Scalable Synthesis of 5-Chloro-4-Aminopyridazine: A Breakthrough in High-Purity Heterocyclic Manufacturing
Scalable Synthesis of 5-Chloro-4-Aminopyridazine: A Breakthrough in High-Purity Heterocyclic Manufacturing
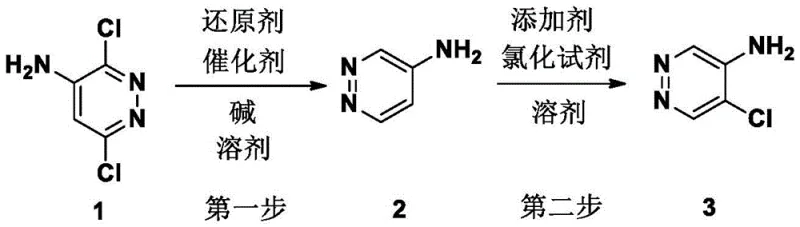
The pharmaceutical and agrochemical industries constantly demand access to complex heterocyclic building blocks that possess high purity and structural integrity. A significant advancement in this domain is detailed in patent CN110903248B, which discloses a robust and economically viable synthesis method for 5-chloro-4-aminopyridazine. This compound serves as a critical intermediate for constructing biologically active molecules with potential anticancer, antiviral, and cardiovascular applications. The disclosed technology addresses long-standing challenges in pyridazine chemistry, specifically regarding regioselectivity and process safety. By utilizing 3,6-dichloro-4-aminopyridazine as a readily available starting material, the process achieves a high-yield transformation through a strategic two-step sequence involving catalytic hydrodechlorination followed by controlled radical chlorination. This approach not only simplifies the synthetic pathway but also ensures that the final product meets the stringent quality standards required for commercial mass production of active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
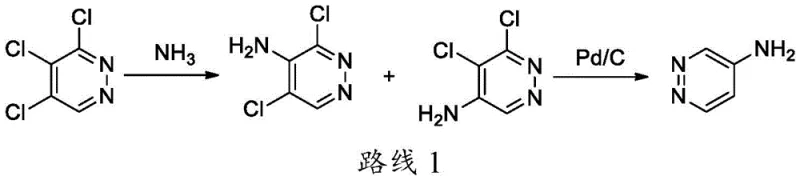
Historically, the synthesis of aminopyridazine derivatives has been plagued by significant technical hurdles that hinder large-scale manufacturing. One traditional route, often referred to as Route 1 in technical literature, involves the ammoniation of 3,4,5-trichloropyridazine. This method suffers from inherent selectivity issues because the three chlorine atoms on the pyridazine ring exhibit similar reactivity profiles. Consequently, the reaction yields a complex mixture of isomers, including 3,5-dichloro-4-aminopyridazine and 5,6-dichloro-4-aminopyridazine, alongside various diamino impurities. Separating these structurally similar byproducts is notoriously difficult and often requires extensive purification steps that drastically reduce overall yield. Furthermore, another historical approach, known as Route 2, relies on the Chichibabin reaction using sodium amide. This method presents severe safety liabilities due to the instability of metal amino mixtures, which are prone to absorbing moisture and carbon dioxide from the air, potentially forming explosive mixtures. These conventional pathways are ill-suited for modern industrial requirements where safety, purity, and cost-efficiency are paramount.
The Novel Approach
In stark contrast to the problematic legacy methods, the novel synthesis strategy outlined in the patent offers a streamlined and highly selective alternative. The process begins with the selective hydrodechlorination of 3,6-dichloro-4-aminopyridazine, a commercially accessible and inexpensive raw material. Under mild conditions using a palladium catalyst and a base in an alcoholic solvent, the chlorine atoms at the 3 and 6 positions are removed to generate the key intermediate, 4-aminopyridazine, with exceptional yield. The second step involves a meticulously controlled chlorination reaction. Unlike previous attempts that resulted in poor selectivity, this method employs N-chlorosuccinimide (NCS) in the presence of a catalytic amount of a radical initiator. This specific combination allows for the precise introduction of a chlorine atom at the 5-position of the pyridazine ring. The result is a clean reaction profile that avoids the formation of the undesirable 3-chloro isomer, thereby enabling purification through simple recrystallization rather than complex chromatographic techniques.
Mechanistic Insights into Radical-Mediated Regioselective Chlorination
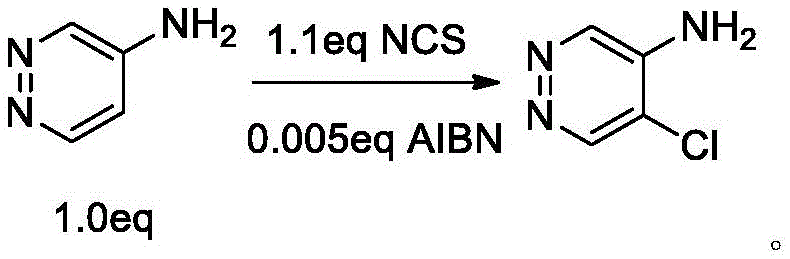
The core innovation of this synthesis lies in the mechanistic understanding of the chlorination step, which overcomes the electronic challenges inherent to the pyridazine ring system. The pyridazine nucleus is electron-deficient, making electrophilic substitution difficult, yet the presence of the electron-donating amino group at the 4-position activates the ring towards substitution. However, the amino group is a strong ortho-para director, which theoretically favors substitution at the 3 and 5 positions. In practice, without specific control, the reaction tends to produce a mixture of 3-chloro and 5-chloro isomers, which have very similar polarities and physical properties, making separation a nightmare for process chemists. The patent reveals that by employing a radical mechanism initiated by azobisisobutyronitrile (AIBN) at low temperatures, the reaction pathway is steered decisively towards the 5-position. The radical species generated interact with the substrate in a manner that kinetically favors the formation of the 5-chloro derivative, effectively suppressing the formation of the 3-chloro byproduct. This level of control is critical for maintaining high purity without resorting to preparative HPLC or flash chromatography.
Furthermore, the impurity control mechanism is reinforced by the choice of reaction conditions and quenching procedures. The process dictates a slow addition of the chlorinating agent at temperatures between 0°C and 5°C, which helps manage the exothermic nature of the reaction and prevents thermal runaway that could lead to over-chlorination or decomposition. Following the reaction, the immediate quenching with saturated ammonium chloride solution effectively neutralizes any remaining reactive species. The subsequent workup involves extraction and washing with saturated sodium carbonate and saline solutions, which removes acidic byproducts and succinimide residues. The final purification via recrystallization from an ethyl acetate and methanol mixture ensures that trace impurities are left behind in the mother liquor. This robust impurity profile is essential for downstream applications, as residual halogenated impurities can interfere with subsequent coupling reactions in drug synthesis, potentially affecting the efficacy and safety of the final therapeutic agent.
How to Synthesize 5-Chloro-4-Aminopyridazine Efficiently
The execution of this synthesis requires careful attention to stoichiometry and environmental controls to replicate the high yields reported in the patent examples. The process is designed to be operationally simple, utilizing standard equipment found in most fine chemical manufacturing facilities. The first step utilizes a hydrogenation kettle capable of handling pressure and inert atmospheres, while the second step requires a reactor equipped with precise temperature control and dropping funnels for reagent addition. The following guide outlines the critical operational parameters derived from the patent data to ensure successful replication of the process. For the complete standardized operating procedure and safety protocols, please refer to the detailed synthesis steps provided below.
- Perform hydrodechlorination of 3,6-dichloro-4-aminopyridazine using Pd/C catalyst and base in ethanol under hydrogen atmosphere to obtain 4-aminopyridazine.
- Dissolve the intermediate 4-aminopyridazine in acetonitrile and cool to 0-5°C.
- Add AIBN initiator and slowly drip N-chlorosuccinimide (NCS) solution while maintaining low temperature to ensure 5-position selectivity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented synthesis route offers transformative advantages that directly impact the bottom line and operational reliability. Traditional methods for synthesizing pyridazine derivatives often rely on exotic starting materials or hazardous reagents that complicate logistics and increase inventory costs. By shifting to a process based on 3,6-dichloro-4-aminopyridazine and N-chlorosuccinimide, manufacturers can leverage a supply chain built on commodity chemicals that are widely available from multiple global vendors. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. Moreover, the elimination of dangerous reagents like sodium amide reduces the regulatory burden and insurance costs associated with handling explosive materials, creating a safer and more compliant manufacturing environment.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the purification workflow. In conventional organic synthesis, the removal of isomeric impurities often necessitates column chromatography, a technique that is prohibitively expensive and slow at an industrial scale due to high solvent consumption and silica gel costs. This novel method achieves high selectivity through reaction engineering rather than downstream purification, allowing for product isolation via crystallization. This shift significantly reduces solvent usage and waste generation, leading to substantial cost savings in both material procurement and waste disposal. Additionally, the use of low-loading palladium catalysts and recyclable solvents like ethanol and acetonitrile further optimizes the cost structure, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the stability and shelf-life of the reagents employed in this synthesis. Unlike moisture-sensitive reagents that require specialized storage and transportation, the key inputs for this process are stable solids and liquids that can be stocked in bulk without degradation. This stability allows for better inventory planning and reduces the lead time associated with sourcing specialty chemicals. The robustness of the reaction conditions, which tolerate mild temperature ranges and standard atmospheric pressures, also means that the process can be transferred between different manufacturing sites with minimal re-validation effort. This flexibility ensures continuous supply continuity even if one production facility faces unforeseen operational challenges.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often introduces new variables that can compromise yield or safety. This synthesis is inherently scalable because it avoids extreme conditions such as cryogenic temperatures or high-pressure autoclaves beyond standard hydrogenation capabilities. The exothermic chlorination step is managed through controlled dosing, a technique that translates easily from liter-scale to cubic-meter-scale reactors. From an environmental standpoint, the process aligns with green chemistry principles by minimizing the use of chlorinated solvents in the first step and avoiding the generation of heavy metal waste streams associated with stoichiometric metal reagents. The ability to recycle solvents and the high atom economy of the transformation contribute to a lower environmental footprint, facilitating easier compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 5-chloro-4-aminopyridazine. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is crucial for process development teams evaluating the feasibility of integrating this intermediate into their own synthetic pipelines. The responses cover aspects of selectivity, safety, and purification to provide a comprehensive overview of the technology's capabilities.
Q: Why is the AIBN additive critical in the chlorination step?
A: Without the radical initiator AIBN, direct chlorination of 4-aminopyridazine lacks regioselectivity, producing significant amounts of the unwanted 3-chloro isomer. The additive ensures high selectivity for the 5-position.
Q: What are the advantages of this route over the Chichibabin reaction?
A: Unlike the Chichibabin reaction which uses hazardous sodium amide and poses explosion risks, this method utilizes stable, commercially available reagents under mild conditions, significantly improving operational safety.
Q: Is column chromatography required for purification?
A: No. A major advantage of this patented process is that the final product can be purified via simple recrystallization, eliminating the need for expensive and time-consuming column chromatography suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chloro-4-Aminopyridazine Supplier
The technical potential of this synthesis route represents a significant opportunity for pharmaceutical companies seeking to optimize their supply chains for pyridazine-based therapeutics. NINGBO INNO PHARMCHEM stands ready to support your development needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including hydrogenation capabilities and precise temperature-controlled reactors. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 5-chloro-4-aminopyridazine meets the highest international standards for pharmaceutical intermediates. Our commitment to quality assurance guarantees that the material you receive is consistent, reliable, and ready for immediate use in your downstream synthesis.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can benefit your specific project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic impact of switching to this superior manufacturing route. We encourage you to contact us today to request specific COA data and route feasibility assessments tailored to your volume needs. Let us help you secure a stable and cost-effective supply of this critical building block for your next generation of innovative medicines.
