Scalable Synthesis of 5-Chloro-4-Aminopyridazine: A High-Yield Route for Pharmaceutical Intermediates
Scalable Synthesis of 5-Chloro-4-Aminopyridazine: A High-Yield Route for Pharmaceutical Intermediates
The development of efficient synthetic routes for heterocyclic building blocks remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activities such as anticancer and antiviral properties. Patent CN110903248A introduces a groundbreaking methodology for the synthesis of 5-chloro-4-aminopyridazine, a critical intermediate that has historically posed significant challenges regarding regioselectivity and scalability. This novel approach utilizes 3,6-dichloro-4-aminopyridazine as a commercially accessible starting material, subjecting it to a controlled hydrodechlorination followed by a highly selective chlorination step. By leveraging specific catalytic systems and radical initiators, the process overcomes the limitations of prior art, delivering high-purity products suitable for large-scale industrial application without the need for hazardous reagents or complex purification techniques.
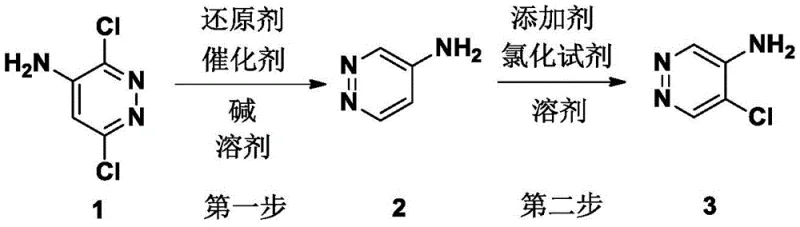
The strategic value of this patent lies in its ability to transform a difficult multi-step problem into a streamlined, two-stage operation that maximizes atom economy and minimizes waste generation. For R&D directors and process chemists, the availability of such a robust protocol means faster timeline-to-market for new drug candidates relying on the pyridazine core. The method ensures that the key intermediate, 4-aminopyridazine, is generated in high yield before undergoing a precise functionalization at the 5-position. This level of control is essential for maintaining strict impurity profiles required by regulatory bodies, making this technology a vital asset for any organization seeking a reliable pharma intermediate supplier capable of meeting rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aminopyridazine derivatives has been plagued by issues related to regioselectivity and the use of dangerous reagents. One prominent conventional route, often cited in literature, involves the ammoniation of 3,4,5-trichloropyridazine. As illustrated in the reaction pathway below, this method suffers from the fact that the three chlorine atoms possess similar reactivity, leading to a complex mixture of isomers including 3,5-dichloro-4-aminopyridazine and 5,6-dichloro-4-aminopyridazine.
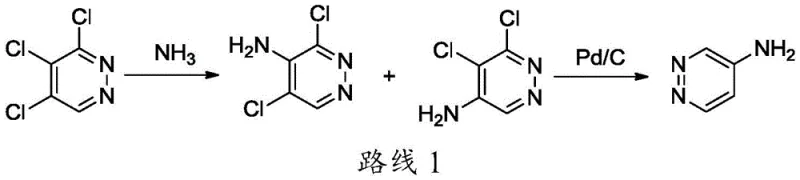
The formation of these structural isomers creates a nightmare for downstream processing, as their physical properties, such as polarity and boiling points, are remarkably similar, making separation via standard crystallization or distillation nearly impossible. Consequently, manufacturers are often forced to resort to preparative column chromatography, a technique that is prohibitively expensive and impractical for commercial scale-up. Furthermore, alternative routes like the Chichibabin reaction utilize sodium amide, a pyrophoric substance that forms explosive mixtures upon exposure to air or moisture, presenting unacceptable safety risks for large-volume production facilities. These inherent flaws in traditional methodologies result in low overall yields, inconsistent batch quality, and inflated production costs.
The Novel Approach
In stark contrast, the methodology disclosed in CN110903248A circumvents these pitfalls by selecting 3,6-dichloro-4-aminopyridazine as the precursor, a material that is both inexpensive and readily available on the global market. The first step employs a mild catalytic hydrodechlorination using palladium on carbon (Pd/C) and a base like sodium hydroxide in an alcohol solvent. This reaction cleanly removes the chlorine atoms at the 3 and 6 positions without affecting the amino group or the aromatic ring integrity, yielding 4-aminopyridazine with exceptional purity. The second step introduces a clever modification to the chlorination process by incorporating a radical initiator, such as azobisisobutyronitrile (AIBN), alongside N-chlorosuccinimide (NCS). This addition fundamentally alters the reaction mechanism, steering the chlorination exclusively to the 5-position and effectively suppressing the formation of the unwanted 3-chloro isomer. This strategic innovation transforms a low-selectivity reaction into a precise synthetic tool.
Mechanistic Insights into Radical-Mediated Selective Chlorination
The success of the second step in this synthesis hinges on a sophisticated understanding of radical chemistry and electronic effects within the pyridazine ring system. The amino group at the 4-position is a strong electron-donating group that activates the ring towards electrophilic attack, specifically at the ortho positions (3 and 5). In the absence of a radical initiator, the reaction between 4-aminopyridazine and NCS proceeds primarily through an electrophilic pathway that lacks sufficient discrimination between the two ortho sites, resulting in a statistical mixture of products. However, the introduction of AIBN changes the paradigm by generating free radicals at elevated temperatures or even at room temperature over time. These radicals facilitate a homolytic cleavage of the N-Cl bond in NCS, generating chlorine radicals that interact with the substrate differently than electrophilic chlorine species.
This radical-mediated pathway appears to favor the thermodynamic or kinetic stability of the transition state leading to the 5-chloro product, possibly due to steric factors or specific interactions with the nitrogen atoms in the pyridazine ring that are less pronounced in the electrophilic pathway. By maintaining the reaction temperature between 0°C and 25°C and carefully controlling the addition rate of the chlorinating agent, the process ensures that the radical concentration remains optimal for selective substitution. This mechanistic control is crucial for impurity management; it prevents the formation of the 3-chloro-4-aminopyridazine byproduct, which, as noted in the patent background, has polarity and properties so close to the target molecule that separation would otherwise require laborious chromatographic methods. The result is a crude product of such high purity that simple recrystallization suffices for final isolation.
How to Synthesize 5-Chloro-4-Aminopyridazine Efficiently
The operational simplicity of this patented process makes it highly attractive for technology transfer and scale-up. The procedure begins with the dissolution of the starting material in a green solvent like ethanol, followed by the addition of the catalyst and base under an inert atmosphere. After the hydrodechlorination is complete, the intermediate is isolated and immediately subjected to the chlorination conditions in acetonitrile. The detailed standardized synthesis steps, including precise molar ratios, temperature profiles, and workup procedures, are outlined in the guide below to ensure reproducibility and safety during implementation.
- Perform hydrodechlorination of 3,6-dichloro-4-aminopyridazine using Pd/C catalyst and NaOH in ethanol under hydrogen atmosphere to obtain 4-aminopyridazine.
- Dissolve the intermediate 4-aminopyridazine in acetonitrile and cool to 0-5°C, adding a catalytic amount of AIBN initiator.
- Slowly add N-chlorosuccinimide (NCS) solution while maintaining low temperature, then warm to room temperature to achieve selective 5-position chlorination.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route offers profound strategic benefits that extend beyond mere chemical yield. The shift away from hazardous reagents like sodium amide and toxic chlorine gas significantly reduces the regulatory burden and insurance costs associated with chemical manufacturing. By utilizing stable, solid reagents such as NCS and AIBN, the process eliminates the need for specialized containment equipment required for handling pressurized gases or pyrophoric solids, thereby simplifying facility requirements and enhancing operational safety. This reduction in hazard profile directly translates to a more resilient supply chain, as the risk of production stoppages due to safety incidents or regulatory compliance issues is drastically minimized.
- Cost Reduction in Manufacturing: The economic implications of this route are substantial, driven primarily by the elimination of expensive purification steps. Traditional methods often necessitate column chromatography to separate isomeric impurities, a process that consumes vast quantities of silica gel and solvents while offering low throughput. By achieving high regioselectivity through the use of the AIBN additive, this new method allows for purification via simple recrystallization, which is exponentially cheaper and faster. Furthermore, the starting material, 3,6-dichloro-4-aminopyridazine, is a commodity chemical with a stable market price, avoiding the volatility associated with custom-synthesized precursors. The ability to recycle solvents like acetonitrile and ethanol further drives down the variable costs per kilogram, ensuring a competitive pricing structure for the final API intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on niche reagents or complex multi-step sequences with low overall yields. This two-step process boasts a high cumulative yield, meaning less raw material is required to produce the same amount of finished goods, effectively buffering against upstream supply shocks. The robustness of the reaction conditions, which tolerate mild temperatures and standard atmospheric pressure, ensures that production can be maintained consistently across different manufacturing sites without the need for highly specialized engineering controls. This flexibility allows for diversified sourcing strategies, enabling companies to qualify multiple contract manufacturing organizations (CMOs) without significant retooling investments.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the ability to demonstrate a "green" manufacturing process is a key differentiator. This synthesis avoids the generation of heavy metal waste associated with stoichiometric metal reagents and reduces the volume of organic waste by eliminating chromatography. The use of catalytic amounts of palladium, which can be recovered and reused, aligns with principles of sustainable chemistry. Scaling this process from pilot plant to commercial tonnage is straightforward because the exothermic nature of the reactions is manageable under the described conditions, preventing thermal runaways that often plague scale-up efforts. This ease of scale-up ensures that lead times for high-purity pharmaceutical intermediates can be met reliably, even as demand surges.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy routes in terms of safety, purity, and efficiency. Understanding these nuances is critical for stakeholders evaluating the feasibility of integrating this intermediate into their existing supply chains.
Q: Why is the AIBN additive critical in the chlorination step?
A: The amino group on the pyridazine ring activates both ortho positions (3 and 5). Without the radical initiator AIBN, chlorination with NCS lacks selectivity, producing significant amounts of the unwanted 3-chloro isomer which is difficult to separate. AIBN promotes a radical mechanism that favors the 5-position, drastically improving purity.
Q: What are the safety advantages of this new route compared to the Chichibabin reaction?
A: Traditional routes often utilize sodium amide (NaNH2) in liquid ammonia, which poses severe explosion risks and requires cryogenic conditions. This patented method operates at mild temperatures (0-25°C) using stable reagents like NCS and Pd/C, eliminating the handling of pyrophoric materials and high-pressure ammonia.
Q: How does this process improve purification efficiency?
A: Conventional methods often result in complex isomeric mixtures requiring energy-intensive column chromatography. This optimized route achieves high regioselectivity, allowing the final product to be purified simply through recrystallization from ethyl acetate and methanol, significantly reducing solvent consumption and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chloro-4-Aminopyridazine Supplier
The technological advancements detailed in patent CN110903248A represent a significant leap forward in the manufacturing of pyridazine-based building blocks, offering a pathway to higher purity and lower environmental impact. NINGBO INNO PHARMCHEM stands at the forefront of adopting such innovative processes, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch of 5-chloro-4-aminopyridazine meets the exacting standards required for global pharmaceutical applications. We understand that consistency is key in drug development, and our state-of-the-art facilities are designed to deliver that reliability.
We invite potential partners to engage with our technical team to explore how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this superior manufacturing method. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your supply chain is built on the most robust and efficient chemical foundations available in the market.
