, ligand, and carbonyl source before the addition of the organic substrates. Maintaining an inert atmosphere is generally recommended to protect the catalyst, although the specific robustness of this system allows for some flexibility. The reaction time, typically ranging from 16 to 30 hours, ensures complete conversion of the starting materials, which is vital for maximizing throughput in a manufacturing setting.
- Mix palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and a nitro compound in an organic solvent like dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours to allow for carbonylation and cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final quinazolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers compelling strategic benefits that extend beyond mere technical feasibility. The primary driver for cost reduction is the substitution of expensive and hazardous reagents with commodity chemicals. Nitro compounds are produced on a massive industrial scale for various applications, making them significantly cheaper and more readily available than specialized amines or anhydrides required by other methods. Furthermore, the elimination of high-pressure carbon monoxide infrastructure reduces the capital intensity of the manufacturing facility, allowing for production in standard multipurpose plants. This flexibility enhances supply chain resilience, as manufacturers are not bottlenecked by specialized equipment availability or stringent safety regulations associated with high-pressure gas handling.
- Cost Reduction in Manufacturing: The economic profile of this process is substantially improved by the use of earth-abundant palladium catalysts in conjunction with inexpensive ligands, replacing costly ruthenium or platinum systems. Additionally, the use of Mo(CO)6 as a solid CO source eliminates the logistical costs and safety premiums associated with transporting and storing high-pressure gas cylinders. The high atom economy of the one-pot design means fewer unit operations, less solvent consumption, and reduced energy usage for heating and cooling cycles between steps. These factors collectively contribute to a lower cost of goods sold (COGS), providing a competitive edge in pricing for the final pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing reliability is paramount for continuous manufacturing operations. Since the key starting materials—trifluoroethylimidoyl chloride and various nitroarenes—are widely produced by multiple global suppliers, the risk of supply disruption is minimized. The robustness of the reaction conditions also means that variations in raw material quality (within specification) are less likely to cause batch failures, ensuring consistent output. This stability allows supply chain managers to forecast production schedules with greater accuracy and maintain leaner inventory levels without compromising on delivery commitments to downstream API manufacturers.
- Scalability and Environmental Compliance: The transition from gram-scale laboratory synthesis to kilogram or ton-scale production is facilitated by the simplicity of the work-up procedure. The absence of toxic heavy metal residues (common with Ru/Pt catalysts) simplifies the purification process, reducing the burden on waste treatment facilities. The reaction generates fewer by-products compared to multi-step alternatives, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. This compliance with increasingly stringent environmental regulations future-proofs the supply chain against potential regulatory crackdowns on hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when integrating this chemistry into their operations. Understanding these nuances helps in making informed decisions regarding process adoption and resource allocation.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method avoids harsh high-pressure carbon monoxide conditions and expensive ruthenium or platinum catalysts. It utilizes cheap, readily available nitro compounds and operates under atmospheric pressure using Mo(CO)6 as a safe CO surrogate.
Q: What is the substrate scope for this reaction?
A: The reaction demonstrates excellent compatibility with various functional groups including halogens (F, Cl, Br), alkyl groups (methyl), and trifluoromethyl groups on both the nitro compound and the imidoyl chloride components.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states the method can be expanded to the gram level and is designed for large-scale industrial application due to simple operation and easy post-treatment procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team of expert process chemists has thoroughly analyzed the methodology described in patent CN112480015A and is fully equipped to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. Our state-of-the-art facilities are designed to handle complex organometallic reactions safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest international standards.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Whether you require custom synthesis of novel derivatives or large-scale supply of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in palladium-catalyzed transformations can accelerate your timeline and reduce your overall development costs.
